
Please set your exam date
Congenital heart diseases: disorders with decreased pulmonary blood flow
Study Questions
Practice Exercise 1
A nurse is attending a staff education program on congenital heart defects. Which of the following cardiac anomaly produces a left-to-right shunt?
Explanation
Left-to-right shunts occur when a structural opening allows oxygenated blood from the high-pressure systemic circulation to recirculate into the lower-pressure pulmonary circulation. This hemodynamic shift increases pulmonary blood flow, leading to pulmonary hypertension and potential right-sided heart failure. Prolonged exposure to high flow causes irreversible vascular remodeling and may eventually lead to shunt reversal, a phenomenon known as Eisenmenger syndrome.
Rationale for correct answer:
A. Atrial septal defect involves a persistent opening in the interatrial septum, typically at the ostium secundum. Because left atrial pressure exceeds right atrial pressure, blood flows left-to-right, causing volume overload of the right ventricle and pulmonary artery. This defect is often asymptomatic in early childhood but leads to exercise intolerance and arrhythmias in adulthood.
Rationale for incorrect answers:
B. Pulmonic stenosis is an obstructive lesion that restricts blood flow from the right ventricle to the pulmonary artery. It does not create a shunt between the systemic and pulmonary circuits but rather increases afterload on the right ventricle. Over time, this pressure overload results in concentric right ventricular hypertrophy and potential right heart failure.
C. Tetralogy of Fallot is a cyanotic heart defect characterized by a right-to-left shunt through a large ventricular septal defect. The combination of pulmonary stenosis and right ventricular hypertrophy elevates right-sided pressures above systemic levels, forcing deoxygenated blood into the aorta. This results in systemic hypoxemia and the classic clinical presentation of bluish skin and hypercyanotic spells.
D. Total anomalous pulmonary venous return is a complex cyanotic defect where pulmonary veins fail to connect to the left atrium. All oxygenated pulmonary venous return drains into the right atrium, creating a total mixing of systemic and pulmonary blood. This condition requires an associated right-to-left shunt, like a patent foramen ovale, for systemic cardiac output to occur.
Test-taking strategy:
- Categorize congenital heart defects into right-to-left and left-to-right groups based on the direction of blood flow.
- Analyze the pressure gradients of the heart chambers; blood naturally moves from areas of high pressure (left side) to low pressure (right side) unless an obstruction exists.
- Identify defects with the prefix "A" or "V" (atrial, ventricular, atrioventricular) as typical candidates for left-to-right shunting in the absence of other complications.
- Rule out obstructive lesions like stenosis or coarctation, which represent flow resistance rather than a septal communication or shunt.
- Recognize that "Total" or "Transposition" defects usually involve mixing or right-to-left shunting and typically present with immediate neonatal cyanosis.
- Associate the term "shunt" specifically with a pathway between the two sides of the heart or the great vessels.
Take home points
- Defects like ASD, VSD, and PDA are characterized by increased pulmonary blood flow due to left-to-right shunting.
- Defects that cause cyanosis often involve decreased pulmonary blood flow or mixed blood flow, resulting in decreased systemic oxygen saturation.
- Chronic left-to-right shunts can lead to Eisenmenger syndrome, where pulmonary resistance exceeds systemic resistance, reversing the shunt.
- The primary clinical concern in large left-to-right shunts is the development of congestive heart failure and pulmonary overcirculation.
A child with an atrial septal defect repair is entering postoperative day C. Which intervention would be most appropriate?
Explanation
Postoperative management of pediatric cardiac surgery necessitates effective multimodal analgesia to ensure respiratory compliance and hemodynamic stability. Adequate pain control prevents the sympathoadrenal response, which can increase myocardial oxygen demand and induce tachycardia in a recovering heart. Pediatric patients often require a combination of intravenous opioids and non-opioid medications, transitioned to oral routes as gastric motility returns following the cessation of anesthesia.
Rationale for correct answer:
D. Administering analgesics is the priority on postoperative day 3 to facilitate early mobilization and deep breathing exercises. Effective pain management prevents atelectasis and pneumonia by allowing the child to cough and clear secretions without significant chest wall discomfort. Validated pediatric pain scales must be used to titrate medication appropriately to the child’s developmental level and reported pain intensity.
Rationale for incorrect answers:
A. By postoperative day 3, most children have already progressed from clear liquids to a regular diet as tolerated. Maintaining a status of nothing by mouth would be inappropriate unless the patient developed a complication like paralytic ileus or required re-intubation. Adequate nutritional intake is essential for surgical wound healing and providing the metabolic energy required for the recovery process.
B. Strict bed rest is contraindicated by the third postoperative day because it increases the risk of thromboembolism and pulmonary complications. Nurses should encourage progressive ambulation to improve circulation and promote the return of normal bowel and bladder function. Sitting in a chair or walking short distances in the hallway helps maintain muscle tone and psychological well-being during the recovery phase.
C. Taking vital signs only every 8 hours is insufficient for a patient who is only 72 hours post-cardiac sternotomy or thoracotomy. Standard postoperative protocols usually require assessments every 2 to 4 hours to monitor for late complications such as pericardial effusion or rhythm disturbances. Frequent monitoring ensures that any subtle changes in perfusion or blood pressure are detected and addressed by the clinical team immediately.
Test-taking strategy:
- Apply the nursing process by identifying which intervention directly addresses the patient's physiological need for comfort and functional recovery on day C.
- Utilize the principle of early mobilization; in postoperative scenarios, any answer choice advocating for prolonged immobility or strict bed rest is usually incorrect.
- Evaluate the timeline provided in the question; day 1 interventions (like NPO or frequent vitals) are typically relaxed by day 3 in an uncomplicated recovery.
- Recognize that pain management is a prerequisite for other nursing interventions, such as incentive spirometry and physical therapy.
- Rule out options that represent substandard care, such as infrequent vital sign monitoring for a high-acuity surgical patient.
- Prioritize interventions that promote the prevention of complications, as pain-induced shallow breathing is a primary risk factor for respiratory failure.
Take home points
- Pain management in the postoperative pediatric patient is essential to prevent respiratory splinting and promote early ambulation.
- Progression of diet and activity is expected by postoperative day 3 in most uncomplicated atrial septal defect repairs.
- Nursing assessments must remain frequent enough to catch post-pericardiotomy syndrome or other delayed cardiac complications.
- Developmental age-appropriate tools should be used to assess pain intensity rather than relying solely on vital sign changes.
A nurse is caring for a child with an untreated atrial septal defect. Which findings may be present? Select all that apply
Explanation
Atrial septal defect is a congenital cardiac lesion characterized by a persistent opening in the interatrial septum, allowing communication between the systemic and pulmonary circulations. This structural defect results in a left-to-right shunt, leading to chronic volume overload of the right heart chambers and pulmonary overcirculation. If left untreated, the increased pulmonary venous return and structural remodeling can predispose the patient to atrial dysrhythmias and progressive heart failure.
Rationale for correct answers:
A. Recurrent respiratory infections occur due to pulmonary congestion and interstitial edema caused by increased pulmonary blood flow. The excess fluid in the lung tissue provides a medium for bacterial growth and impairs mucociliary clearance, making the child highly susceptible to pneumonia and bronchitis. This increased pulmonary vascularity is a hallmark finding on chest radiography in patients with significant left-to-right shunts.
B. Right atrial enlargement is a direct consequence of the volume overload shifting from the higher-pressure left atrium through the septal opening. The right atrium must accommodate both the normal systemic venous return and the shunted blood, leading to chamber dilation and hypertrophy over time. This enlargement can be visualized on an echocardiogram and often results in a prominent a-wave in the jugular venous pulse.
D. Fatigue with activity manifests because the heart's stroke volume is inefficiently distributed, with a portion of oxygenated blood recirculating through the lungs rather than the systemic tissues. During exertion, the right ventricle may struggle to meet the increased oxygen demand, leading to exercise intolerance and diaphoresis. In infants, this often presents as "feeding exhaustion," where the child must stop frequently to rest during bottle or breastfeeding.
Rationale for incorrect answers:
C. Decreased pulmonary blood flow is characteristic of cyanotic heart defects, such as tetralogy of Fallot or pulmonic stenosis, where blood is obstructed from reaching the lungs. In an atrial septal defect, there is actually increased pulmonary blood flow because the left-to-right shunt adds extra volume to the pulmonary circuit. This excess flow is the primary driver of the clinical symptoms and long-term vascular damage associated with the condition.
E. Left ventricular hypertrophy is not typically associated with an atrial septal defect, as the primary burden is placed on the right-sided chambers. Left-to-right shunting actually results in a "volume-depleted" left ventricle relative to the overloaded right side, often causing the interventricular septum to flatten or bow toward the left. Left ventricular hypertrophy is more commonly seen in conditions like aortic stenosis or systemic hypertension where the left heart faces high afterload.
Test-taking strategy:
- Categorize the defect first: Is it cyanotic (decreased flow) or acyanotic (increased flow). ASD is acyanotic, so flow to the lungs must be increased.
- Trace the blood flow path: Left atrium to right atrium, then into the right ventricle and lungs; therefore, the right side and lungs are the structures that "suffer."
- Link pulmonary overcirculation to clinical symptoms: Extra fluid in the lungs always equals a higher risk for respiratory infections and crackles.
- Differentiate between atrial and ventricular burdens: ASD predominantly affects the atria and the right ventricle, whereas VSD affects both ventricles.
- Use the "path of least resistance" rule: Blood flows from the high-pressure left side to the lower-pressure right side, creating a volume load for the right side.
- Rule out options that describe obstructive physiology like stenosis or systemic pressure issues when dealing with a simple septal opening.
Take home points
- Atrial septal defect leads to increased pulmonary blood flow, which clinically manifests as frequent lower respiratory tract infections.
- Right-sided heart enlargement (atrial and ventricular) is the classic structural compensation for the chronic left-to-right shunt.
- The characteristic physical exam finding is a fixed, widely split second heart sound (S2) regardless of the respiratory cycle.
- Untreated defects in adults significantly increase the risk for paradoxical embolic strokes and pulmonary arterial hypertension.
A nurse anticipates which client with an atrial septal defect will most likely require surgical intervention?
Explanation
Atrial septal defect management is determined by the hemodynamic significance of the left-to-right shunt and its impact on cardiac structure. While many small defects undergo spontaneous closure during the first year of life, larger defects cause a chronic volume load on the right heart. This leads to remodeling of the myocardium, permanent dilation of the right-sided chambers, and a high risk for pulmonary hypertension if the defect is not mechanically closed.
Rationale for correct answer:
C. Surgical or transcatheter intervention is indicated when there is objective evidence of right-sided heart enlargement or a significant pulmonary-to-systemic flow ratio. This structural change proves the shunt is large enough to cause pathological stretching of the right atrium and ventricle. Closing the defect at this stage prevents the progression to irreversible heart failure and atrial arrhythmias that typically manifest later in life.
Rationale for incorrect answers:
A. An asymptomatic infant is rarely a candidate for immediate surgery because many atrial septal defects, especially small secundum types, close without intervention. Physicians typically monitor these patients with serial echocardiography to track the size of the hole as the heart grows. Unless the infant exhibits severe failure to thrive or congestive heart failure, the standard of care is to wait until the child is older.
B. A child with a small defect and no symptoms does not meet the clinical criteria for invasive closure. Small defects that do not result in volume overload of the right heart are often hemodynamically insignificant and carry a low risk for complications. These patients may lead a normal life with regular cardiology follow-up rather than undergoing the risks associated with general anesthesia and cardiac surgery.
D. A newborn with a transient murmur often has a patent foramen ovale or a functional murmur that resolves as pulmonary vascular resistance drops. Most murmurs heard in the first 48 hours of life are benign and do not necessitate surgical planning. Only defects that persist beyond the neonatal period and show evidence of a significant shunt require a long-term plan for surgical or device-based repair.
Test-taking strategy:
- Apply the principle of hemodynamic impact: Surgery is reserved for defects that cause physical changes to the heart's anatomy.
- Identify right-sided heart enlargement as the "red flag" indicator that the heart can no longer compensate for the extra volume.
- Recognize the natural history of ASDs; smaller defects frequently close on their own, making immediate surgery unnecessary for small or asymptomatic cases.
- Prioritize objective evidence such as enlargement over subjective findings such as a transient murmur when choosing the most likely candidate for an invasive procedure.
- Use the age-appropriate rule: Surgical intervention for ASD is usually delayed until age 2 to 4 unless the patient is symptomatic.
- Differentiate between a pathological shunt and a transient neonatal finding; "transient" implies a condition that will fix itself without medical intervention.
Take home points
- Indications for ASD closure include a Qp/Qs ratio > A.5:1 or evidence of right ventricular volume overload.
- Most pediatric ASD repairs are performed electively between the ages of 2 and 5 years.
- Transcatheter device closure is the preferred method for secundum ASDs with sufficient septal rims.
- Untreated large ASDs lead to a shortened life expectancy due to pulmonary vascular disease and right heart failure.
A nurse provides education to caregivers of a child with an untreated atrial septal defect. Which long-term complications should the nurse include? Select all that apply
Explanation
Atrial septal defect is a congenital cardiac anomaly where a deficiency in the interatrial septum creates a persistent communication between the atria. The resulting left-to-right shunt causes a chronic increase in pulmonary blood flow and volume overload of the right heart chambers. Over decades, this hemodynamic stress leads to irreversible structural changes in the pulmonary vasculature and cardiac remodeling, significantly increasing the risk of morbidity and mortality in adulthood.
Rationale for correct answers:
A. Pulmonary hypertension develops due to the chronic exposure of the pulmonary arterial bed to excessive flow. This high-volume state triggers endothelial dysfunction and medial hypertrophy of the small pulmonary arteries, increasing vascular resistance. Eventually, the pulmonary pressures may equal or exceed systemic pressures, leading to a shunt reversal known as Eisenmenger syndrome, characterized by central cyanosis and digital clubbing.
B. Right-sided heart failure occurs because the right ventricle is subjected to chronic volume overload. To accommodate the shunted blood from the left atrium, the right ventricle undergoes dilation and eventual hypertrophy to maintain cardiac output. Over time, the myocardial wall stress exceeds the compensatory mechanisms, leading to systolic dysfunction, peripheral edema, hepatomegaly, and exercise intolerance.
C. Stroke later in life is a significant risk due to the potential for paradoxical embolism. A thrombus originating in the systemic venous circulation can cross through the atrial septal defect into the left atrium, bypassing the pulmonary filter. Once in the left-sided circulation, the embolus can travel to the cerebral arteries, causing an ischemic stroke, a risk that is heightened if the patient develops atrial fibrillation due to atrial stretching.
Rationale for incorrect answers:
D. Decreased coronary perfusion is not a typical long-term complication of an atrial septal defect. Coronary blood flow is primarily determined by the aortic diastolic pressure and coronary vascular resistance. In an uncomplicated atrial septal defect, the left ventricular output and aortic pressures are generally maintained within normal limits unless the patient reaches the end stages of heart failure or severe Eisenmenger physiology.
E. Systemic hypotension is generally not seen in patients with an untreated atrial septal defect. The body employs compensatory mechanisms, such as increasing the heart rate and systemic vascular resistance, to maintain an adequate mean arterial pressure. Hypotension would only occur as a terminal event in decompensated heart failure rather than serving as a characteristic long-term complication of the shunt itself.
Test-taking strategy:
- Evaluate the direction of flow: Left-to-right shunts always put the "stress" on the lungs and the right side of the heart first.
- Connect anatomical changes to clinical outcomes: Atrial stretching leads to arrhythmias, and right-sided overload leads to right heart failure.
- Identify the unique risk of paradoxical embolism: Any hole in the heart that connects the right and left sides carries an inherent risk of a clot "skipping" the lungs.
- Distinguish between pulmonary and systemic effects. ASD is primarily a pulmonary overcirculation disease, not a systemic low-pressure disease.
- Recall the natural history of ASD: It is often a "silent" defect in childhood that manifests with severe complications like pulmonary hypertension in the 3rd or 4th decade of life.
- Rule out options that describe left-sided pressure issues (like coronary perfusion) unless the defect directly involves the aorta or left ventricular outflow.
Take home points
- Chronic pulmonary overcirculation from an ASD leads to irreversible pulmonary arterial hypertension.
- Atrial septal defects are a known risk factor for cryptogenic strokes via paradoxical embolization of venous thrombi.
- Right-sided heart failure is the eventual result of chronic right ventricular volume and pressure overload.
- Early closure of the defect is the primary intervention used to prevent these long-term cardiovascular sequelae.
Practice Exercise 2
An infant with a ventricular septal defect is receiving digoxin (Lanoxin). Which intervention by the nurse is most appropriate before digoxin administration?
Explanation
Ventricular septal defect is a common congenital cardiac anomaly involving an opening in the interventricular septum, leading to a left-to-right shunt. This shunt causes pulmonary overcirculation and eventual congestive heart failure, necessitating pharmacologic support with inotropic agents like digoxin. Digoxin enhances myocardial contractility while simultaneously exerting a negative chronotropic effect by increasing vagal tone and slowing conduction through the atrioventricular node.
Rationale for correct answer:
D. Assessing the apical pulse for a full 60 seconds is the standard nursing intervention to identify bradycardia or arrhythmias prior to digoxin administration. In infants, the heart rate is highly variable and prone to sinus arrhythmia, making a full minute of auscultation necessary for accuracy. Digoxin must be withheld, and the provider notified, if the infant’s heart rate falls below the specific threshold, typically 90 beats per minute for an infant.
Rationale for incorrect answers:
A. Taking the infant's blood pressure is not a prerequisite for digoxin administration, as the drug primarily affects contractility and rate rather than systemic vascular resistance. While blood pressure monitoring is vital in assessing overall cardiac output and the efficacy of heart failure management, it does not provide the safety data required to prevent digoxin toxicity. Digoxin is not an antihypertensive medication, and its immediate side effects are reflected in rhythm and rate changes rather than acute blood pressure fluctuations.
B. Checking the respiratory rate for 1 minute is a critical part of a pediatric cardiovascular assessment, but it is not the determining factor for giving digoxin. Tachypnea is a hallmark sign of pulmonary congestion in ventricular septal defects, and a decrease in rate may indicate the medication's therapeutic success. However, the drug’s toxic threshold is measured by its effect on the cardiac conduction system, not the respiratory centers of the brain.
C. Checking the radial pulse for 1 minute is unreliable in infants because peripheral pulses are often faint or difficult to palpate accurately. The apical pulse, heard directly over the precordium at the fourth intercostal space, provides the most accurate representation of the actual heart rate. Using a radial site increases the risk of undercounting the rate due to peripheral pulse deficits, potentially leading to an inappropriate decision to withhold or give the dose.
Test-taking strategy:
- Prioritize safety assessments that directly monitor the drug's known mechanism of action and common toxic side effects.
- Select the most accurate measurement technique: For infants, central assessments (apical) always supersede peripheral assessments (radial) for cardiac data.
- Identify the full-minute requirement for pediatric vital signs to account for the irregularities common in young children's heart and respiratory rhythms.
- Distinguish between a therapeutic effect monitor that is the respiratory rate and a safety limit monitor that is the heart rate when assessing for medication administration.
- Use the "hold" parameters as a guide: if you must hold a drug for a low heart rate, then measuring the heart rate is the priority action.
- Rule out options that involve hemodynamic parameters not directly altered by the specific drug class being administered.
Take home points
- Digoxin toxicity in infants often presents as bradycardia, vomiting, and poor feeding.
- The apical pulse must be counted for one full minute to ensure accuracy before every dose.
- Standard practice requires withholding digoxin in infants if the heart rate is less than 90 to 100 beats per minute.
- Digoxin increases cardiac output by strengthening the force of contraction while slowing the heart rate to allow for better filling.
A nurse is assessing a child with a ventricular septal defect. Which of the following signs may be seen in this client?
Explanation
Ventricular septal defect is a frequent congenital heart lesion characterized by a deficiency in the interventricular septum, which allows a left-to-right shunt of blood. This hemodynamic abnormality results in pulmonary overcirculation as high-pressure oxygenated blood from the left ventricle is forced into the lower-pressure right ventricle. The clinical severity depends on the defect size, with larger openings leading to congestive heart failure, tachypnea, and increased metabolic expenditure.
Rationale for correct answer:
D. Most children with an uncomplicated ventricular septal defect remain acyanotic because the shunt is left-to-right, ensuring that systemic circulation consists of oxygenated blood. Consequently, the patient will typically display pink nailbeds and a brisk capillary refill of less than 2 seconds, indicating adequate peripheral perfusion. Unless the defect is so large that it causes profound low-output heart failure, systemic oxygen delivery to the tissues remains sufficient for normal capillary refill.
Rationale for incorrect answers:
A. Cyanosis of the nailbeds is not an expected finding in an acyanotic defect like a ventricular septal defect unless shunt reversal has occurred. Simple left-to-right shunting does not result in deoxygenated blood entering the aorta, so the skin and mucous membranes remain pink. The presence of cyanosis in a child with a known septal defect would suggest a progression toward Eisenmenger syndrome or an additional obstructive right-sided lesion.
B. Children with significant ventricular septal defects are rarely above-average in height and frequently exhibit growth retardation. The increased metabolic demand of a heart working against a volume load, combined with respiratory distress, often results in a failure to meet linear growth milestones. These children often fall below the 10th percentile on standardized growth charts due to the physiologic stress of the cardiac lesion.
C. Above-average weight gain is highly unlikely because infants with ventricular septal defects suffer from poor feeding and high caloric expenditure. The effort required to breathe while eating leads to early exhaustion, resulting in a caloric deficit and failure to thrive. While fluid retention from congestive heart failure might cause a sudden weight spike, it does not represent healthy or above-average growth on a standard pediatric chart.
Test-taking strategy:
- Categorize the defect: A ventricular septal defect is an acyanotic defect, so eliminate any options suggesting cyanosis or blue discoloration.
- Apply the metabolic cost principle: Congenital heart defects generally cause the body to "work harder," which leads to below-average weight and height, not above.
- Evaluate peripheral perfusion: In left-to-right shunts, the systemic blood is well-oxygenated, therefore capillary refill and nailbed color should be normal.
- Distinguish between fluid weight and growth: Heart failure can cause edema, but "above-average weight gain" in a nursing context usually refers to healthy growth milestones.
- Focus on systemic oxygenation: Since the left ventricle is pumping oxygenated blood to the body, there is no physiological reason for a simple VSD to cause dusky nailbeds.
Take home points
- Ventricular septal defects are acyanotic because oxygenated blood recirculates through the lungs rather than deoxygenated blood entering the body.
- Failure to thrive and poor weight gain are the most common physical findings in symptomatic children with large defects.
- Peripheral pulses and capillary refill are typically normal in these children unless they are in advanced cardiogenic shock.
- Assessment of growth parameters using standardized charts is a critical nursing intervention for monitoring the severity of the shunt.
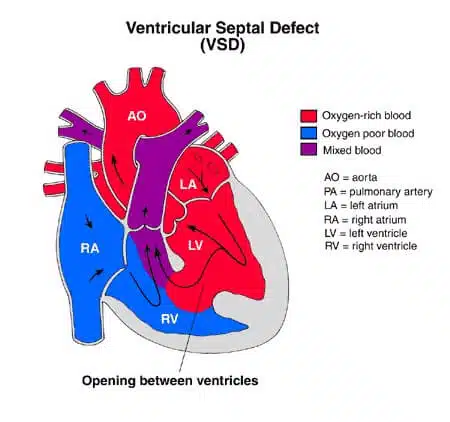
A nurse is caring for a child diagnosed with a ventricular septal defect. Which description would the nurse incorporate when teaching the parents about this condition?
Explanation
Ventricular septal defect is a congenital heart malformation characterized by an abnormal opening in the interventricular septum, which normally serves as the muscular and membranous barrier between the lower chambers. This defect allows a left-to-right shunt because the left ventricle generates significantly higher pressure during systole than the right ventricle. The resulting flow causes pulmonary overcirculation, leading to increased pulmonary venous return and potential volume overload of the left atrium and left ventricle.
Rationale for correct answer:
D. A ventricular septal defect represents a failure of the septum to close completely during embryologic development, specifically between the right and left ventricles. This allows oxygenated blood to leak from the high-pressure left side back into the right side, where it is pumped to the lungs again. The nurse must explain that this inefficient circulation forces the heart to work harder to maintain systemic output while simultaneously flooding the pulmonary system.
Rationale for incorrect answers:
A. A narrowing of the aortic arch is the defining characteristic of coarctation of the aorta, which is an obstructive defect rather than a septal one. This condition leads to high blood pressure in the upper extremities and diminished pulses in the lower extremities due to the physical restriction of blood flow. It does not involve a shunt between the cardiac chambers but rather increases the afterload on the left ventricle as it struggles to pump blood past the narrowing.
B. A failure of a septum to develop completely between the atria describes an atrial septal defect, which occurs in the upper chambers of the heart. While both ASD and VSD involve left-to-right shunting, the pressure dynamics and clinical timing of symptoms differ significantly between the two. Atrial shunts are usually lower pressure and may remain asymptomatic for years, whereas ventricular shunts often present with signs of heart failure much earlier in infancy.
C. A narrowing of the valves at the entrance of the pulmonary artery is known as pulmonic stenosis, which obstructs blood flow from the right ventricle to the lungs. This creates a pressure overload on the right side of the heart and may cause right ventricular hypertrophy, but it does not involve a septal opening. In some complex cases, pulmonic stenosis may be part of a larger syndrome, such as tetralogy of Fallot, where it is accompanied by a ventricular septal defect.
Test-taking strategy:
- Focus on the anatomical terminology in the question; "ventricular" refers specifically to the lower chambers of the heart.
- Match the prefix or root of the defect name (ventricular) with the corresponding anatomical structure (ventricles) in the answer choices.
- Rule out options that describe narrowing or stenosis, as these are obstructive lesions, not "defects" in the septal walls.
- Distinguish between the atria (upper chambers) and ventricles (lower chambers) to differentiate between ASD and VSD.
- Utilize the pathophysiology of shunting to confirm that "failure of a septum to develop" is the scientific mechanism for any septal hole.
- Eliminate coarctation or aortic arch issues when the question specifically asks about a septal defect.
Take home points
- Ventricular septal defect is the most common congenital heart defect, occurring in the wall between the two lower chambers.
- The pressure difference between the ventricles causes a left-to-right shunt, leading to pulmonary congestion.
- Small VSDs may be asymptomatic and close spontaneously, while large VSDs require surgical repair to prevent heart failure.
- A loud, harsh holosystolic murmur heard at the left lower sternal border is the classic physical finding.
A nurse is caring for an infant with ventricular septal defect and heart failure. Which interventions are appropriate? Select all that apply
Explanation
Heart failure in the setting of a ventricular septal defect is characterized by pulmonary overcirculation due to the left-to-right shunting of blood. This volume overload leads to increased hydrostatic pressure in the pulmonary capillaries, causing interstitial edema and decreased lung compliance. The myocardium compensatory mechanisms, including the renin-angiotensin-aldosterone system, result in salt and water retention, further exacerbating systemic and pulmonary venous congestion.
Rationale for correct answers:
A. Diuretics such as furosemide are the cornerstone of management to reduce the circulating blood volume and pulmonary congestion. By inhibiting sodium reabsorption in the loop of Henle, these agents promote the excretion of excess fluid, thereby decreasing the preload on the weakened heart. Reducing the fluid volume helps alleviate symptoms of tachypnea and work of breathing in the infant.
C. Implementing a fluid restriction is necessary to prevent the worsening of volume overload and systemic edema. In infants with heart failure, the total daily intake must be carefully calculated to ensure adequate metabolic support without exceeding the heart's capacity to process the fluid. This intervention helps maintain a neutral fluid balance and reduces the stress on the ventricles.
E. Oxygen administration is appropriate for infants showing signs of respiratory distress or hypoxia due to pulmonary edema. Supplemental oxygen acts as a pulmonary vasodilator, which can help improve oxygenation. However, it must be used judiciously as it may also decrease pulmonary vascular resistance and increase the left-to-right shunt. The goal is to maintain target saturations while minimizing the work of the respiratory muscles.
Rationale for incorrect answers:
B. Low-calorie feedings are contraindicated because infants with heart failure have increased metabolic demands and often suffer from failure to thrive. These infants require high-calorie formulas (e.g., 24 to 30 kcal/oz) to provide enough energy for growth while maintaining fluid restrictions. Providing low-calorie nutrition would lead to malnutrition and impair the infant's ability to recover or tolerate future surgical intervention.
D. Increasing sodium intake would be detrimental as it promotes water retention and further increases the intravascular volume. The pathophysiology of heart failure already involves a state of sodium sensitivity and fluid accumulation. Most infants are placed on low-sodium protocols or are monitored closely to prevent the exacerbation of edema and hypertension associated with high salt intake.
Test-taking strategy:
- Identify the primary problem: Heart failure in an infant is a "too much fluid" problem, so interventions must focus on fluid removal.
- Apply the ABCs: Oxygen addresses the airway/breathing compromised by pulmonary congestion.
- Use the physiological demand principle: Cardiac babies burn more energy, so they need more calories (high-density), not fewer.
- Recall pharmacology basics: Diuretics are the standard "gold-label" treatment for any congestive heart failure scenario.
- Analyze electrolyte balance: Sodium follows water so if you want to get rid of water, you must restrict, not increase, sodium.
- Think about organ protection: Fluid restriction protects the lungs and heart from being overwhelmed by the shunt's volume.
Take home points
- The goal of heart failure management in VSD is to reduce preload and pulmonary congestion.
- Nutritional support for cardiac infants must prioritize high caloric density within a restricted fluid volume.
- Diuretics require close monitoring of serum electrolytes, especially potassium levels.
- Oxygen should be titrated to the lowest effective dose to avoid worsening the left-to-right shunt through vasodilation.
A nurse reviews manifestations of increased pulmonary blood flow in a client with a ventricular septal defect. Which of the following findings are expected? Select all that apply
Explanation
Ventricular septal defect is a congenital cardiovascular malformation involving a deficiency in the interventricular septum, which allows for a high-pressure left-to-right shunt. This hemodynamic shift causes excessive blood volume to enter the pulmonary circulation, leading to pulmonary overcirculation and interstitial edema. The resulting decrease in lung compliance and increased cardiac workload trigger systemic compensatory mechanisms and significant metabolic stress in the affected infant or child.
Rationale for correct answers:
A. Tachypnea is a primary clinical manifestation resulting from pulmonary congestion and decreased lung compliance. The presence of excess fluid in the interstitial spaces of the lungs stimulates stretch receptors, leading to a rapid, shallow breathing pattern. This increased respiratory rate is the body's attempt to maintain adequate gas exchange despite the fluid-filled environment of the pulmonary parenchyma.
B. Frequent respiratory infections occur because the pulmonary overcirculation creates an environment of chronic interstitial edema. This excess fluid impairs the mucociliary escalator and provides a fertile medium for bacterial growth within the lower respiratory tract. Children with significant left-to-right shunts often present with recurrent bouts of pneumonia or bronchiolitis that are slower to resolve than in healthy peers.
D. Failure to thrive is an expected finding due to the massive caloric expenditure required to support a heart and respiratory system working at high capacity. The infant often experiences diaphoresis during feeding and becomes exhausted before consuming enough nutrients for growth. This results in a persistent inability to gain weight, often placing the child below the 5th percentile on standardized growth charts.
Rationale for incorrect answers:
C. Clubbing of fingers is a sign of chronic systemic hypoxemia and is typically seen in cyanotic heart defects or long-standing right-to-left shunts. In an uncomplicated ventricular septal defect, the blood reaching the systemic tissues is oxygenated, so clubbing does not occur. This finding would only appear if the patient developed Eisenmenger syndrome, representing a late and severe complication of the untreated defect.
E. Infants with ventricular septal defects actually have a significantly increased oxygen demand. The extra work of breathing and the inefficient hyperdynamic heart require more oxygen and energy than a normal physiological state. Management strategies focus on reducing this demand by providing rest periods and maintaining a neutral thermal environment.
F. Cyanosis at rest is not a feature of an acyanotic defect like a ventricular septal defect where the shunt is left-to-right. Systemic arterial oxygen saturation remains within normal limits because the blood traveling to the body has already been oxygenated in the lungs. Cyanosis only occurs in "blue baby" defects, such as tetralogy of Fallot, where deoxygenated blood bypasses the lungs entirely.
Test-taking strategy
- Classify the defect: VSD is an acyanotic defect, so immediately eliminate any options associated with low oxygen such as cyanosis and clubbing.
- Link "increased flow" to "wet lungs": Excess blood in the lungs always leads to tachypnea and a higher risk for infections.
- Apply the metabolic cost rule: Cardiac defects that flood the lungs cause the body to "burn" more energy, leading to failure to thrive.
- Evaluate the direction of the shunt: In a left-to-right shunt, the body gets enough oxygen, but the lungs get too much blood.
- Distinguish between acute and chronic signs: Clubbing takes months or years of low oxygen to develop and is not typical for a simple VSD.
- Rule out options that imply the body is "working less"; cardiac defects always result in increased work and demand.
Take home points
- Increased pulmonary blood flow leads to pulmonary edema, characterized by tachypnea and crackles upon auscultation.
- Chronic pulmonary congestion increases the frequency and severity of lower respiratory tract infections.
- Nutritional failure in VSD is multifactorial, involving both increased metabolic rate and poor oral intake due to fatigue.
- Left-to-right shunts do not cause cyanosis unless pulmonary pressures rise high enough to reverse the shunt.
Practice Exercise 3
A nurse is attending a staff education program on congenital heart defects. What type of congenital defect occurs when the endocardial cushions fail to fuse completely?
Explanation
Atrioventricular canal defect, also known as endocardial cushion defect, results from the incomplete embryonic development of the endocardial cushions. These specialized structures are essential for the formation of the lower portion of the atrial septum, the upper portion of the ventricular septum, and the mitral and tricuspid valves. The failure of these cushions to fuse results in a large central "hole" in the heart and a common atrioventricular valve, which frequently leads to massive left-to-right shunting and early-onset pulmonary hypertension.
Rationale for correct answer:
A. Atrioventricular canal defect occurs when the endocardial cushions fail to fuse during the 6th week of gestation. This results in a combined primum atrial septal defect and a ventricular septal defect, along with malformed atrioventricular valves. This defect is strongly associated with Trisomy 21 and requires early surgical intervention to prevent irreversible pulmonary vascular disease. The deficiency of the endocardial cushions prevents the normal separation of the four heart chambers.
Rationale for incorrect answers:
B. Atrial septal defect is a generic term for several types of openings in the interatrial wall, but only the primum type is related to endocardial cushion issues. The most common type, the secundum defect, involves the center of the septum (fossa ovalis) and is unrelated to cushion fusion. Atrial septal defects allow left-to-right shunting but do not involve the ventricular septum or the common atrioventricular valve structure characteristic of canal defects.
C. Coarctation of the aorta is an obstructive defect involving a narrowing of the aorta, typically near the ductus arteriosus insertion site. It does not involve the internal septa of the heart or the endocardial cushions but is rather a vascular malformation. This condition leads to a pressure gradient between the upper and lower extremities, manifesting as hypertension in the arms and diminished femoral pulses.
D. Aortic stenosis involves a narrowing or malformation of the aortic valve, obstructing blood flow from the left ventricle into the systemic circulation. This is an obstructive valvular lesion and does not result from the failure of septal fusion at the center of the heart. The pathophysiology involves left ventricular hypertrophy due to increased afterload rather than the shunting of blood between chambers seen in cushion defects.
Test-taking strategy:
- Connect the embryological structure, the endocardial cushions directly with its namesake defect (endocardial cushion defect), which is a synonym for AV canal.
- Recognize the anatomic scope: Cushion defects affect the "canal" between the atria and ventricles, involving both levels of the heart.
- Identify syndromic associations: If the question mentioned Down syndrome, AV canal would be the most statistically likely answer.
- Distinguish between septal/shunting defects and obstructive/stenotic defects; cushion issues always fall into the shunting category.
- Use word association: "Fusion" refers to the merging of walls; only options 1 and 2 describe wall (septal) issues, and 1 is more comprehensive.
Take home points
- Atrioventricular canal defect is the definitive result of failed endocardial cushion fusion.
- This defect is the most common cardiac anomaly found in infants with Down syndrome.
- It produces a common AV valve instead of separate mitral and tricuspid valves, leading to significant regurgitation.
- Surgical repair is typically required within the first 6 months of life to prevent heart failure.
A nurse is assessing an infant with atrioventricular canal defect. Which findings are expected? Select all that apply
Explanation
Atrioventricular (AV) canal defect, also known as endocardial cushion defect, is a severe congenital heart anomaly where there is a large hole in the center of the heart. This involves both an atrial septal defect and a ventricular septal defect, combined with a single, common atrioventricular valve instead of separate mitral and tricuspid valves. This configuration allows massive left-to-right shunting of blood, resulting in a volume and pressure overload of the lungs and all four heart chambers.
Rationale for correct answers:
A. Signs of heart failure are common because the large central opening allows a massive amount of blood to be shunted from the high-pressure left side of the heart to the low-pressure right side. The heart must work much harder to pump this extra blood, leading to symptoms like tachypnea, tachycardia, and diaphoresis, particularly during feedings.
B. Poor weight gain occurs because the infant is in a constant hypermetabolic state. The heart and lungs are working overtime, burning through calories at an accelerated rate. Additionally, the infant often becomes too exhausted to finish a bottle or breastfeed, leading to a condition known as failure to thrive.
C. Recurrent respiratory infections result from pulmonary overcirculation. The excess blood flow to the lungs causes pulmonary congestion and interstitial edema. This "wet" environment in the lungs makes them a breeding ground for bacteria and viruses, leading to frequent bouts of pneumonia and bronchiolitis.
Rationale for incorrect answers:
D. Decreased pulmonary blood flow is incorrect because an AV canal defect causes a massive increase in pulmonary blood flow due to the left-to-right shunt. Decreased flow is typically seen in cyanotic defects like tetralogy of Fallot, where blood is physically blocked from reaching the lungs. In AV canal defects, the lungs are actually "flooded" with too much blood.
E. Bradycardia is not an expected finding. Infants with AV canal defects and resulting heart failure almost always exhibit tachycardia as the body tries to compensate for the inefficient circulation. While surgical repair of an AV canal can sometimes damage the electrical conduction system and cause heart block leading to bradycardia, it is not a primary manifestation of the untreated defect.
Test-taking strategy
- Categorize the shunt: AV canal is a "left-to-right" shunt, which always means the lungs get too much blood.
- Link the lungs to infections: Any defect that makes the lungs "wet" (increased flow) will lead to frequent respiratory illnesses.
- Connect heart failure to growth: If the heart is failing, the baby is burning too many calories and will not gain weight well.
- Watch for "opposites": Options 1 and 4 are opposites. If a defect causes heart failure via a shunt, it almost always involves increased flow, not decreased.
- Know the association: Remember that this defect is the one most commonly associated with Down syndrome (Trisomy 21).
Take home points
- AV canal defect creates a "common" valve that allows blood to mix freely between all four chambers.
- It is a high-flow, high-pressure defect that leads to early and severe heart failure.
- Surgical repair is usually required within the first 3 to 6 months of life to prevent permanent lung damage.
- Nursing care focuses on high-calorie nutrition, fluid management, and monitoring for respiratory distress.
A nurse recognizes that atrioventricular canal defect is most commonly associated with which condition?
Explanation
Atrioventricular (AV) canal defect, also known as endocardial cushion defect, is a severe congenital heart anomaly that occurs when the central tissues of the heart fail to fuse during embryonic development. This failure creates a large central "hole" involving both the atrial and ventricular septa and results in a common atrioventricular valve instead of separate mitral and tricuspid valves.
Rationale for correct answer:
B. Down syndrome (Trisomy 21) is the condition most strongly associated with atrioventricular canal defects. Approximately 40% to 50% of children born with Down syndrome have a congenital heart defect, and of those, the AV canal defect is the most common. Because of this high correlation, every newborn diagnosed with Down syndrome must undergo a comprehensive cardiac evaluation, including an echocardiogram, to screen for this specific malformation.
Rationale for incorrect answers:
A. Turner syndrome (45,X) is more commonly associated with left-sided obstructive lesions, specifically coarctation of the aorta and bicuspid aortic valves. While children with Turner syndrome can have septal defects, the "canal" defect is not their hallmark cardiac association.
C. Marfan syndrome is a connective tissue disorder that primarily affects the aorta and the valves later in life. The most frequent cardiovascular complications include aortic root dilation, aortic dissection, and mitral valve prolapse. It is not typically associated with the embryological failure of the endocardial cushions seen in AV canal defects.
D. Cystic fibrosis is a genetic disorder affecting the exocrine glands, primarily impacting the respiratory and digestive systems. It is not a congenital heart defect nor is it associated with structural cardiac malformations at birth. Any cardiac involvement in cystic fibrosis usually develops later as cor pulmonale (right-sided heart failure) due to chronic lung disease.
Test-taking strategy
- Memorize the "Classic Duo": In pediatric nursing, always link AV canal/Endocardial cushion defect with Down Syndrome. This is a high-yield association for exams.
- Differentiate the syndromes:
- Down syndrome is linked to AV canal.
- Turner syndrome is linked to coarctation of the aorta.
- Marfan syndrome is linked to aortic aneurysm/dilation.
- DiGeorge syndrome is linked to tetralogy of Fallot/truncus arteriosus.
- Think "Central": The AV canal is a defect in the "center" of the heart; Down syndrome is a "central" genetic topic in pediatrics.
Take home points
- AV canal defect is the most frequent cardiac anomaly in children with Trisomy 2A.
- The defect involves a "common" valve that leads to severe shunting and early heart failure.
- Surgical repair is essential and is usually performed before 6 months of age to prevent permanent pulmonary hypertension.
A nurse anticipates that surgical repair of atrioventricular canal defect is typically performed:
Explanation
Atrioventricular (AV) canal defect is a high-pressure, high-flow lesion that involves both atrial and ventricular septal defects and a common, often leaky, atrioventricular valve. Because this defect allows massive amounts of blood to be shunted to the lungs under high pressure, it can lead to irreversible pulmonary vascular obstructive disease (Eisenmenger syndrome) much faster than smaller or simpler defects.
Rationale for correct answer:
C. Surgical repair is typically performed within the first year of life, usually between 3 and 6 months of age. This timeline is chosen to intervene before the chronic high pressure from the left-to-right shunt causes permanent damage to the pulmonary arteries. Repairing the defect during infancy allows the child to grow and prevents the development of severe, irreversible pulmonary hypertension, which would make later surgery impossible.
Rationale for incorrect answers:
A. Immediately after birth is generally not required unless the infant is in profound, intractable heart failure that cannot be managed with medication. Surgeons prefer to allow the infant to grow slightly and for pulmonary vascular resistance to drop naturally after birth, which makes the surgical repair and postoperative recovery safer.
B. Waiting until adolescence is far too late. By the time a child with a complete AV canal defect reaches adolescence, they would almost certainly have developed irreversible pulmonary hypertension and heart failure. The damage to the lung's blood vessels would be permanent, and the heart would likely be severely enlarged and weakened.
D. Delaying surgery until adulthood is not a viable strategy for this defect. While some simple atrial septal defects (ASDs) are found in adulthood, a complete AV canal defect is a "complete" failure of the center of the heart and is too hemodynamically severe to remain untreated for decades.
Test-taking strategy:
- Identify the severity: AV canal is a "large hole" defect. Large holes require early intervention to protect the lungs.
- Know the "safe window": In pediatric cardiology, the "first year of life" is the standard window for repairing defects that cause high pulmonary pressure.
- Recall the goal: The primary goal of early repair in shunting defects is to prevent Eisenmenger syndrome (irreversible lung damage).
- Rule out extremes: "Immediately after birth" is rarely the answer for septal repairs; "Adulthood" is only for very minor or hidden defects.
Take home points
- Most infants with AV canal defects are managed with heart failure medications (like diuretics) to "bridge" them to surgery at 3-6 months.
- Early surgery is critical for children with Down syndrome, who are more prone to developing pulmonary hypertension early.
- The surgery involves patching the holes and creating two separate valves (mitral and tricuspid) from the single common valve.
A nurse explains that infants with atrioventricular canal defect develop heart failure early due to:
Explanation
Atrioventricular (AV) canal defect represents a "complete" failure of the heart’s center to form. Because there is a large, continuous opening between the atria and the ventricles, blood follows the path of least resistance. In a newborn, the left side of the heart generates much higher pressure than the right side, creating a massive left-to-right shunt.
Rationale for correct answer:
B. The large left-to-right shunt is the primary driver of early heart failure. Oxygenated blood that should be going out to the body is instead forced back into the right side of the heart and recirculated through the lungs. This creates a "volume overload" cycle since the heart has to pump the same blood twice, which quickly exhausts the heart muscle and floods the lungs with excess fluid (pulmonary congestion).
Rationale for incorrect answers:
A. Severe obstruction of systemic blood flow is characteristic of "left-sided" obstructive defects, such as aortic stenosis or coarctation of the aorta. AV canal is a shunting defect, not a blockage. While systemic flow can be slightly reduced because so much blood is diverted to the lungs, "obstruction" is not the underlying mechanism.
C. Decreased cardiac preload is the opposite of what occurs. In AV canal defects, there is actually increased preload, the volume entering the heart chambers, because the shunted blood adds to the normal venous return. This extra volume stretches the heart chambers, eventually leading to dilation and failure.
D. Increased systemic resistance refers to the resistance the heart must pump against in the body. AV canal defects are generally associated with decreased pulmonary resistance relative to the systemic side, which is why the blood shunts into the lungs. Increased systemic resistance would actually make the left-to-right shunt even worse, but it is not the cause of the initial heart failure.
Test-taking strategy:
- Identify the defect type: AV canal is an acyanotic, shunting defect. Shunting defects cause heart failure via volume overload, not obstruction.
- Trace the flow: Always remember that blood flows from high pressure (left) to low pressure (right). This "left-to-right" movement is the root of the problem in VSDs, ASDs, and AV canals.
- Keyword association: Heart failure with a septal hole is a shunt.
- Rule out "obstruction": If the defect name doesn't include "stenosis," "coarctation," or "atresia," it is likely not an obstructive lesion.
Take home points
- Heart failure in AV canal infants usually appears within the first 4 to 8 weeks of life as pulmonary resistance naturally drops.
- The "flooding" of the lungs leads to the classic symptoms: fast breathing, retractions, and difficulty feeding.
- Because the shunt involves both the atria and ventricles, the volume of blood being misdirected is much larger than in a simple isolated VSD.
Practice Exercise 4
An infant is diagnosed with patent ductus arteriosus. Which drug should the nurse anticipate administering to attempt to close the defect?
Explanation
Patent ductus arteriosus (PDA) occurs when the fetal shunt between the pulmonary artery and the aorta fails to close after birth. In the womb, the ductus arteriosus allows blood to bypass the fluid-filled lungs. After birth, this vessel should constrict and close; if it remains open (patent), oxygenated blood from the high-pressure aorta shunts back into the pulmonary artery, flooding the lungs.
Rationale for correct answer:
D. Indomethacin (Indocin) or Ibuprofen are the primary pharmacological treatments used to close a PDA, particularly in premature infants. These medications are prostaglandin inhibitors (NSAIDs). Because circulating prostaglandins are responsible for keeping the ductus arteriosus open during fetal life, inhibiting them triggers the smooth muscle in the ductus to constrict and close the vessel.
Rationale for incorrect answers:
A. Digoxin (Lanoxin) is a cardiac glycoside used to increase the force of heart contractions and slow the heart rate. While it may be used to manage the symptoms of heart failure caused by a large PDA, it has no physiological mechanism to actually close the ductus arteriosus.
B. Prednisone is a corticosteroid used to reduce inflammation. It is not used in the treatment of PDA. While steroids are sometimes given to mothers before preterm birth to help mature the infant's lungs, they do not play a role in the postnatal closure of the ductus.
C. Furosemide (Lasix) is a loop diuretic used to remove excess fluid from the lungs and body. Like Digoxin, it manages the symptoms of pulmonary congestion (the "wet lungs") associated with PDA, but it does not address the underlying structural defect.
Test-taking strategy:
- Identify the goal: The question asks for a drug to close the defect, not just treat the symptoms.
- Recall the "P" Rule:
- To keep it Patent (open): Give Prostaglandins.
- To close it: Give a Prostaglandin inhibitor (Indomethacin).
- Know your drug classes: Recognize Indomethacin and Ibuprofen as the "standard" non-surgical interventions for PDA in the NICU setting.
- Differentiate treatment vs. management: Diuretics and inotropes (Digoxin/Lasix) manage the effects of the hole; NSAIDs treat the hole itself.
Take home points
- Indomethacin is most effective when administered in the first few days of life.
- Before giving Indomethacin, the nurse must check the infant's renal function (urine output and creatinine) and platelet count, as the drug can cause kidney injury and increase the risk of bleeding.
- If pharmacological treatment fails, the infant may require surgical ligation or a transcatheter occlusion.
A nurse in the newborn unit is caring for several clients. While assessing a newborn with respiratory distress, the nurse auscultates a machine-like heart murmur. Other findings are a wide pulse pressure, periods of apnea, increased Pa CO2, and decreased POB. The nurse suspects that the newborn has:
Explanation
Patent ductus arteriosus (PDA) is a clinical condition where the fetal vascular connection between the pulmonary artery and the descending aorta fails to close after birth. This failure typically occurs within 72 hours of life in term infants but is frequently delayed in premature neonates due to low oxygen tension and high levels of circulating prostaglandins. The resulting left-to-right shunt causes pulmonary overcirculation and left-sided heart strain, potentially leading to pulmonary edema and respiratory compromise.
Rationale for correct answer:
B. The presence of a continuous machine-like murmur heard at the left upper sternal border is pathognomonic for a PDA. A wide pulse pressure occurs because blood shunts from the aorta into the pulmonary artery during diastole, lowering the diastolic pressure. The respiratory findings of apnea and altered blood gases such as increased PaCO2 and decreased PO2 result from decreased lung compliance caused by excessive pulmonary blood flow.
Rationale for incorrect answers:
A. Pulmonary hypertension in newborns involves high resistance in the pulmonary vascular bed, often leading to right-to-left shunting through fetal pathways. While it causes hypoxemia, it typically presents with a loud second heart sound (S2) and a systolic murmur of tricuspid regurgitation. It does not produce the classic continuous machinery-like sound or the wide pulse pressure associated with a persistent ductal connection.
C. A ventricular septal defect (VSD) is characterized by a harsh holosystolic murmur best heard at the left lower sternal border. While a large VSD can cause heart failure and respiratory distress, it does not create a continuous murmur because the pressure gradient exists primarily during systole. Furthermore, VSDs are not typically associated with the bounding pulses or wide pulse pressures seen in ductal shunting.
D. Bronchopulmonary dysplasia is a chronic lung disease resulting from long-term mechanical ventilation and oxygen therapy in premature infants. It manifests as tachypnea, crackles, and wheezing due to alveolar damage and fibrosis rather than a structural heart defect. While these infants may have heart strain (cor pulmonale), they do not present with the specific vascular murmur described in the assessment.
Test-taking strategy:
- Recognize pathognomonic buzzwords. The phrase "machine-like murmur" is the most specific clinical indicator for patent ductus arteriosus in pediatric board exams.
- Analyze blood pressure dynamics. A "wide pulse pressure" (the difference between systolic and diastolic) points toward a runoff of blood from the aorta, common in PDA.
- Connect respiratory and cardiac symptoms: In newborns, excess pulmonary blood flow from a shunt "drowns" the lungs, leading to poor gas exchange (high PaCO2).
- Rule out timing-specific murmurs: VSD is systolic only; PDA is continuous (machinery) because the pressure in the aorta is always higher than in the pulmonary artery.
- Consider neonatal history: Respiratory distress in a newborn often triggers the reopening or persistence of the ductus due to hypoxia.
Take home points
- The ductus arteriosus normally closes functionally within 15 hours and anatomically within weeks in full-term infants.
- Indomethacin or ibuprofen, which are prostaglandin inhibitors, are often used pharmacologically to induce closure of a PDA.
- Bounding peripheral pulses are a frequent physical finding due to the increased stroke volume and low diastolic pressure.
- Surgical ligation or transcatheter closure is indicated if medical management fails or if the infant remains ventilator-dependent.
A newborn is diagnosed with a congenital heart defect (CHD). The test results reveal that the lumen of the duct between the aorta and pulmonary artery remains open. This defect is known as:
Explanation
The ductus arteriosus is a vital fetal structure, a blood vessel that connects the pulmonary artery to the aorta. In utero, it allows blood to bypass the baby's fluid-filled, non-functioning lungs. When this vessel fails to close after birth, it is referred to as patent.
Rationale for the correct answer:
C. Patent ductus arteriosus (PDA) is the correct term for this specific defect. "Patent" means open, and "ductus arteriosus" is the name of the vessel itself. When it remains open, high-pressure blood from the aorta flows back into the pulmonary artery, a left-to-right shunt, which can lead to pulmonary congestion and heart failure if the opening is large.
Rationale for incorrect answers:
A. Atrial stenosis is not a standard term for a common congenital heart defect. Stenosis refers to the narrowing of a valve or vessel, such as aortic or pulmonic stenosis. It does not describe an open communication between the aorta and pulmonary artery.
B. Atrial septal defect (ASD) refers to an opening in the septum (wall) between the two upper chambers of the heart, the atria. While it is a left-to-right shunt, it occurs within the heart itself, not between the great arteries (aorta and pulmonary artery).
D. Ventricular septal defect (VSD) refers to an opening in the septum between the two lower chambers of the heart, the ventricles. This is the most common congenital heart defect, but like the ASD, it is an internal heart defect rather than a persistent fetal vessel.
Test-taking strategy:
- Break down the medical terms: "Patent" = Open. "Ductus Arteriosus" = the specific fetal duct. The name of the defect describes exactly what is happening.
- Identify the anatomy: The question specifies the "aorta and pulmonary artery." Only the PDA involves these two major vessels directly via a persistent lumen.
- Visualize the shunt: Remember that PDA is an extra-cardiac shunt (outside the heart), whereas ASD and VSD are intra-cardiac shunts (inside the heart).
Take home points
- PDA is most common in premature infants because their tissues are less responsive to the oxygen signals that trigger closure.
- The classic sign of a PDA is a continuous, machinery-like murmur.
- Medical closure is often attempted using Indomethacin or Ibuprofen, which are prostaglandin inhibitors.
A nurse explains that patent ductus arteriosus increases pulmonary blood flow because:
Explanation
Patent ductus arteriosus (PDA) involves the persistence of the fetal connection between the aorta and the pulmonary artery. After birth, systemic vascular resistance rises while pulmonary vascular resistance falls, creating a pressure gradient. This hemodynamic shift causes blood to shunt from the high-pressure systemic circulation into the lower-pressure pulmonary circulation, leading to pulmonary overcirculation and potential congestive heart failure.
Rationale for correct answer:
B. In the postnatal period, the aortic pressure significantly exceeds the pressure within the pulmonary artery throughout the cardiac cycle. Consequently, a patent ductus allows oxygenated blood to flow "backward" from the aorta into the pulmonary artery. This additional volume increases the total amount of blood entering the lungs, which can lead to pulmonary edema and respiratory distress in the neonate.
Rationale for incorrect answers:
A. Blood flowing from the pulmonary artery to the aorta describes a right-to-left shunt, which occurs primarily in fetal life or in certain cyanotic defects. In a typical PDA after the first few days of life, this direction is reversed because the systemic pressure is higher. If blood moved in this direction postnatally, it would result in systemic hypoxemia and cyanosis of the lower extremities.
C. In the transition to extrauterine life, systemic vascular resistance actually increases due to the removal of the low-resistance placental circuit. A decrease in systemic resistance would reduce the pressure gradient that drives the left-to-right shunt through the ductus. The pathophysiology of PDA is driven by the fact that systemic resistance is relatively high, forcing blood through any available escape path.
D. If pulmonary resistance exceeded systemic resistance, blood would shunt away from the lungs toward the aorta, a condition known as Eisenmenger syndrome. This reversal of the shunt occurs only after prolonged, untreated pulmonary overcirculation leads to vascular remodeling. In the initial presentation of PDA, pulmonary resistance is lower than systemic resistance, which is why pulmonary blood flow increases.
Test-taking strategy:
- Understand the pressure gradients in the heart: Left-sided or systemic structures such as the aorta always have higher pressure than right-sided or pulmonary structures such as the pulmonary artery in a normal postnatal heart.
- Apply the "Path of least resistance" rule: Blood will always flow from an area of high pressure to an area of low pressure if a hole (like a PDA) exists.
- Distinguish between fetal and neonatal circulation: In the womb, pulmonary resistance is high; after the first breath, it drops, which changes the direction of flow through the ductus.
- Connect the anatomical names to the flow: "Aorta to pulmonary artery" equals "systemic to pulmonary," which explains why the lungs get "flooded" with too much blood.
- Eliminate answers that suggest cyanosis unless the defect is specifically a right-to-left shunt. Since PDA increases pulmonary flow, it is initially an acyanotic condition.
Take home points
- PDA is an acyanotic heart defect because oxygenated blood is recirculated through the lungs.
- The magnitude of the shunt depends on the diameter of the ductus and the ratio of pulmonary to systemic vascular resistance.
- Chronic left-to-right shunting through a PDA results in left atrial and left ventricular enlargement due to volume overload.
- Prostaglandin inhibitors like Indomethacin are used to promote ductal closure by Constricting the smooth muscle within the ductus wall.
A nurse attended a staff education program on congenital heart defects. The nurse identifies which of the following clients as at increased risk for patent ductus arteriosus? Select all that apply
Explanation
Patent ductus arteriosus (PDA) occurs when the fetal connection between the aorta and the pulmonary artery fails to close after birth. While the exact cause can be multifactorial, certain environmental and physiological factors significantly interfere with the smooth muscle constriction required to seal the ductus.
Rationale for correct answers:
A. Prematurity is the most common risk factor for PDA. In preterm neonates, the muscular layer of the ductus is underdeveloped and less sensitive to the increase in arterial oxygen tension that normally triggers closure. Additionally, premature infants often have higher levels of circulating prostaglandins, which act as vasodilators to keep the ductus open.
B. Maternal infection with the rubella virus during the first trimester of pregnancy is a well-documented cause of "Congenital rubella syndrome." This syndrome frequently results in a triad of defects: cataracts, deafness, and cardiac anomalies, most notably PDA and pulmonary artery stenosis.
E. At high altitudes, the partial pressure of atmospheric oxygen is lower. Because the closure of the ductus arteriosus is primarily triggered by a rise in the infant's blood oxygen levels (PaO2) after the first breath, infants born in low-oxygen environments are statistically more likely to experience a persistent, open ductus.
Rationale for incorrect answers:
C. Marfan syndrome is a connective tissue disorder that primarily affects the aorta leading to dilation or aneurysm and the cardiac valves leading to mitral valve prolapse. It is not specifically associated with the failure of the ductus arteriosus to close during the neonatal period.
D. Birth weight itself is not a primary risk factor for PDA. In fact, small-for-gestational-age (SGA) infants or those with low birth weight (due to prematurity) are at much higher risk. Large-for-gestation-age (LGA) infants, often born to diabetic mothers, may face other cardiac issues like hypertrophic cardiomyopathy, but PDA is not more prevalent in this group than in the general population.
Test-taking strategy:
- Think "oxygen": Since oxygen closes the ductus, any condition where oxygen is low (prematurity/lungs not ready) or atmospheric oxygen is low (high altitude) will increase risk.
- Recall classic associations: In nursing school, rubella = PDA is a classic "must-know" pairing.
- Rule out growth: Size usually doesn't dictate "shunts" unless it's related to the developmental age (prematurity).
Take home points
- The ductus typically closes functionally within 12–72 hours in full-term infants.
- In the NICU, Indomethacin (a prostaglandin inhibitor) is the go-to medication for these at-risk premature infants.
- Infants born at high altitudes may have a PDA incidence nearly 20 times higher than those born at sea level.
An infant is diagnosed with patent ductus arteriosus. Which drug should the nurse anticipate administering to attempt to close the defect?
Explanation
Patent ductus arteriosus (PDA) occurs when the fetal shunt between the pulmonary artery and the aorta fails to close after birth. In the womb, the ductus arteriosus allows blood to bypass the fluid-filled lungs. After birth, this vessel should constrict and close; if it remains open (patent), oxygenated blood from the high-pressure aorta shunts back into the pulmonary artery, flooding the lungs.
Rationale for correct answer:
D. Indomethacin (Indocin) or Ibuprofen are the primary pharmacological treatments used to close a PDA, particularly in premature infants. These medications are prostaglandin inhibitors (NSAIDs). Because circulating prostaglandins are responsible for keeping the ductus arteriosus open during fetal life, inhibiting them triggers the smooth muscle in the ductus to constrict and close the vessel.
Rationale for incorrect answers:
A. Digoxin (Lanoxin) is a cardiac glycoside used to increase the force of heart contractions and slow the heart rate. While it may be used to manage the symptoms of heart failure caused by a large PDA, it has no physiological mechanism to actually close the ductus arteriosus.
B. Prednisone is a corticosteroid used to reduce inflammation. It is not used in the treatment of PDA. While steroids are sometimes given to mothers before preterm birth to help mature the infant's lungs, they do not play a role in the postnatal closure of the ductus.
C. Furosemide (Lasix) is a loop diuretic used to remove excess fluid from the lungs and body. Like Digoxin, it manages the symptoms of pulmonary congestion (the "wet lungs") associated with PDA, but it does not address the underlying structural defect.
Test-taking strategy:
- Identify the goal: The question asks for a drug to close the defect, not just treat the symptoms.
- Recall the "P" Rule:
- To keep it Patent (open): Give Prostaglandins.
- To close it: Give a Prostaglandin inhibitor (Indomethacin).
- Know your drug classes: Recognize Indomethacin and Ibuprofen as the "standard" non-surgical interventions for PDA in the NICU setting.
- Differentiate treatment vs. management: Diuretics and inotropes (Digoxin/Lasix) manage the effects of the hole; NSAIDs treat the hole itself.
Take home points
- Indomethacin is most effective when administered in the first few days of life.
- Before giving Indomethacin, the nurse must check the infant's renal function (urine output and creatinine) and platelet count, as the drug can cause kidney injury and increase the risk of bleeding.
- If pharmacological treatment fails, the infant may require surgical ligation or a transcatheter occlusion.
Comprehensive Questions
An infant age 2-months has a tentative diagnosis of congenital heart defect. During physical assessment, the nurse notes that the infant has a pulse rate of 168 beats/minute and a respiratory rate of 72 breaths/ minute. In which position should the nurse place the infant?
Explanation
The infant in this scenario is demonstrating significant clinical signs of congestive heart failure (CHF) and respiratory distress. A normal heart rate for a 2-month-old is typically 100–160 beats/minute, and a normal respiratory rate is 30–60 breaths/minute. The infant's values (168 bpm and 72 breaths/min) indicate that the heart is struggling to meet metabolic demands and the lungs are likely congested.
Rationale for the correct answer:
A. Upright in an infant seat is the most appropriate position. Placing the infant in a semi-upright position (approximately 45 degrees) uses gravity to lower the diaphragm, allowing for maximum chest expansion and easier breathing. Furthermore, this position decreases venous return to the heart, which helps alleviate pulmonary congestion and reduces the workload on an already stressed cardiac system.
Rationale for incorrect answers:
B. Lying on the back is contraindicated for an infant in respiratory distress. In this position, the abdominal organs push upward against the diaphragm, restricting lung expansion. Additionally, fluid in the lungs (pulmonary edema) tends to pool more broadly, worsening the ventilation-perfusion mismatch and increasing the infant's work of breathing.
C. Lying on the abdomen is dangerous for a 2-month-old infant due to the high risk of sudden infant death syndrome (SIDS). While the prone position can sometimes improve oxygenation in controlled intensive care settings (prone positioning therapy), it is not a standard nursing intervention for a general cardiac assessment and significantly restricts the nurse's ability to observe the infant's face and respiratory effort.
D. Sitting in high Fowler’s position (90 degrees) is generally too extreme for a 2-month-old. Infants this age lack the head and trunk control to maintain a strictly vertical position. Attempting to keep an infant at 90 degrees usually results in the head falling forward onto the chest, which can occlude the airway and worsen the respiratory distress.
Test-taking strategy:
- Identify the physiological stress: The vital signs (168 bpm, 72 breaths/min) tell you the infant is "working too hard."
- Prioritize breathing: In pediatric nursing, always choose the position that makes breathing easier. "Upright" is the universal answer for respiratory distress.
- Safety and development: Rule out positions that the infant cannot developmentally support (90-degree sitting) or that violate safety protocols such as prone sleeping.
- Preload management: Remember that in heart failure, you want to "keep the fluid down", away from the heart or lungs hence an upright tilt achieves this.
Take home points
- High heart and respiratory rates in an infant with a heart defect suggest the onset of congestive heart failure.
- Positioning is a non-pharmacological intervention to improve cardiac output and oxygenation.
- Nurse's Role: Monitor for "red flags" like perioral cyanosis, nasal flaring, and grunting while the infant is in the seat.
A nurse is caring for a 1-month-old client. Which factor indicating a cardiac defect might be found when assessing this client?
Explanation
Congenital heart defects in infants frequently result in a state of congestive heart failure or chronic hypoxemia, which significantly increases the basal metabolic rate. The physiological stress of maintaining cardiac output leads to sympathetic activation, resulting in diaphoresis and tachycardia. Because feeding is the most strenuous physical activity for a neonate, infants with cardiac pathology often exhibit exercise intolerance manifested during nursing or bottle-feeding sessions.
Rationale for correct answer:
C. Poor nutritional intake occurs because the infant becomes easily fatigued during the physical act of sucking and swallowing. This "feeding exhaustion" is a critical clinical marker; the infant may take a few sucks, stop to catch their breath, and eventually fall asleep before consuming adequate volume. This cycle leads to failure to thrive, as the caloric intake cannot compensate for the high metabolic demands of the heart.
Rationale for incorrect answers:
A. Infants with structural heart defects typically experience weight loss or difficulty gaining weight rather than excessive weight gain. While fluid retention can cause a deceptive increase in weight, the overarching clinical picture is one of malnutrition due to energy imbalance. In the context of a 1-month-old, a lack of weight gain along the expected growth curve is a major red flag for cardiac dysfunction.
B. Hyperactivity is not a finding associated with congenital heart disease; instead, these infants often present with lethargy and weakness. The limited cardiac reserve means the infant must conserve energy for vital organ perfusion, leading to decreased activity levels. Parents often report that the child is "too quiet" or sleeps excessively because they lack the oxygen delivery required for normal infant alertness.
D. Pink mucous membranes are a sign of adequate systemic oxygenation and are considered a normal finding in healthy infants. Many cardiac defects, especially cyanotic ones like tetralogy of Fallot, result in cyanosis or a dusky blue hue to the membranes. Even in acyanotic defects, poor perfusion can lead to pallor, making "pink" an unlikely indicator of an underlying structural heart defect.
Test-taking strategy:
- Focus on infant milestones: The primary role of a 1-month-old is to eat and grow; therefore, any defect will likely manifest as a failure in these two areas.
- Apply the energy budget principle: A heart defect requires extra energy from the rest of the body hence look for answers that describe a lack of energy such as fatigue or poor feeding.
- Distinguish between symptoms and health: Pink membranes and weight gain (under normal circumstances) signify health, whereas the question asks for indicators of a defect.
- Recognize metabolic reality: Structural heart disease is an "energy-wasting" condition; hyperactivity is physiologically impossible when the heart cannot meet basic oxygen demands.
- Use clinical priorities: In pediatric nursing, "poor feeding" is often the first subtle sign of a major systemic issue, including cardiac, respiratory, or infectious crises.
Take home points
- Feeding difficulties in infants with heart defects are often accompanied by forehead sweating (diaphoresis) and tachypnea.
- Failure to thrive is defined as a weight that falls below the 5th percentile or crosses two major growth percentiles.
- Small, frequent feedings and high-calorie formulas are used to reduce the workload on the heart while promoting growth.
- Nurses should observe a full feeding session to assess for "suck-rest-suck" patterns and signs of respiratory distress.
The nurse is caring for an infant with a heart defect that involves increased pulmonary blood flow. Which illustration shows a congenital heart disorder with increased pulmonary blood flow?
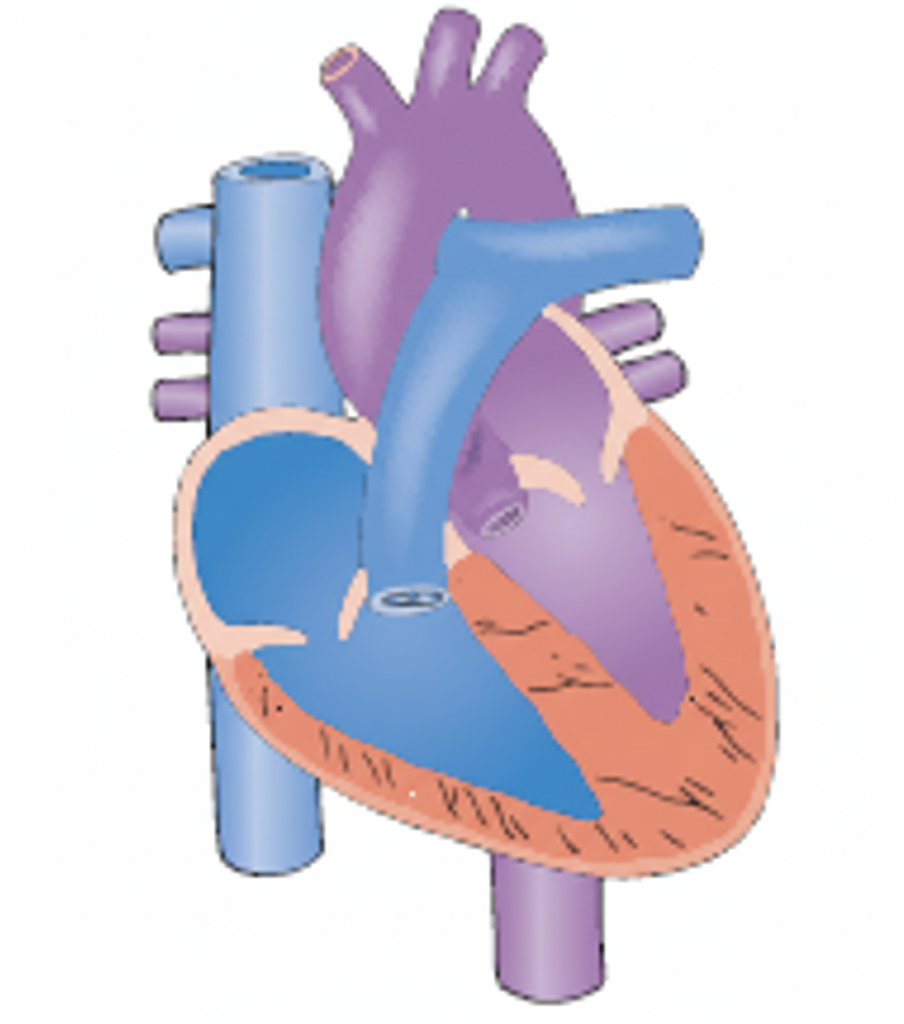
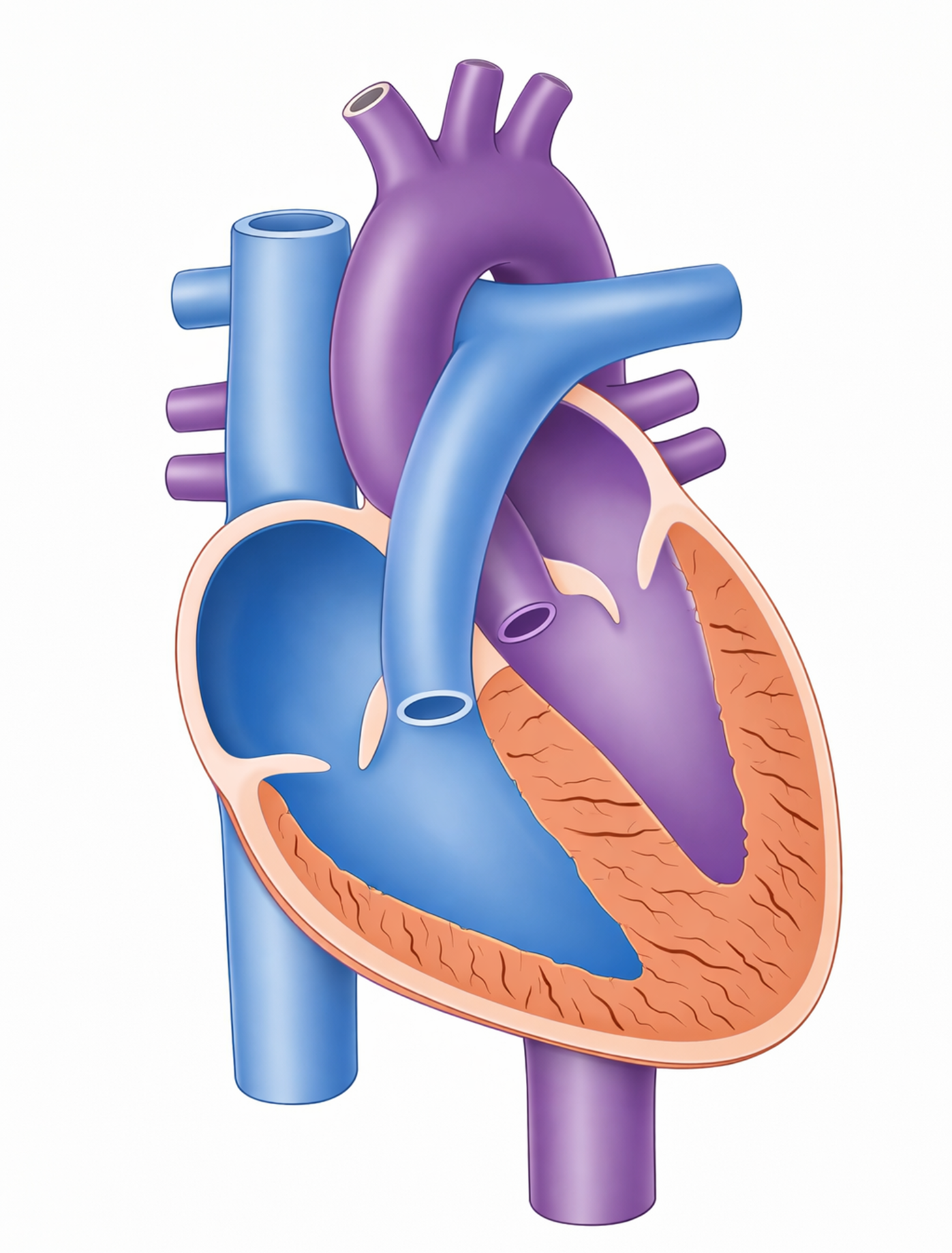
B.
. 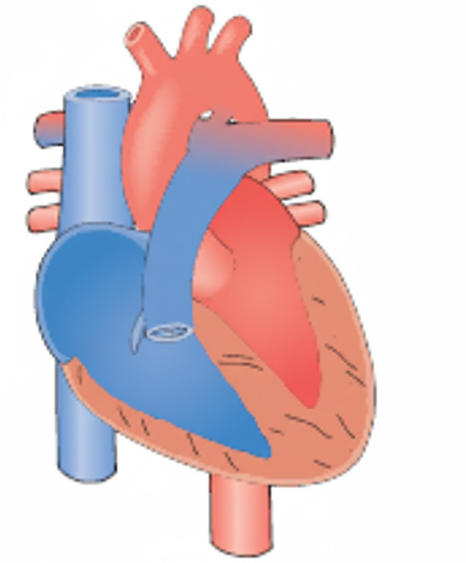
C.
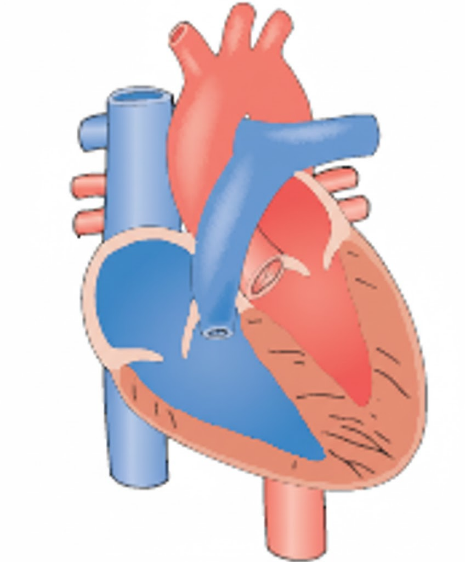
D.
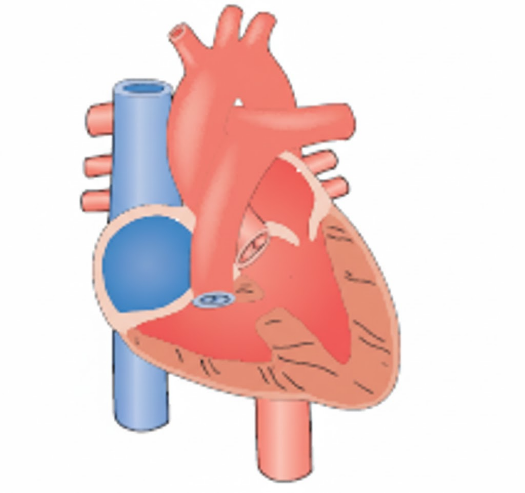
Explanation
Congenital heart defects are generally classified by their hemodynamic effect, whether they increase pulmonary blood flow, decrease it, or obstruct systemic flow. Defects that increase pulmonary blood flow (acyanotic defects) involve a left-to-right shunt, where oxygenated blood from the high-pressure left side of the heart leaks into the low-pressure right side, sending extra blood to the lungs.
Rationale for the correct answer:
B. Patent ductus arteriosus (PDA) is the correct choice. In this defect, a fetal vessel remains open between the aorta and the pulmonary artery. Because the aorta is a high-pressure system, blood is forced into the pulmonary artery, significantly increasing the volume of blood flowing to the lungs. This "flooding" of the lungs leads to pulmonary congestion and increased respiratory effort.
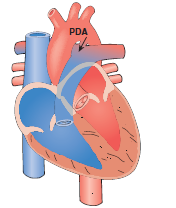
Rationale for incorrect answers:
A. Aortic stenosis is an obstructive defect. It involves a narrowing of the aortic valve, which makes it harder for the left ventricle to pump blood to the body. It does not involve a shunt that increases blood flow to the lungs but rather, it increases the workload (pressure) of the left ventricle.
Image Title: Aortic Stenosis
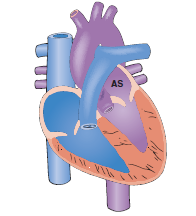
C. Pulmonic stenosis is also an obstructive defect. The narrowing occurs at the pulmonary valve. This actually decreases or restricts blood flow into the lungs because the right ventricle cannot easily push blood past the narrowed valve.
D. Tricuspid atresia is a cyanotic defect that typically results in decreased pulmonary blood flow. In this condition, the tricuspid valve fails to develop, meaning no blood can flow directly from the right atrium to the right ventricle. Blood must cross through an ASD to the left side, often bypassing the lungs unless a secondary defect (like a VSD) is present.
Test-taking strategy:
- Categorize by flow:
- Increased flow (L-to-R shunts): PDA, VSD, ASD, AV Canal.
- Decreased flow (R-to-L shunts/obstruction): Tetralogy of Fallot, tricuspid atresia.
- Obstruction to body: Coarctation of the Aorta, Aortic Stenosis.
- Visualize the "Hole": If there is a shunt and the baby is pink (acyanotic), blood is almost certainly moving toward the lungs, increasing flow.
- Identify "stenosis": The word "stenosis" means narrowing/blockage. Blockages usually restrict flow rather than increase it.
Take home points
- Increased pulmonary blood flow leads to "wet lungs," which manifests as crackles, tachypnea, and frequent respiratory infections.
- These infants are at high risk for congestive heart failure (CHF).
- Long-term untreated increased flow can lead to permanent damage to the pulmonary blood vessels, that is pulmonary hypertension.
A 3-year-old child is on postoperative day 5 for an atrial septal defect repair. Which nursing diagnosis would be most appropriate?
Explanation
Postoperative recovery following cardiac surgery involves a significant metabolic demand while the cardiovascular system adapts to corrected hemodynamics. During the initial recovery phase, the child often experiences a reduction in physiologic reserve, leading to an inability to endure or complete required daily activities. This functional impairment is exacerbated by surgical trauma, residual effects of anesthesia, and the temporary deconditioning associated with the inflammatory response triggered by cardiopulmonary bypass.
Rationale for correct answer:
A. Activity intolerance is the most appropriate diagnosis because the child is likely to experience fatigue and dyspnea when increasing physical exertion on day E. Surgical recovery requires balancing the need for progressive mobilization with the heart's limited ability to increase cardiac output. The nurse must monitor for signs such as tachycardia or diaphoresis, which indicate that the child’s current activity level exceeds their physiological tolerance.
Rationale for incorrect answers:
B. Chronic pain is defined as pain lasting longer than 3 to 6 months, which is not applicable to a patient on postoperative day E. The pain experienced in the immediate aftermath of an atrial septal defect repair is classified as acute pain resulting from tissue injury. Management focuses on analgesic administration to facilitate recovery, whereas chronic pain would involve a much more complex, long-term multidisciplinary approach to pain modulation.
C. Social isolation is generally not a primary nursing diagnosis for a 3-year-old in the acute phase of surgical recovery. While the hospital environment limits normal peer interactions, the presence of caregivers and child life specialists typically mitigates the risk of true social withdrawal. Efforts are focused on developmental play within the room rather than addressing a profound lack of social contact or belonging.
D. Risk for imbalanced fluid volume is a high priority in the first 24 to 48 hours but usually stabilizes by day E. By this stage, the patient has typically transitioned to oral intake and the initial risks of third-spacing or acute renal failure have diminished. While fluid status is always monitored, activity intolerance becomes the more prominent functional challenge as the child begins to navigate the hospital environment.
Test-taking strategy:
- Distinguish between actual and risk diagnoses. On day 5, the physical impact of surgery is an actual state affecting the patient's performance.
- Evaluate the timeframe provided; "day 5" suggests the patient is moving beyond the critical acute stabilization phase and into the functional recovery phase.
- Differentiate between acute and chronic descriptors. Surgical pain is a textbook example of acute pain, making any "chronic" label automatically incorrect.
- Focus on the developmental stage, for a 3-year-old, the drive to move and play is often hindered by the physical limitations of a healing sternum.
- Prioritize physiological needs over psychosocial ones unless the stem specifically describes behaviors indicating withdrawal or lack of support systems.
- Recognize that activity intolerance is a common sequela of any major thoracic surgery involving a temporary decrease in respiratory and cardiac efficiency.
Take home points
- Activity intolerance is characterized by an insufficient physiological energy to endure or complete necessary or desired daily activities.
- Postoperative day 5 marks a transition where nurses encourage increased ambulation while closely monitoring vital sign stability.
- Acute pain is the expected finding post-surgery, while chronic pain is an inappropriate label for a patient in the first week of recovery.
- Nursing interventions for activity intolerance include clustering care and providing frequent rest periods between physical therapy sessions.
A nurse is educating the parents of a child with an atrial septal defect regarding the child’s condition.
Which of the following information would be appropriate for the nurse to provide?
Explanation
Atrial septal defect is a non-cyanotic congenital heart lesion characterized by a deficiency in the interatrial septum. This structural opening allows for a left-to-right shunt because the pressure in the left atrium is chronically higher than that in the right atrium. The resulting volume overload leads to dilation of the right atrium and right ventricle, potentially causing atrial arrhythmias and a palpable parasternal lift on physical examination.
Rationale for correct answer:
C. In an atrial septal defect, oxygenated blood shunts from the left atrium back into the right atrium, essentially recirculating through the lungs. This creates an inefficient circuit where the right side of the heart must pump an increased stroke volume to accommodate both systemic return and the shunted blood. The increased workload on the right ventricle can eventually lead to heart failure if the defect is hemodynamically significant and remains unrepaired.
Rationale for incorrect answers:
A. Cyanosis typically occurs in defects that allow deoxygenated blood to bypass the lungs and enter the systemic circulation via a right-to-left shunt. In an uncomplicated atrial septal defect, the shunt is left-to-right, meaning the systemic blood remains fully oxygenated. If a patient with an atrial septal defect becomes cyanotic, it usually indicates the development of Eisenmenger syndrome, where pulmonary pressures have surpassed systemic pressures.
B. A hole between the aorta and the pulmonary artery describes a patent ductus arteriosus, not an atrial septal defect. While both conditions involve a left-to-right shunt, the anatomic location and the timing of the associated murmur differ significantly. The murmur of an atrial septal defect is actually a systolic ejection murmur heard at the upper left sternal border, caused by increased flow across the pulmonic valve.
D. A high number of red blood cells, or polycythemia, is a compensatory mechanism found in cyanotic heart disease to improve oxygen-carrying capacity. It does not cause a slowed heart rate; rather, infants with heart defects often exhibit tachycardia as a compensatory response to decreased cardiac efficiency. Chronic hypoxemia triggers erythropoietin production, but this is not a feature of simple, acyanotic atrial septal defects where systemic oxygenation is normal.
Test-taking strategy:
- Focus on the physiological consequence of the shunt. Left-to-right flow always results in pulmonary overcirculation and increased cardiac work.
- Eliminate options that describe anatomical misplacements of other defects, such as the patent ductus arteriosus or transposition of the great arteries.
- Recognize that cyanosis is an "early" sign only in right-to-left shunts; in left-to-right shunts, it is a very late and grave sign.
- Use the principle of hemodynamics to understand that blood follows the path of least resistance from high-pressure chambers to lower-pressure chambers.
- Rule out any choices that suggest bradycardia is a standard compensatory mechanism for congenital heart disease, as the opposite is usually true.
Take home points
- Atrial septal defect causes a left-to-right shunt, leading to increased pulmonary blood flow and right-sided heart volume overload.
- Most children with an ASD are asymptomatic but may present with a characteristic fixed split second heart sound (S2).
- Long-term complications of an unrepaired ASD include pulmonary hypertension, atrial fibrillation, and right-sided heart failure.
- Surgical or percutaneous closure is usually recommended in preschool-aged children to prevent adult-onset cardiovascular complications.
A nurse is educating student nurses on congenital heart defects. Eisenmenger’s complex consists of pulmonary vascular resistance exceeding systemic pressure. This can occur in which cardiac anomaly when untreated?
Explanation
Eisenmenger syndrome represents the end-stage physiological transition of a long-standing, unrepaired left-to-right shunt. Chronic exposure of the pulmonary vasculature to high-volume and high-pressure blood flow induces endothelial injury and permanent structural remodeling of the pulmonary arterioles. This results in pulmonary hypertension so severe that pulmonary vascular resistance eventually exceeds systemic vascular resistance. At this critical threshold, the shunt direction reverses to right-to-left, allowing deoxygenated blood to enter the systemic circulation, causing secondary erythrocytosis and central cyanosis.
Rationale for correct answer:
D. Ventricular septal defect is the primary precursor to Eisenmenger syndrome due to the high-pressure systolic shunting from the left ventricle directly into the pulmonary circuit. This direct transmission of systemic pressure accelerates vascular sclerosis and medial hypertrophy far more rapidly than atrial-level shunts. If a large ventricular septal defect remains unrepaired, the transition to irreversible hypertension can occur by late childhood or early adolescence.
Rationale for incorrect answers:
A. Aortic stenosis is an obstructive lesion of the left ventricular outflow tract that increases afterload on the left ventricle. It does not involve a septal communication or an initial left-to-right shunt, which is a prerequisite for Eisenmenger syndrome. The primary pathophysiology involves concentric hypertrophy of the left ventricle rather than pulmonary overcirculation and subsequent vascular remodeling.
B. Atrial septal defect allows oxygenated blood to flow from the left atrium to the right atrium, increasing pulmonary volume. Although it can lead to Eisenmenger syndrome, it typically takes decades to develop because the pressure gradient is significantly lower than in ventricular defects. In the context of a student education program, the ventricular septal defect is the classic and most frequent example of high-pressure progression.
C. Pulmonary stenosis involves an obstruction of blood flow from the right ventricle to the pulmonary arteries, which actually protects the lungs from high pressure. This condition results in decreased pulmonary flow and right ventricular pressure overload, the opposite of the overcirculation seen in shunting defects. Because the pulmonary vascular bed is shielded from high pressures, it cannot develop the vasculopathy required for shunt reversal.
Test-taking strategy:
- Define the core requirement: Eisenmenger syndrome requires a pre-existing "hole" that initially shunts blood from the left side to the right side.
- Identify acyanotic defects: Only defects that start as left-to-right shunts (VSD, ASD, PDA) can "reverse" into the Eisenmenger complex.
- Rule out obstructive lesions: Stenosis (aortic or pulmonic) involves narrowing of valves/vessels without a primary inter-chamber shunt pathway.
- Apply the pressure-flow logic: Eisenmenger is a disease of "too much flow" leading to "too much resistance"; look for defects that flood the lungs.
- Differentiate by pressure intensity: Ventricular shunts are higher pressure than atrial shunts and thus more likely to cause rapid vascular damage.
- Use process of elimination: If a defect does not allow blood to cross from the left heart to the right heart, it cannot be the correct answer.
Take home points
- Eisenmenger syndrome is characterized by the reversal of a left-to-right shunt to a right-to-left shunt due to severe pulmonary hypertension.
- Clinical manifestations include clubbing, cyanosis, and polycythemia due to chronic systemic hypoxemia.
- Once Eisenmenger syndrome develops, surgical repair of the original defect is generally contraindicated as the shunt acts as a "relief valve."
- Management focuses on pulmonary vasodilators and preventing complications like brain abscesses or hyperviscosity syndrome.
A nurse is caring for a child with a ventricular septal defect. Which curative surgical intervention is recommended for this client?
Explanation
Ventricular septal defect represents a pathological opening in the interventricular septum allowing for a left-to-right shunt of blood between the lower chambers. The definitive management of hemodynamically significant defects involves the anatomical closure of the orifice to restore ventricular independence and normalize pulmonary pressures. Surgical correction is typically performed under cardiopulmonary bypass, ensuring a bloodless field for precise visualization and repair of the septal wall while protecting myocardial function.
Rationale for correct answer:
C. The curative treatment for a ventricular septal defect is surgical closure using either a purse-string suture for small defects or a Dacron patch for larger ones. During this procedure, the surgeon secures the synthetic patch over the defect using permanent sutures, which eventually becomes covered by the heart's own endocardial tissue. This mechanical barrier effectively eliminates the left-to-right shunt, thereby preventing further pulmonary overcirculation and right ventricular volume overload.
Rationale for incorrect answers:
A. Pulmonary artery banding is a palliative procedure, not a curative one, used to reduce pulmonary blood flow and protect the lungs in infants who are too small or unstable for definitive repair. This intervention involve placing a constricting band around the main pulmonary artery to increase resistance and decrease the volume of the shunt. While it manages symptoms and prevents pulmonary vasculopathy, the original septal defect remains open and requires a second, definitive surgery later.
B. While some secundum atrial septal defects are easily closed via cardiac catheterization, most ventricular septal defects, particularly those in the perimembranous region, are traditionally managed through open-heart surgery. Transcatheter closure of a VSD carries a higher risk of interfering with the atrioventricular node and causing permanent heart block due to the proximity of the conduction system. While device closure is an option for certain muscular VSDs, it is not the standard curative recommendation for the majority of cases.
D. Delaying surgery until severe pulmonary hypertension is noted is contraindicated and dangerous, as it increases the risk of developing irreversible Eisenmenger syndrome. Once pulmonary vascular resistance becomes fixed and elevated, surgical closure of the defect may lead to acute right-sided heart failure and death because the shunt no longer acts as a pressure-relief valve. The goal of intervention is to repair the defect before permanent damage to the pulmonary vasculature occurs to ensure a normal life expectancy.
Test-taking strategy:
- Distinguish between curative and palliative terminology; "banding" is always palliative in the context of septal defects.
- Identify the standard of care for VSDs, which remains surgical patch repair due to the complex anatomy of the ventricular conduction system.
- Apply the principle of timing in cardiac surgery: intervention should occur before the onset of irreversible complications like severe pulmonary hypertension.
- Recognize the materials used in cardiac repair; Dacron and Gore-Tex are classic synthetic materials mentioned in nursing exams for patching holes.
- Differentiate between atrial and ventricular catheterization limits. Remember that VSDs are harder to "plug" safely than ASDs due to their location near heart valves and nerves.
- Rule out options that describe reactive treatments that is waiting for hypertension before management versus preventative curative measures.
Take home points
- Dacron patch repair is the gold standard curative treatment for large, symptomatic ventricular septal defects.
- Pulmonary artery banding is a temporary measure used to bridge high-risk infants to a future definitive repair.
- Surgery is ideally performed before the age of 2 years to prevent permanent pulmonary vascular remodeling.
- Postoperative monitoring must include assessment for heart block or arrhythmias due to surgical proximity to the Bundle of His.
A child with a ventricular septal defect repair is receiving dopamine (Intropin) post-operatively. The nurse should teach the child’s parents that this medication is most likely to be given for which action?
Explanation
Dopamine is a naturally occurring catecholamine and potent sympathomimetic agent frequently utilized in the postoperative management of pediatric cardiac patients. It acts as a precursor to norepinephrine and stimulates dopaminergic, beta-1 adrenergic, and alpha-1 adrenergic receptors in a dose-dependent manner. In the context of a ventricular septal defect repair, dopamine is primarily titrated to maintain hemodynamic stability by counteracting the transient myocardial depression often seen following cardiopulmonary bypass and surgical manipulation of the interventricular septum.
Rationale for correct answer:
C. Dopamine is administered to increase cardiac output through its stimulation of beta-1 adrenergic receptors in the myocardium. This stimulation results in positive inotropic effects, which increase the force of myocardial contraction and stroke volume. By enhancing the efficiency of the heart as a pump, dopamine ensures adequate systemic perfusion to vital organs during the critical recovery phase after the surgical patch has been placed.
Rationale for incorrect answers:
A. Dopamine does not decrease the heart rate but often causes tachycardia as a common side effect. Through its action on beta-1 receptors, it exerts a positive chronotropic effect, which increases the rate of sinoatrial node firing. A medication that decreases heart rate, such as a beta-blocker or digoxin, would be counterproductive if the primary goal is to support a struggling postoperative heart with low output.
B. One of the unique benefits of dopamine at low to moderate doses is its ability to increase, not decrease, urine output. By stimulating dopaminergic receptors in the renal vasculature, it promotes vasodilation and increases renal blood flow and glomerular filtration rate. Monitoring for increased urine output is a key nursing assessment to determine if the drug is successfully improving visceral perfusion and preventing acute kidney injury.
D. Dopamine increases, rather than decreases, cardiac contractility via its potent inotropic properties. A medication that decreases contractility would be a negative inotrope, which is typically avoided in the immediate postoperative period for a child who has just undergone a major cardiac repair. The objective post-VSD repair is to provide circulatory support to help the ventricles adjust to the new, corrected pressure gradients without the left-to-right shunt.
Test-taking strategy:
- Identify the drug class: Dopamine is an inotrope/vasopressor used in critical care to "boost" the heart and blood pressure.
- Analyze the intended outcome: In a postoperative cardiac setting, the goal is almost always to improve perfusion and support the heart's pumping ability that is cardiac output.
- Use the opposites rule: If you know dopamine is a "stimulant" for the heart, you can immediately eliminate any options containing "decrease" that is choices 1, 2, and 4).
- Differentiate between therapeutic effects and side effects; while dopamine can increase heart rate, its primary therapeutic reason for use in this scenario is contractility and output.
- Connect renal physiology to the drug: Recall the "renal dose" of dopamine which is famous for improving kidney blood flow and urine production.
- Recognize the surgical context: Cardiopulmonary bypass causes "stunned myocardium," making an agent that increases contractility essential for weaning off the pump.
Take home points
- Dopamine increases cardiac output by improving myocardial contractility and heart rate.
- At low doses, dopamine improves renal perfusion, leading to an increase in urine output.
- Tachycardia and cardiac arrhythmias are significant side effects that require continuous ECG monitoring.
- The medication must be administered via a central line whenever possible to prevent tissue necrosis from extravasation.
The parents of a 3-month-old ask why their baby will not have an operation to correct a ventricular septal defect (VSD). The nurse’s best response is:
Explanation
Ventricular septal defect is a congenital cardiac malformation involving an opening in the interventricular septum. The natural history of this condition is highly dependent on the anatomic location and size of the defect. Many small, muscular, or perimembranous defects undergo spontaneous closure as the heart grows and the septal tissue hypertrophies. If the infant remains hemodynamically stable without signs of congestive heart failure or pulmonary hypertension, a conservative "watchful waiting" approach is standard medical practice.
Rationale for correct answer:
B. Small ventricular septal defects have a high rate of spontaneous closure, with approximately 75% to 80% closing within the first 12 months of life. Because the defect is small, it does not produce significant pulmonary overcirculation or compromise the infant's growth and development. Informing the parents of this common outcome provides evidence-based reassurance and explains the physiological reasoning behind delaying invasive surgical intervention.
Rationale for incorrect answers:
A. Suggesting a second opinion is a non-therapeutic response that may undermine the parents' trust in their current healthcare team. While patients have a right to seek additional consultations, this statement fails to address the parents' specific question about the science of the condition. The nurse’s role is to provide accurate education regarding the standard of care for small cardiac defects rather than deferring the explanation to another provider.
C. Waiting until an infant develops respiratory distress is an incorrect and dangerous management strategy. The goal of cardiac management is to intervene before significant symptoms like tachypnea, grunting, or retractions occur to prevent permanent lung damage. If a child were to develop respiratory distress from a VSD, it would indicate that the defect is hemodynamically significant and requires prompt medical or surgical management, not further waiting.
D. Delaying surgery until 10 years of age is not the standard protocol for a small ventricular septal defect. If a small defect does not close on its own by early childhood, it usually remains clinically insignificant and may never require surgery at all. If surgery were required for a symptomatic defect, it is typically performed much earlier, often between 2 and 5 years of age, to prevent long-term complications such as endocarditis or aortic valve prolapse.
Test-taking strategy:
- Identify the natural history of the defect: Remember that "small" holes in the heart (ASD/VSD) often fix themselves through natural growth.
- Use therapeutic communication principles: Eliminate responses that are dismissive, undermine other professionals, or provide false/dangerous clinical timelines.
- Recognize standard pediatric timelines: The first year of life is the critical window for spontaneous closure of many congenital anomalies.
- Evaluate clinical triggers: Surgery is triggered by "failure to thrive" or "pulmonary hypertension," not by waiting for a child to reach a specific double-digit age like 10.
- Focus on parental education: The best response is one that explains the "why" behind a medical decision using specific, accurate physiological data.
- Rule out pessimistic options: Option 3 suggests waiting for a crisis, which contradicts the preventative nature of modern pediatric cardiology.
Take home points
- Small ventricular septal defects frequently close spontaneously during the first year of life without any intervention.
- Conservative management involves serial echocardiograms to monitor the size of the shunt and right ventricular pressures.
- Spontaneous closure is most common in muscular VSDs compared to other anatomical types.
- Prophylaxis for infective endocarditis is no longer routinely recommended for isolated, unrepaired small VSDs unless other criteria are met.
A baby, exhibiting no obvious signs of congestive heart failure, has been diagnosed with a small ventricular septal defect. Which of the following information should the nurse explain to the baby’s parents?
Explanation
Ventricular septal defect is a congenital cardiovascular malformation characterized by a communication in the interventricular septum. The embryological development of this septum occurs primarily between the 4th and 8th weeks of gestation. Small defects, particularly those located in the muscular portion of the septum, often undergo spontaneous closure as the heart grows and the surrounding muscular tissue hypertrophies. If the shunt volume is negligible, the infant remains hemodynamically stable without evidence of pulmonary overcirculation or myocardial strain.
Rationale for correct answer:
B. Small ventricular septal defects have a high probability of spontaneous closure, with approximately 75% to 80% closing within the first year of life. Since the infant exhibits no signs of congestive heart failure, the shunt is hemodynamically insignificant, requiring only periodic echocardiographic monitoring. Informing parents of this natural progression provides evidence-based reassurance and avoids unnecessary anxiety regarding invasive surgical or pharmacological interventions that are not currently indicated.
Rationale for incorrect answers:
A. Open-heart surgery within a week is only indicated for infants with life-threatening defects or intractable heart failure that does not respond to medical management. In an asymptomatic baby with a small defect, the risks of cardiopulmonary bypass and general anesthesia far outweigh the benefits of closing a hole that is likely to resolve on its own. Surgical repair is typically deferred until it is clear the defect will not close spontaneously and is causing pathological changes.
C. Cardiac development and septation occur during the first trimester, specifically between weeks 4 and 8 of post-conception age. Attributing the defect to the second trimester is embryologically incorrect, as the basic structures of the heart are already formed by that stage. Most congenital heart defects are the result of multifactorial inheritance or environmental triggers occurring during the highly sensitive period of organogenesis in early pregnancy.
D. High-calorie formula is a nutritional intervention reserved for infants who demonstrate failure to thrive or increased metabolic demands due to heart failure. Since this baby exhibits no signs of congestive heart failure, their caloric requirements should be similar to those of a healthy infant. Unnecessary caloric fortification can lead to excessive weight gain and gastrointestinal distress if the infant's metabolic rate is not actually elevated by cardiac pathology.
Test-taking strategy
- Categorize the severity: The stem states the defect is small and the infant is asymptomatic, which points toward a conservative management approach.
- Evaluate the natural history: Recall that "small" septal defects (VSD/ASD) are famous for their high rate of spontaneous resolution.
- Apply embryology facts: Eliminate any choice suggesting heart formation happens in the second or third trimester; the heart is the first functional organ and develops very early.
- Match the intervention to the symptom: Do not select "high-calorie formula" or "surgery" if the infant does not have the symptoms such as poor growth or heart failure, that justify them.
- Use the "least invasive" principle: In stable pediatric cases, the least invasive path (observation) is often the correct answer over immediate surgery.
- Rule out urgent timelines: Words like "within a week" are red flags in a question describing a stable, non-emergency clinical presentation.
Take home points
- Most small ventricular septal defects close spontaneously during the first year of life as cardiac tissue grows.
- Asymptomatic VSDs do not require immediate surgical intervention or aggressive nutritional supplementation.
- Congenital heart defects typically originate during the first 8 weeks of gestation.
- Periodic cardiology follow-up is necessary to ensure the defect does not enlarge or cause latent pulmonary hypertension.
A nurse is caring for a child with AV canal defect. Upon entering the room, the nurse sees the mother lying in bed with the child, who is lying flat and drinking from a bottle. Which intervention should be implemented?
Explanation
Atrioventricular canal defect, also known as endocardial cushion defect, involves a large hole in the center of the heart affecting both atrial and ventricular septa and the atrioventricular valves. This defect leads to massive left-to-right shunting and severe pulmonary overcirculation, causing early-onset heart failure and pulmonary hypertension. Infants with this condition have significantly increased metabolic demands and decreased respiratory reserve, making feeding an exhaustive activity that requires specific positioning to optimize oxygenation and prevent aspiration.
Rationale for correct answer:
B. Explaining the need for an upright position and frequent breaks addresses the infant's physiological struggle to coordinate sucking, swallowing, and breathing while in a high-flow state. Holding the child upright uses gravity to decrease venous return to the heart and reduce pressure on the diaphragm, which improves lung expansion. Frequent rest periods during feedings prevent hypoxia and minimize the caloric expenditure associated with the increased work of breathing.
Rationale for incorrect answers:
A. Immediately raising the child into an upright position without explanation is an abrupt intervention that may startle both the mother and the infant. While the position change is necessary for safety, the nurse should prioritize education and collaboration to ensure the caregiver understands the rationale for the change. Sudden movements in an infant with a fragile hemodynamic status can trigger unnecessary stress and increase myocardial oxygen consumption.
C. Taking the bottle from the mother is a non-therapeutic action that undermines the parent's role and can damage the nurse-client relationship. A more effective teaching strategy is to model the behavior or provide verbal guidance while the mother remains an active participant in the care. Empathetic education encourages caregiver confidence and ensures that the proper feeding techniques are continued after the nurse leaves the room.
D. Informing the mother she should not be in bed for safety reasons focuses on hospital policy rather than the immediate physiological needs of the cardiac infant. While co-sleeping is discouraged in hospital settings due to the risk of falls or suffocation, it does not address the primary issue of feeding-related respiratory distress. The priority must remain on aspiration prevention and reducing the workload of the heart during nutritional intake.
Test-taking strategy:
- Prioritize educational responses that empower the caregiver while addressing the immediate safety concern.
- Identify the pathophysiology of the defect: AV canal patients are "wet" and tired, so they need help breathing while eating.
- Rule out aggressive or rude actions, such as grabbing the bottle or reprimanding the parent, as these are never the "best" nursing response.
- Apply the ABCs of feeding: positioning affects the airway and breathing, which is the most critical intervention for a cardiac infant.
- Focus on the long-term goal of parental mastery; explaining the "why" ensures the behavior is repeated at home.
- Recognize that upright positioning is a standard nursing intervention for any child with pulmonary congestion or a high risk of aspiration.
Take home points
- Atrioventricular canal defects cause severe heart failure that necessitates high-energy feeding strategies.
- Upright positioning during feeding reduces the work of breathing and helps prevent pulmonary aspiration.
- Frequent rest periods during feedings allow the infant to recover from the high metabolic cost of eating.
- Nursing education should be delivered in a supportive, non-judgmental manner to foster effective home care.
A newborn with patent ductus arteriosus was delivered 6 hours earlier and is being held by his mother. As the nurse enters the room to assess the neonate’s vital signs, the mother says, “The physician says that my baby has a heart murmur. Does that mean he has a bad heart?” Which response by the nurse would be the most appropriate?
Explanation
In the first few hours of life, a newborn is in a state of physiologic transition. During fetal development, the ductus arteriosus is a necessary structure that shunts blood away from the non-functional, fluid-filled lungs directly into the aorta. Once the baby takes their first breath, pulmonary resistance drops, and the increase in arterial oxygen typically triggers the ductus to begin constricting. This process is not instantaneous and can take anywhere from 24 to 72 hours to complete.
Rationale for correct answer:
D. Telling the mother the murmur is caused by the natural opening, which can take a day or two to close and it’s a normal part of your baby’s transition is the most appropriate response because it provides evidence-based reassurance while educating the mother on normal neonatal physiology. Many newborns have "transient" murmurs as the ductus arteriosus and the foramen ovale transition toward closure. By framing it as a natural part of the transition, the nurse alleviates the mother's anxiety without dismissing her concerns or pathologizing a potentially normal finding at the 6-hour mark.
Rationale for incorrect answers:
A. Telling a mother that the baby needs "more tests" at this stage is likely to increase her anxiety significantly. While follow-up (like an echocardiogram) might be necessary if the murmur persists or symptoms of heart failure develop, it is not the primary explanation for a murmur heard so shortly after birth.
B. Stating that the baby will require home oxygen therapy is premature and likely incorrect. A patent ductus arteriosus (PDA) is an acyanotic defect (left-to-right shunt), meaning the baby is getting plenty of oxygen to the body. Oxygen therapy is not a standard treatment for an isolated PDA and suggests a much graver condition than a transitional murmur.
C. "He'll be fine. Don't worry about him" is a classic example of non-therapeutic communication. It offers false reassurance and shuts down the mother's expression of concern. It fails to provide the information the parent is seeking and minimizes her feelings, which can damage the nurse-client relationship.
Test-taking strategy
- Identify the timing: The baby is only 6 hours old. In nursing exams, cardiac findings in the first 24 hours are often related to "transitional" changes rather than permanent defects.
- Choose therapeutic communication: Look for the answer that educates the parent while remaining calm and supportive.
- Rule out "Alarmist" responses: Options 1 and 2 jump to conclusions about serious illness or long-term treatment before the baby has even finished transitioning.
- Avoid false reassurance: Rule out any option that tells the parent "don't worry" without providing a scientific reason why.
Take home points
- A "continuous, machinery-like murmur" is the classic description of a PDA.
- Physical closure of the ductus arteriosus in a term infant usually occurs within 48 hours of birth.
- Nursing assessment at this stage focuses on the baby's color, feeding tolerance, and respiratory effort rather than just the sound of the murmur.
A nurse is preparing to administer indomethacin (Indocin®) to an infant diagnosed with patent ductus arteriosus (PDA). By which route should the nurse expect to administer the indomethacin for this infant?
Explanation
In the neonatal intensive care unit (NICU) setting, Indomethacin (Indocin IV®) is the gold standard pharmacological treatment for the closure of a patent ductus arteriosus (PDA) in premature infants. While Indomethacin exists in various forms for adults (such as oral capsules or rectal suppositories for inflammatory conditions), the specialized formulation used for neonates is designed for precise, controlled delivery to the systemic circulation.
Rationale for correct answer:
A. Intravenously (IV) is the expected route for an infant with a PDA. Intravenous administration ensures 100% bioavailability and rapid therapeutic levels in the bloodstream, which is critical for inducing the constriction of the ductus arteriosus. Most infants requiring this medication are premature and may have compromised gastrointestinal absorption (risk of necrotizing enterocolitis) or are NPO (nothing by mouth), making the IV route the safest and most reliable method for drug delivery.
Rationale for incorrect answers:
B. Orally is generally avoided for Indomethacin in neonates with PDA. Premature infants often have immature digestive systems, and oral administration is less predictable due to variable absorption rates. Furthermore, Indomethacin can decrease blood flow to the mesenteric arteries, and giving it orally could potentially increase the risk of intestinal irritation or injury in a fragile newborn.
C. Rectally is not a standard route for this specific clinical indication. While rectal suppositories exist for older children and adults for pain or fever, they do not allow for the precise titration and consistent serum levels required to manage a cardiac shunt in a neonate.
D. Intramuscularly (IM) is not used for Indomethacin. IM injections are painful and can cause local tissue irritation or hematomas, especially in premature infants who may have low muscle mass and fluctuating platelet counts. Additionally, the absorption from muscle tissue is not as consistent or rapid as the intravenous route.
Test-taking strategy:
- Consider the patient population: Most babies with symptomatic PDA are premature neonates. In the NICU, the IV route is the standard for critical medications to ensure accuracy and bypass the immature GI tract.
- Identify the goal: Closing a cardiac shunt is an acute intervention. Acute interventions in hospital settings almost always favor the IV route for speed and control.
- Safety first: Remember that NSAIDs like Indomethacin can be hard on the stomach and kidneys; IV administration allows for the most controlled "dosage-to-effect" ratio.
Take home points
- Indomethacin is usually given in a series of three doses at specific intervals (usually 12 to 24 hours apart).
- The nurse must monitor urine output extremely closely during IV Indomethacin therapy, as the drug can cause significant renal vasoconstriction.
- Do not administer if the infant has active bleeding, especially intracranial hemorrhage, a significantly low platelet count, or suspected necrotizing enterocolitis (NEC).
The parents of a newborn child have just been told that he has a heart defect known as patent ductus arteriosus. Which statement made by the parents indicates that teaching has been effective?
Explanation
Patent ductus arteriosus (PDA) involves the persistence of a fetal vessel that connects the pulmonary artery to the aorta. While the functional closure of this vessel typically occurs within the first 24 to 72 hours of life in full-term infants, the permanent anatomical closure, where the vessel turns into a ligament, usually completes by 2 to 3 weeks, or certainly by 6 weeks of age.
Rationale for correct answer:
B. The statement that the ductus normally closes by 6 weeks of age shows that the parents understand the natural timeline for neonatal transition. In healthy infants, the increase in arterial oxygen and the drop in circulating prostaglandins after birth trigger the smooth muscle of the ductus to constrict. If this process is delayed, it is considered a "patent" ductus, but the biological expectation remains closure within the first month or two of life.
Rationale for incorrect answers:
A. Heart failure is actually a common complication of a large patent ductus arteriosus. Because the aorta has much higher pressure than the pulmonary artery, oxygenated blood is "stolen" from the body and forced back into the lungs. This volume overload causes the heart to work harder, eventually leading to symptoms of congestive heart failure (CHF) like tachypnea and poor feeding.
C. An open ductus causes increased blood flow to the lungs, not decreased. This is a left-to-right shunt. The lungs are effectively "flooded" with blood that has already been oxygenated, which can lead to pulmonary congestion and respiratory distress.
D. PDA is an acyanotic defect with increased pulmonary blood flow. Because the blood being shunted into the pulmonary system is already oxygenated (from the aorta), the baby’s skin remains pink (acyanotic). Decreased pulmonary blood flow is seen in "blue baby" defects like tetralogy of Fallot.
Test-taking strategy
- Identify the shunt type: PDA is a "Left-to-Right" shunt. Left-to-right always means increased pulmonary flow and acyanosis (pink baby).
- Connect the anatomy: The ductus connects the aorta (high pressure) to the pulmonary artery (low pressure). Blood moves from high to low, flooding the lungs.
- Recall the timeline: Neonatal "holes" (PDA, foramen ovale) are transitional. If they stay open past the first few weeks, they are considered defects.
- Eliminate "cyanotic": If the blood in the aorta is already oxygenated, a shunt out of the aorta can't make the baby turn blue.
Take home points
- PDA is often associated with a "machinery-like" murmur heard at the upper left sternal border.
- Medical closure is often attempted with Indomethacin or Ibuprofen in premature infants.
- If it remains open, it can lead to pulmonary hypertension and frequent respiratory infections.
A nurse is caring for a client with patent ductus arteriosus. Which assessment finding is consistent with this diagnosis?
Explanation
Patent ductus arteriosus (PDA) is a condition where the fetal vascular connection between the main pulmonary artery and the aorta fails to close after birth. Because the aorta is a high-pressure system and the pulmonary artery is a low-pressure system, blood shunts continuously from the aorta into the lungs throughout the entire cardiac cycle, both systole and diastole.
Rationale for correct answer:
B. The machinelike murmur, also called a continuous or Gibson murmur, is the hallmark physical finding of a PDA. Unlike other septal defects that may only produce sound during contraction, the pressure gradient between the aorta and pulmonary artery persists during both contraction (systole) and relaxation (diastole). This results in a "to-and-fro" or "machinery" sound that is usually best heard at the upper left sternal border or under the left clavicle.
Rationale for incorrect answers:
A. Weak peripheral pulses are not characteristic of PDA. Instead, infants with a significant PDA often have bounding peripheral pulses. Because a large volume of blood leaves the aorta and "runs off" into the pulmonary artery during diastole, the diastolic blood pressure drops significantly, making the pulse feel very strong or "leaping."
C. PDA causes a widened pulse pressure, not a narrowed one. Pulse pressure is the difference between the systolic and diastolic blood pressure. In PDA, the systolic pressure remains normal or slightly elevated, while the diastolic pressure drops due to the blood "shunting" away from the systemic circulation into the lungs. This creates a large (wide) gap between the two numbers.
D. Right ventricular hypertrophy is less common as an initial finding than left-sided changes. In PDA, the extra blood shunted to the lungs returns to the left atrium and left ventricle, causing volume overload and left ventricular hypertrophy. Right-sided hypertrophy typically only develops much later if the patient develops pulmonary hypertension (Eisenmenger syndrome).
Test-taking strategy:
- Keywords: Associate "PDA" with "machinery-like murmur" and "bounding pulses."
- Pressure physics: If blood is "leaking" out of the aorta into the lungs during the "resting" phase (diastole), the diastolic pressure will fall, which always widens the pulse pressure.
- Location: Remember that PDA is an acyanotic (left-to-right) shunt. Blood stays on the "pink" side, so the baby remains pink but the lungs get "wet."
- Eliminate right-sided findings: Most left-to-right shunts (VSD, PDA) primarily overload the left side of the heart first because that is where the blood returns from the lungs.
Take home points
- A PDA allows oxygenated blood to recirculate through the lungs, increasing the workload of the left side of the heart.
- Clinical signs include a continuous murmur, bounding pulses, and widened pulse pressure.
- Small PDAs may be asymptomatic, while large ones lead to heart failure and poor growth.
Exams on Congenital heart diseases: disorders with decreased pulmonary blood flow
Custom Exams
Login to Create a Quiz
Click here to loginLessons
 Naxlex
Just Now
Naxlex
Just Now
Notes Highlighting is available once you sign in. Login Here.
Objectives
- Classify congenital heart diseases characterized by increased pulmonary blood flow, specifically focusing on left-to-right shunting mechanisms.
- Describe the anatomical defects of atrial septal defect (ASD), ventricular septal defect (VSD), and patent ductus arteriosus (PDA).
- Identify the clinical manifestations of pulmonary over-circulation, such as tachypnea, poor feeding, and failure to thrive.
- Explain the pathophysiology of Eisenmenger syndrome.
- Interpret diagnostic findings for these defects from diagnostic tests such as chest X-rays and echocardiogram.
- Outline the medical and surgical management options for all these conditions.
- Develop a nursing care plan for pediatric patients with increased pulmonary blood flow.
- Recognize potential complications following surgical repairs of these defects.
Introduction
- Congenital heart defects (CHDs) characterized by increased pulmonary blood flow represent the most prevalent group of cardiac malformations in the pediatric population.
- These disorders are primarily defined by an abnormal communication between the systemic (left) and pulmonary (right) circulations.
- Because systemic pressure significantly exceeds pulmonary pressure, blood is diverted through these openings in a left-to-right shunt. Consequently, oxygenated blood that has already returned from the lungs is recirculated back to the pulmonary vasculature rather than being distributed to the body.
- The pathophysiology of these defects, most notably atrial septal defect (ASD), ventricular septal defect (VSD), and patent ductus arteriosus (PDA), centers on pulmonary over-circulation. This "re-pumping" of blood creates a dual burden: the lungs become congested and "wet," while the heart chambers must compensate for the massive volume overload.
- Over time, if left untreated, this chronic volume stress can lead to right ventricular hypertrophy and permanent remodeling of the pulmonary arteries, potentially resulting in irreversible pulmonary hypertension.
- From a nursing perspective, these patients typically present as "acyanotic" or "pink" infants. Instead of the blue tint seen in obstructive or cyanotic defects, these children exhibit the classic signs of congestive heart failure (CHF). Nurses must be vigilant for subtle cues such as tachypnea, tachycardia, and periorbital edema.
- A hallmark clinical sign is feeding intolerance since infants often become diaphoretic and exhausted during feeds, leading to a "failure to thrive" as their metabolic demands outpace their caloric intake.
- Understanding these defects is essential for the nurse to anticipate complications and provide the necessary education to families preparing for eventual surgical or transcatheter repairs.
Atrial septal defect
Definition
- Atrial septal defect (ASD) is a common congenital heart anomaly characterized by a persistent opening in the interatrial septum, the muscular wall separating the left and right atria. This defect allows for a left-to-right shunting of oxygenated blood after birth, leading to increased pulmonary blood flow and potential right-sided heart volume overload.
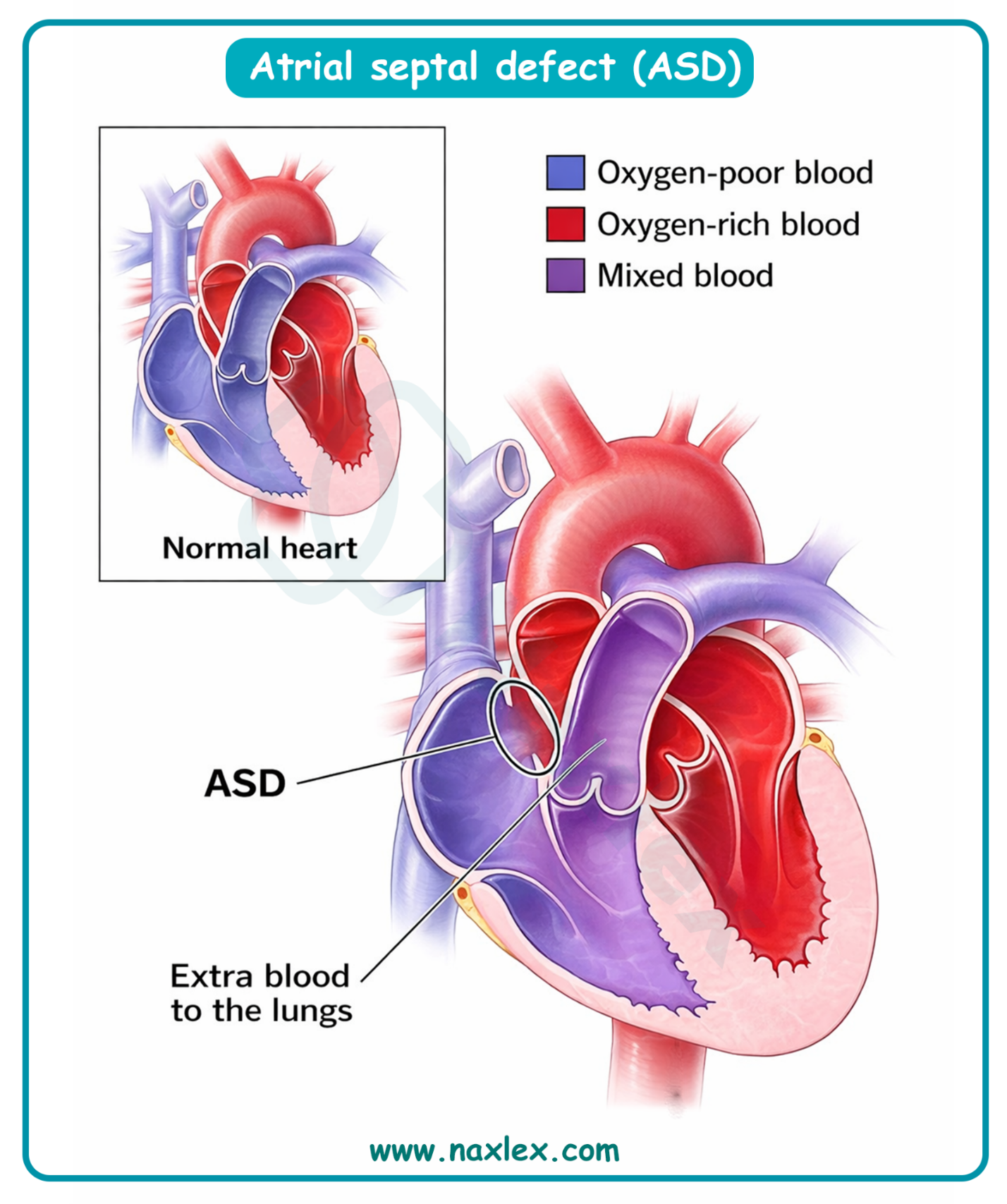
Epidemiology
- ASD accounts for approximately 6% to 10% of all congenital heart defects.
- The incidence is approximately 1 in 1,500 live births.
- There is a significant female-to-male predilection, with a ratio of approximately 2:1.
- Secundum ASD is the most common type, representing 80% of all cases.
Etiology
- Genetic factors: Associated with chromosomal abnormalities such as Trisomy 21 (Down Syndrome), Holt-Oram Syndrome, and mutations in the NKX2.5 gene.
- Environmental factors: Maternal exposure to rubella, alcohol consumption (Fetal Alcohol Syndrome), tobacco use, or certain medications (e.g., SSRIs) during the first trimester.
- Embryological failure: Incomplete formation of the septum primum or septum secundum, or failure of the foramen ovale to close postnatally.
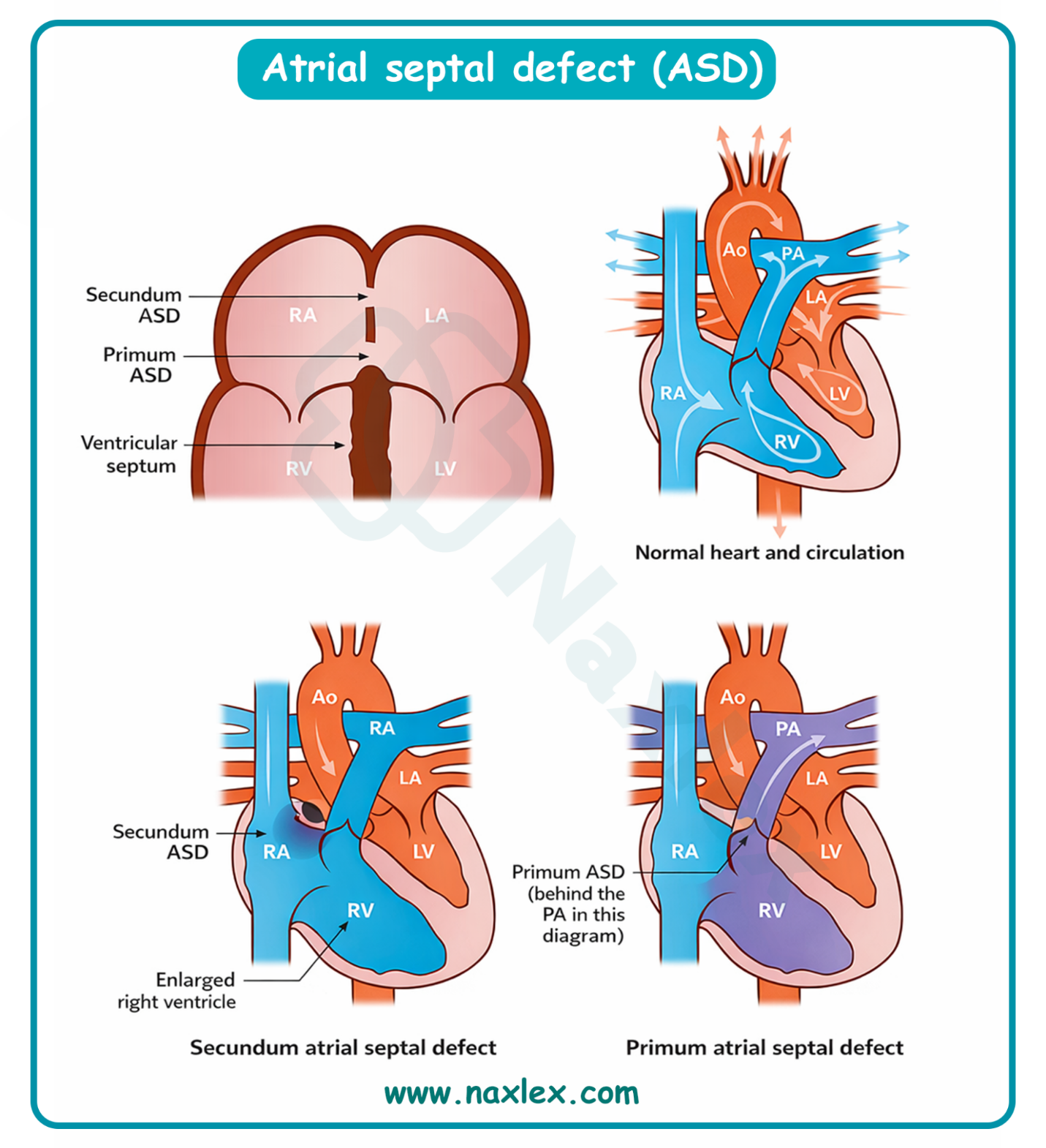
Pathophysiology
- Pressure gradient: Systemic left atrial pressure is normally higher than right atrial pressure.
- Left-to-right shunt: Blood flows from the left atrium (high pressure) to the right atrium (low pressure) through the defect.
- Volume overload: The right atrium and right ventricle must accommodate the extra shunted blood plus the normal systemic venous return.
- Pulmonary hypercirculation: Increased volume is ejected into the pulmonary artery, leading to increased pulmonary blood flow.
- Remodeling: Over time, chronic volume overload causes right atrial and ventricular dilation. If untreated, this can progress to pulmonary hypertension and, rarely in childhood, Eisenmenger syndrome (reversal of shunt).
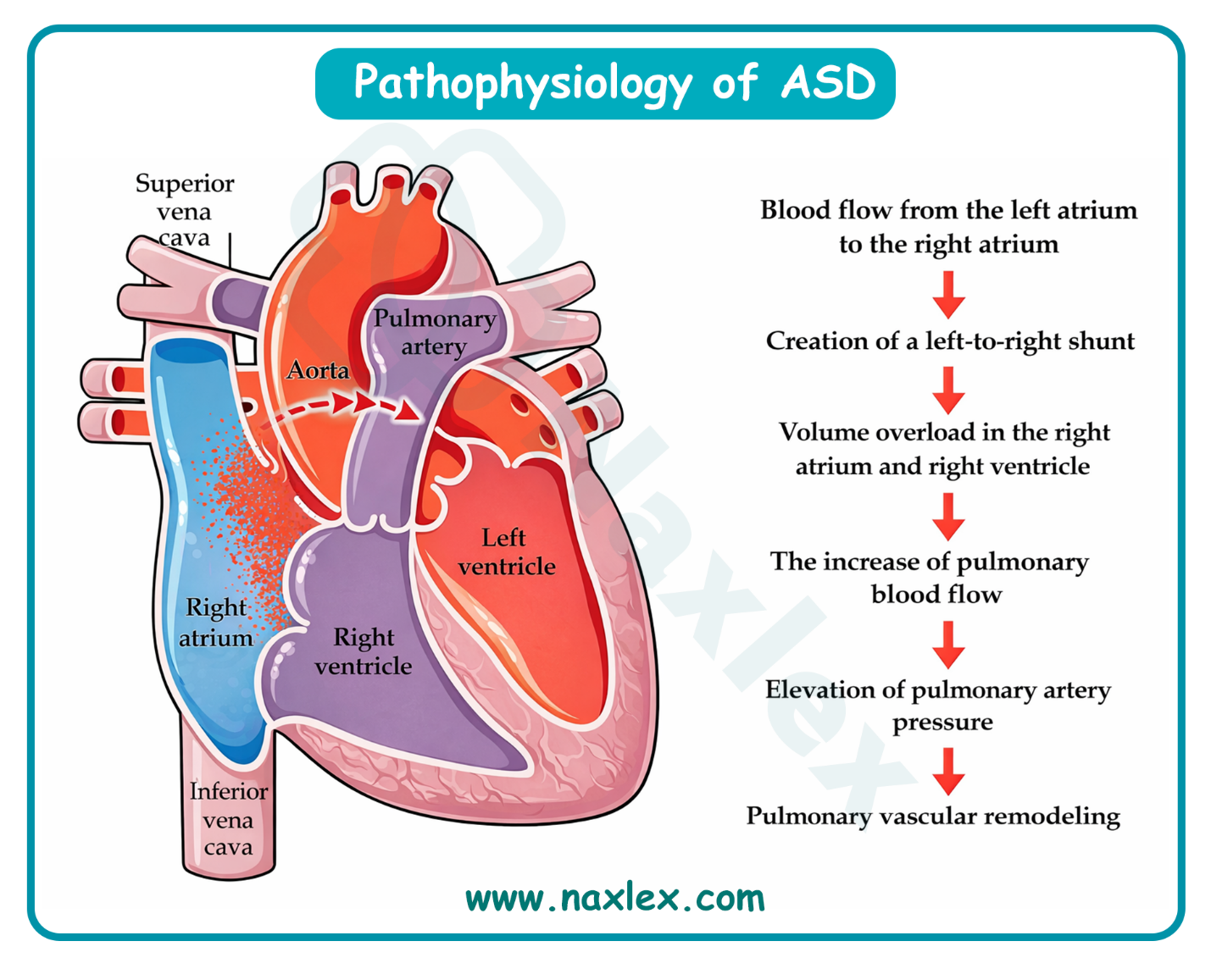
Clinical Manifestations
Many pediatric patients remain asymptomatic for years. Symptoms typically emerge as the shunt size increases or the heart compensates.
- General: Fatigue, dyspnea on exertion, and poor weight gain (failure to thrive) in severe cases.
- Respiratory: Recurrent lower respiratory tract infections due to pulmonary congestion.
- Cardiac auscultation:
- S1: Normal or split.
- S2: Characteristically exhibits a fixed, wide splitting. This occurs because the large stroke volume of the right ventricle delays the closure of the pulmonic valve, and this delay does not vary with respiration.
- Murmur: A midsystolic (ejection) systolic murmur is heard best at the upper left sternal border (2nd intercostal space) due to increased flow across the pulmonic valve (relative pulmonic stenosis).
- Diastolic flow rumble: In large shunts, a mid-diastolic murmur may be heard at the lower left sternal border due to increased flow across the tricuspid valve.
Diagnostic Evaluation
- Echocardiography (ECHO): The gold standard. Transthoracic ECHO identifies the location, size of the defect, direction of the shunt, and evidence of right-sided chamber enlargement.
- Electrocardiogram (ECG): May show right axis deviation, right atrial enlargement, or a primum defect pattern (left axis deviation). Often reveals an incomplete right bundle branch block.
- Chest X-ray (CXR): May show cardiomegaly (right-sided) and prominent pulmonary vascular markings (increased pulmonary blood flow).
- Cardiac catheterization: Rarely needed for diagnosis but used for hemodynamic assessment by measuring pulmonary blood flow to systemic blood flow (Qp:Qs ratio) or transcatheter device closure.
Therapeutic Management
A. General Principles
- Observation: Small defects (< 5 mm) in infants often close spontaneously within the first 12 to 18 months of life.
- Medical management: Diuretics (e.g., furosemide) and digoxin may be used if signs of congestive heart failure (CHF) are present.
- Nonsurgical closure: Transcatheter device closure (e.g., Amplatzer Septal Occluder) is the preferred treatment for Secundum ASDs with sufficient tissue margins.
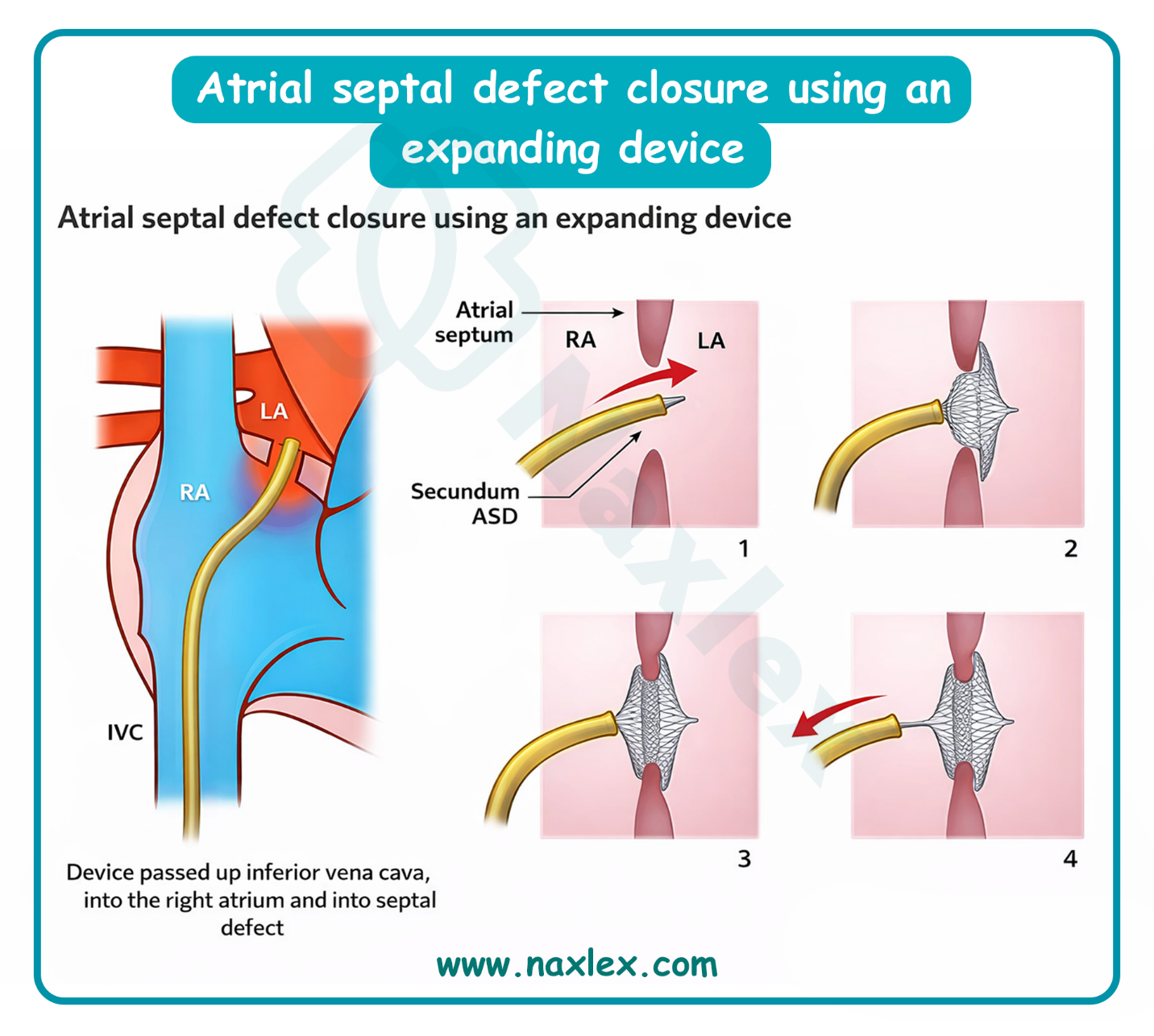
B. Surgical Management
- Indication: Large defects with significant shunting (Qp:Qs > 1.5:1) or failures of device closure.
- Procedure: Open-heart surgery using cardiopulmonary bypass. The defect is closed via primary purse string suture (small defects) or a pericardial/Dacron patch (large defects).
Post-Operative Nursing Interventions
Post-Op Day 0 to 1 (ICU Phase):
- Maintain mechanical ventilation and monitor arterial blood gases (ABGs).
- Monitor hourly chest tube output; report drainage > 3 mL/kg/hr for 3 consecutive hours or 5 mL/kg in 1 hour (risk of hemorrhage).
- Continuous ECG monitoring for arrhythmias (Junctional ectopic tachycardia or A-V blocks).
- Strict I&O monitoring; maintain foley catheter patency.
- Assess peripheral perfusion (capillary refill < 3 seconds).
Post-Op Day 2 to 3 (Transition Phase):
- Encourage weaning from supplemental oxygen.
- Advance diet from NPO to clear liquids as bowel sounds return.
- Incentive spirometry or bubble blowing to prevent atelectasis.
- Pain management using scheduled IV ketorolac or acetaminophen with opioid rescue.
Post-Op Day 4 to Discharge:
- Monitor surgical incision for signs of infection (erythema, drainage).
- Gradual increase in ambulation.
- Parent education on activity restrictions (no lifting by arms for 4 to 6 weeks to protect sternum).
Nursing Diagnosis (Post-Op)
Post-Op Day 1:
- Decreased Cardiac Output related to surgical trauma, arrhythmias, or fluid volume shifts.
- Impaired Gas Exchange related to anesthesia, pain, or pulmonary congestion.
- Risk for Fluid Volume Deficit related to blood loss or diuretic therapy.
Post-Op Day 2 to 3:
- Acute Pain related to sternotomy and chest tube irritation.
- Ineffective Airway Clearance related to retained secretions and reluctance to cough.
- Risk for Infection related to invasive lines and surgical incision.
Post-Op Day 4+:
- Deficient Knowledge (Caregiver) related to home care, medication administration, and follow-up.
- Activity Intolerance related to surgical recovery and generalized weakness.
Complications
- Cardiac: Atrial arrhythmias (Atrial fibrillation/flutter), Congestive Heart Failure, and Sick Sinus Syndrome.
- Pulmonary: Pulmonary Vascular Obstructive Disease (Pulmonary Hypertension).
- Surgical: Postpericardiotomy syndrome (fever, pericardial friction rub, pleurisy), endocarditis, or paradoxical emboli (stroke).
Prognosis
- The prognosis is excellent following successful repair, with most children living a normal life span.
- Surgical mortality is < 1% in uncomplicated cases.
- If repaired early (before age 25), the risk of late-onset arrhythmias and pulmonary hypertension is significantly reduced.
Ventricular septal defect
Definition
- Ventricular septal defect (VSD) is a congenital cardiac anomaly characterized by an abnormal opening in the interventricular septum, which serves as the partition between the right and left ventricles. This defect results in a communication between the high-pressure left ventricle and the low-pressure right ventricle, typically leading to a left-to-right shunt.
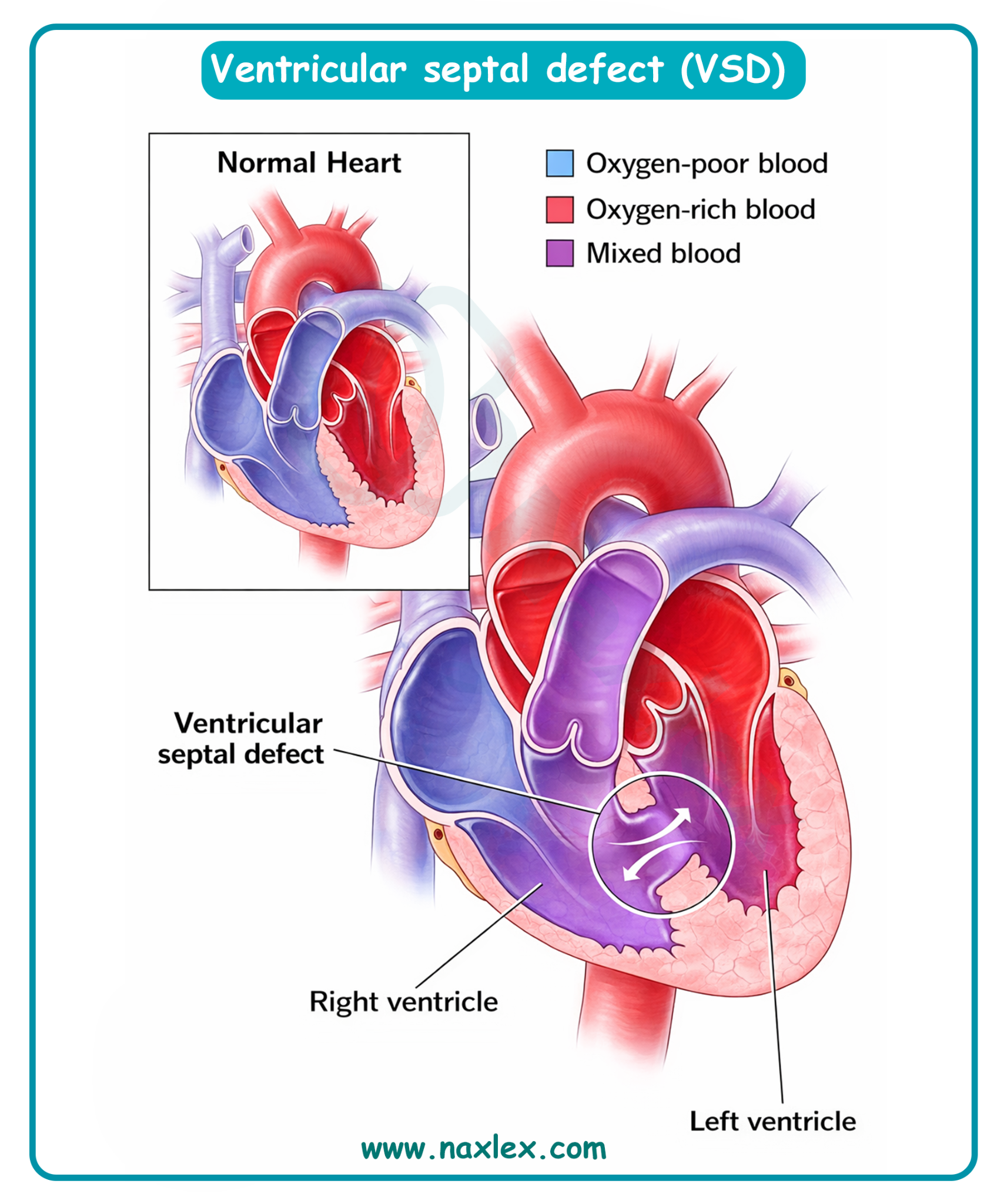
Epidemiology
- VSD is the most common congenital heart defect, accounting for approximately 25% to 35% of all congenital cardiac malformations.
- Incidence is roughly 2 to 7 per 1,000 live births.
- There is no significant gender predilection.
- Approximately 30% to 50% of small VSDs close spontaneously during the first year of life.
Etiology
- Genetic factors: Highly associated with chromosomal trisomies, specifically Trisomy 21 (Down Syndrome), Trisomy 13 (Patau Syndrome), and Trisomy 18 (Edwards Syndrome).
- Teratogenic exposure: Maternal intake of antiepileptic drugs, alcohol (Fetal Alcohol Syndrome), or maternal diabetes.
- Embryological failure: Failure of the muscular or membranous portion of the ventricular septum to fuse with the endocardial cushions during the 4th to 8th week of gestation.
Types of VSD by Location
The VSD is classified based on where it sits in the ventricular septum.
|
Type |
Frequency |
Location & Characteristics |
Nursing Considerations |
|
Membranous (Perimembranous) |
~70-80% |
Upper section of the septum, near the heart valves (aortic and tricuspid). |
Most common. Most likely to require surgery if large. Closely monitor for conduction issues (Bundle of His is nearby). |
|
Muscular (Trabecular) |
~5-20% |
Lower, muscular part of the septum. Can have multiple holes ("Swiss cheese" septum). |
High rate of spontaneous closure. If small, often asymptomatic. |
|
Inlet (AV Canal type) |
~5% |
Posterior septum, where blood enters the ventricles (near tricuspid/mitral valves). |
Often associated with Down Syndrome. Rarely closes on its own. |
|
Outlet (Supracristal) |
~5% |
Superior/anterior septum, near the pulmonary and aortic valves. |
Most common in Asian populations. Risk of aortic valve prolapse due to lack of support. |
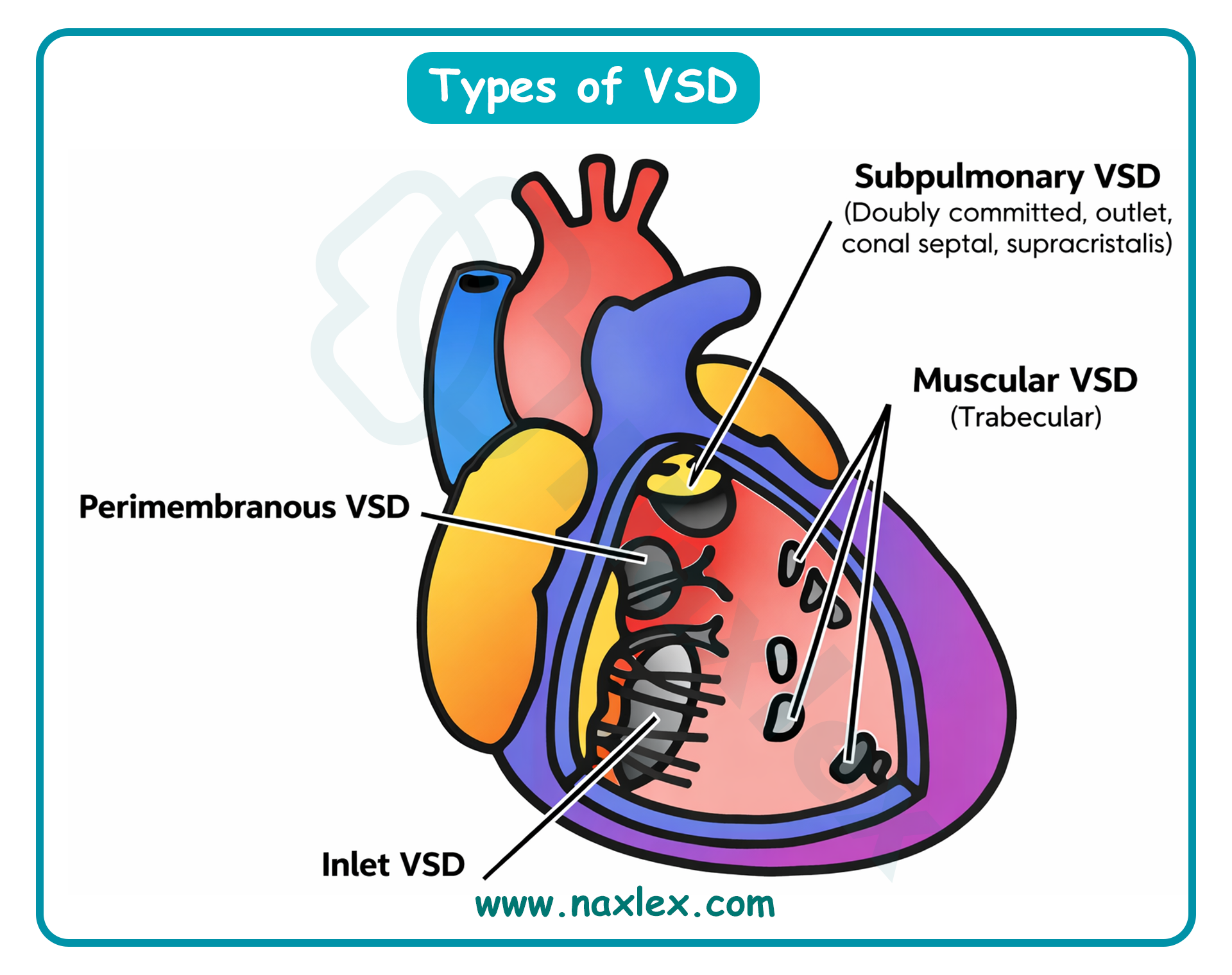
Pathophysiology
- Shunt dynamics: Post-birth, systemic vascular resistance exceeds pulmonary vascular resistance. Blood follows the path of least resistance from the left ventricle (LV) through the defect into the right ventricle (RV).
- Pulmonary overcirculation: The extra volume from the LV is pumped into the pulmonary artery, leading to increased pulmonary blood flow.
- Volume overload: Increased venous return from the lungs enters the left atrium and left ventricle, causing left-sided chamber dilation and hypertrophy.
- Right ventricular pressure: If the VSD is large (restrictive vs. non-restrictive), the RV and pulmonary arteries are exposed to systemic pressures, eventually leading to right ventricular hypertrophy and pulmonary vascular obstructive disease.
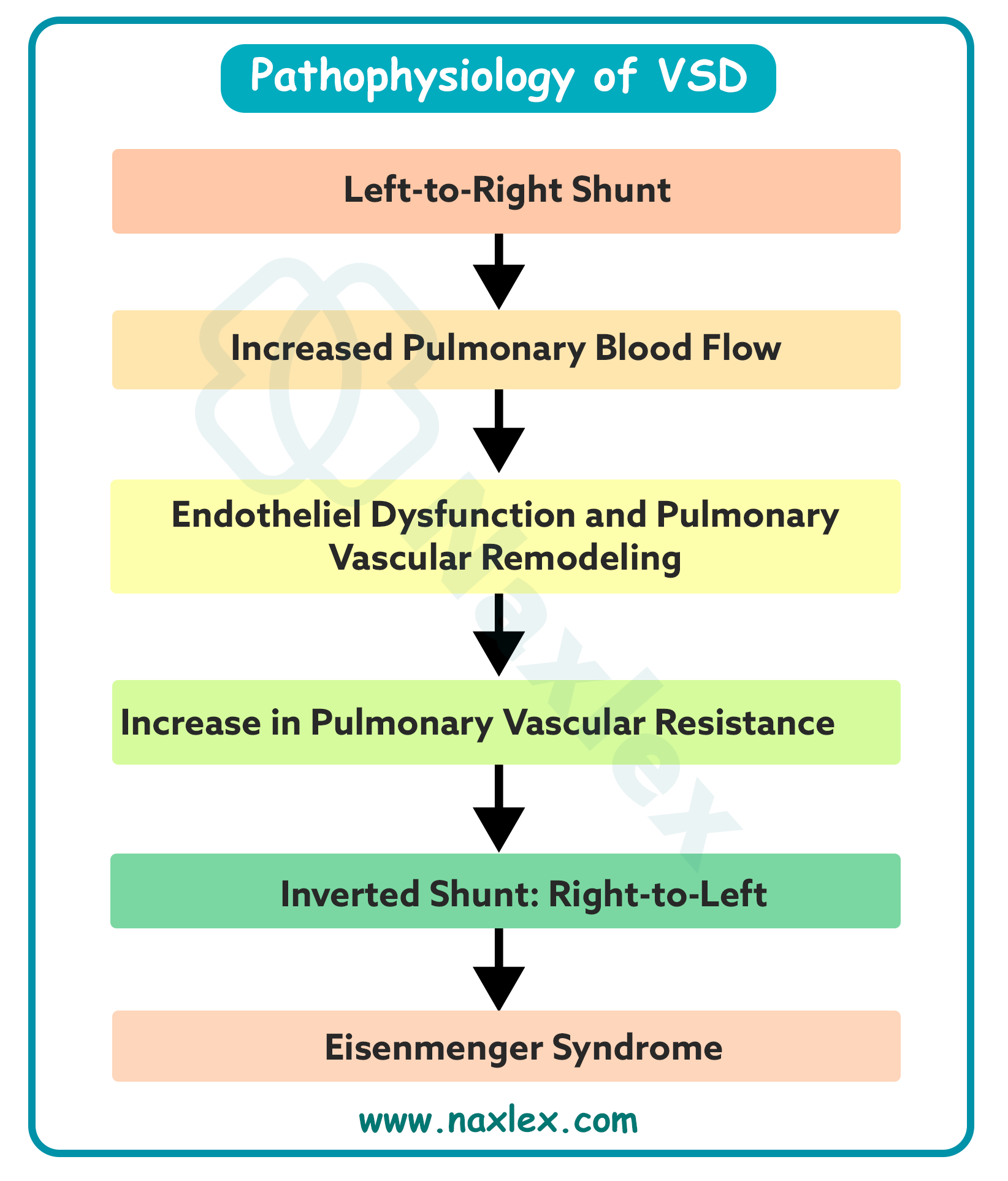
Clinical Manifestations
Symptoms depend on the size of the defect and the degree of pulmonary vascular resistance.
- Small VSD: Often asymptomatic; discovered during routine physical exams.
- Large VSD: Signs of Congestive Heart Failure (CHF), including tachypnea, dyspnea, poor feeding, diaphoresis during feeding, and failure to thrive.
- Cardiac auscultation features:
- Murmur: A loud, harsh, holosystolic (pansystolic) murmur heard best at the left lower sternal border (LLSB).
- Palpation: A systolic thrill may be palpable at the LLSB in restrictive defects.
- S2: If pulmonary hypertension develops, the pulmonic component of the second heart sound (P2) becomes accentuated or loud.
- Mitral flow rumble: A mid-diastolic rumble at the apex may be present in large shunts due to increased flow across the mitral valve.
Diagnostic Evaluation
- Echocardiogram (ECHO): Definitive tool. Two-dimensional ECHO with Color Flow Doppler identifies the location (perimembranous, muscular, supracristal), size, and shunt magnitude.
- Electrocardiogram (ECG): May show left ventricular hypertrophy (LVH) in moderate defects or Biventricular Hypertrophy (BVH) in large defects.
- Chest X-ray (CXR): Reveals cardiomegaly (prominent LV and LA) and increased pulmonary vascular markings (plethora).
- Cardiac catheterization: Performed to assess pulmonary artery pressure and vascular resistance if Eisenmenger syndrome is suspected.
Therapeutic Management
A. General Principles
Management aims to control symptoms of CHF, maintain adequate growth, and prevent pulmonary vascular permanent damage.
B. Medical Management
- Diuretics: Furosemide (Lasix) is used to reduce pulmonary congestion and preload. Monitor for hypokalemia and dehydration.
- Afterload reducers: ACE inhibitors (e.g., Captopril or Enalapril) reduce systemic resistance, making it easier for the LV to pump blood systemically rather than through the shunt.
- Inotropic support (Digoxin): Mechanism: Increases myocardial contractility (positive inotrope) and slows heart rate (negative chronotrope).
- Monitoring: Nursing must check the apical pulse for 1 full minute prior to administration. Withhold if pulse is < 90 to 110 beats/min in infants or < 70 beats/min in older children.
- Toxicity: Monitor for bradycardia, dysrhythmias, and vomiting (earliest sign of toxicity in infants). Monitor serum potassium levels, as hypokalemia potentiates digoxin toxicity.
C. Surgical Management
- Palliative: Pulmonary Artery Banding (PA Banding) to reduce pulmonary blood flow. Rarely performed now except in complex multiple defects.
- Corrective: Complete repair using a Dacron or pericardial patch sutured over the defect via cardiopulmonary bypass.
- Transcatheter closure: Using a septal occluder device for specific muscular VSDs.
Post-Operative Nursing Interventions
Day 0 to Day 1 (Critical Phase):
- Hemodynamic Monitoring: Constant monitoring of CVP, MAP, and LAP.
- Inotropic Support (Dopamine): Mechanism: A precursor to norepinephrine. At low/moderate doses (2 to 10 mcg/kg/min), it stimulates beta-1 receptors to increase cardiac output and myocardial contractility. At higher doses (> 10 mcg/kg/min), it stimulates alpha receptors causing vasoconstriction.
- Nursing: Monitor infusion site for extravasation (can cause tissue necrosis) and monitor for tachycardia.
- Ventilation: Maintain mechanical ventilation; monitor ET tube patency and ABGs.
- Chest Tube: Monitor drainage. Notify surgeon if drainage is > 3 mL/kg/hr for 3 hours or > 5 mL/kg/hr in a single hour.
Day 2 to Day 3 (Intermediate Phase):
- Extubation: Assess for readiness to wean; provide humidified oxygen post-extubation.
- Fluid Balance: Strict I&O; monitor for signs of Postpericardiotomy Syndrome (fever, pericardial rub).
- Pain Management: Transition from continuous IV infusions to intermittent IV or oral analgesics.
Day 4 to Discharge (Recovery Phase):
- Incision Care: Monitor sternal incision for dehiscence or infection.
- Nutrition: Gradual reintroduction of high-calorie feedings (e.g., 24 to 30 kcal/oz formula) to support catch-up growth.
Nursing Diagnosis (Post-Op)
Day 1 to 2:
- Decreased Cardiac Output related to myocardial depression, rhythm disturbances, or fluid shifts.
- Impaired Gas Exchange related to ventilation/perfusion mismatch or atelectasis.
- Risk for Electrolyte Imbalance related to diuretic use and bypass-induced shifts.
Day 3 to Discharge:
- Acute Pain related to surgical incision and chest tube placement.
- Risk for Infection related to invasive lines and sternotomy.
- Deficient Knowledge (Parental) related to home medication administration and activity restrictions.
Complications
- Arrhythmias: Specifically Right Bundle Branch Block (RBBB) or complete heart block if the patch interferes with the Bundle of His.
- Residual VSD: Small leaks around the patch.
- Eisenmenger syndrome: Definition: A condition where chronic left-to-right shunting causes irreversible pulmonary vascular obstructive disease.
- Pathophysiology: High pulmonary blood flow leads to pulmonary artery wall thickening and fibrosis. Pulmonary resistance eventually exceeds systemic resistance.
- Result: The shunt reverses to right-to-left, resulting in deoxygenated blood entering systemic circulation.
- Clinical Signs: Cyanosis, clubbing of digits, and polycythemia. Once this occurs, surgical repair of the VSD is contraindicated.
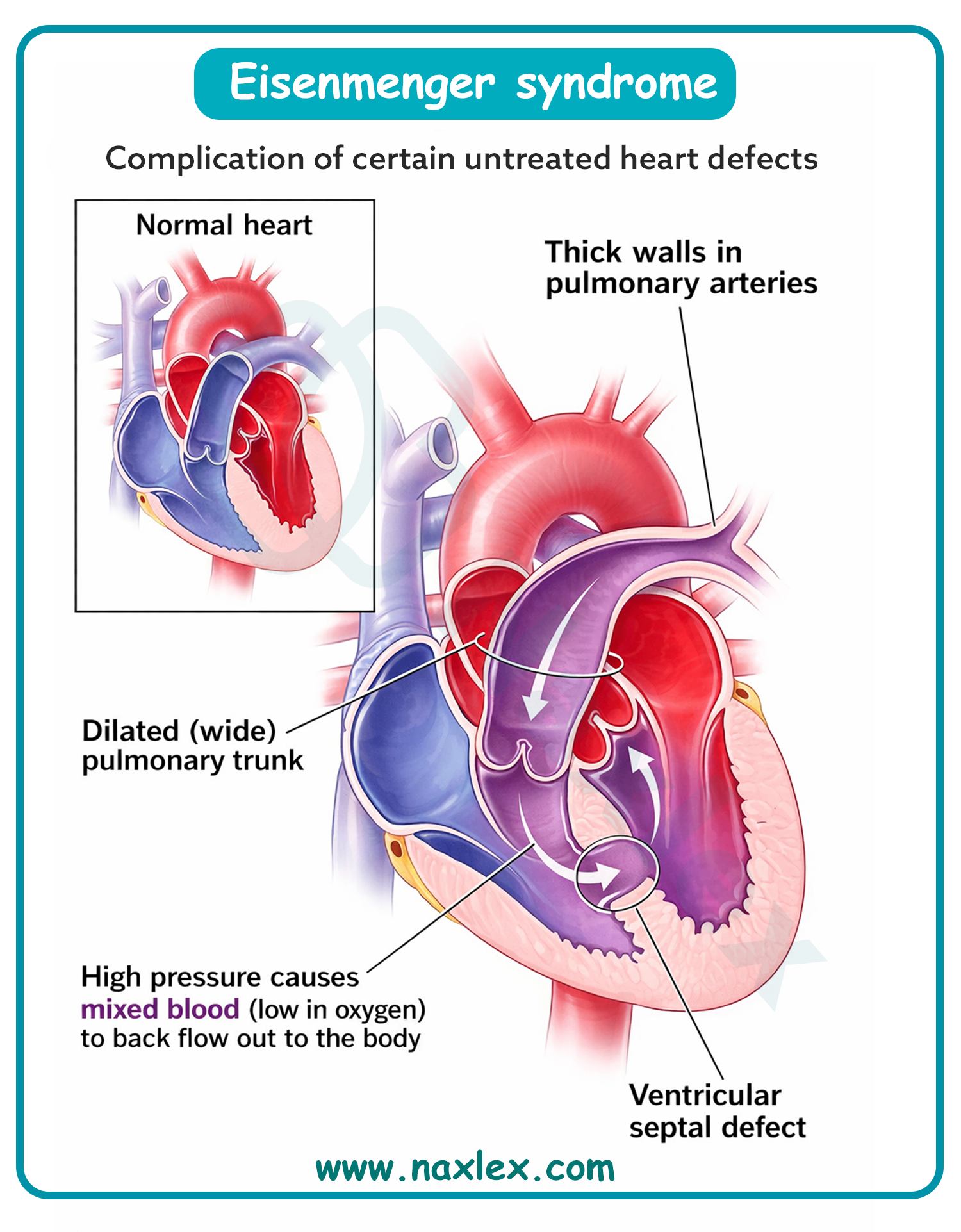
Prognosis
- Small VSD: Excellent; many close spontaneously with no impact on life expectancy.
- Repaired VSD: Excellent. Most children lead normal, active lives.
- Unrepaired Large VSD: Poor; leads to CHF, growth failure, and death from pulmonary hypertension or Eisenmenger syndrome by the 3rd or 4th decade of life.
Atrioventricular canal defect
Definition
- Atrioventricular Canal Defect (AVCD), also known as Atrioventricular Septal Defect (AVSD) or Endocardial Cushion Defect, is a complex congenital heart malformation involving an opening in the atrial septum (primum ASD), an opening in the ventricular septum (inlet VSD), and abnormalities of the mitral and tricuspid valves. In the complete form, there is a single, common atrioventricular valve instead of two separate valves.
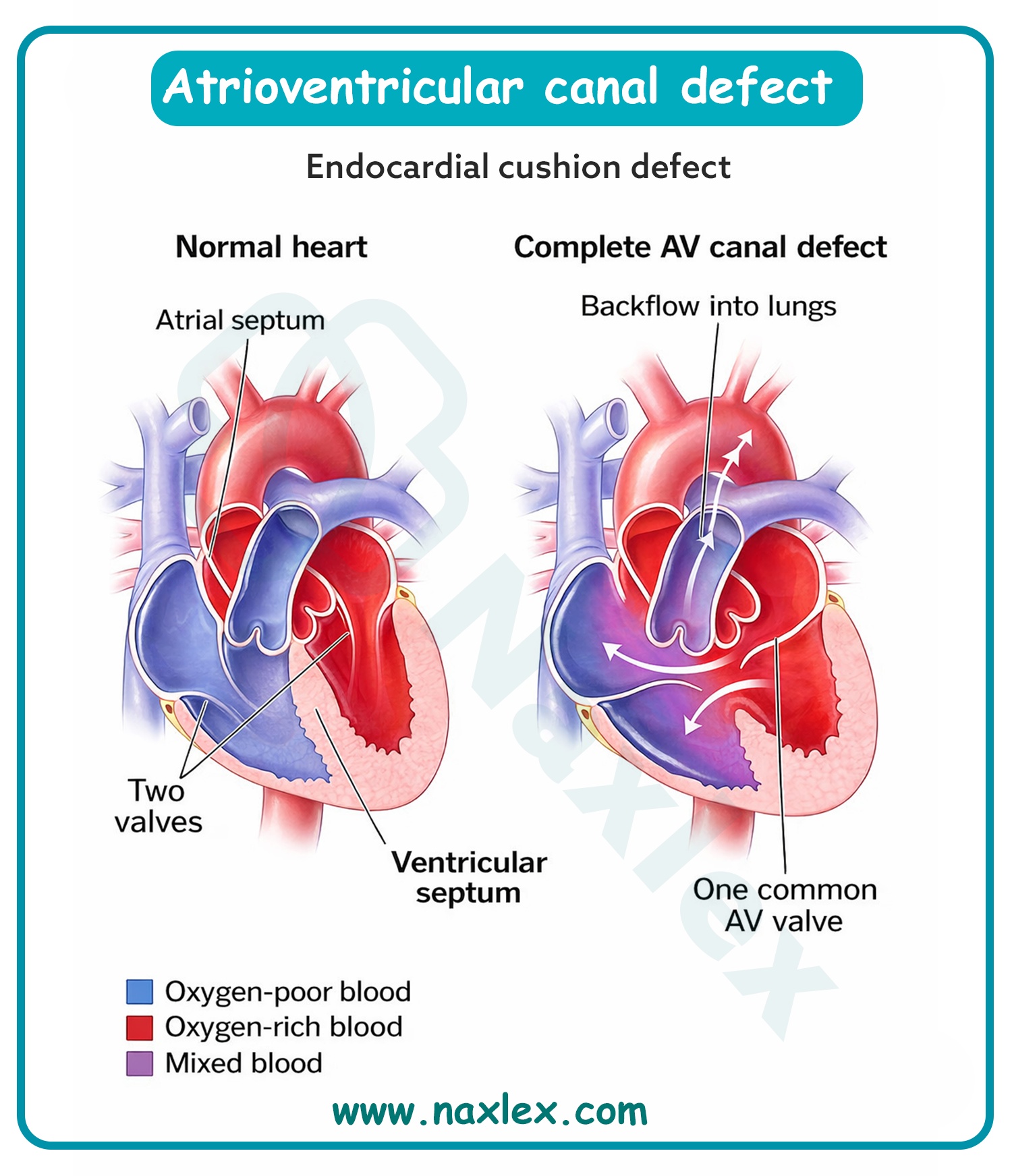
Epidemiology
- AVCD accounts for approximately 4% to 5% of all congenital heart defects.
- It is highly associated with Down Syndrome (Trisomy 21); approximately 45% of children with Down Syndrome have a congenital heart defect, and of those, nearly 40% have AVCD.
- Incidence is roughly 0.35 per 1,000 live births.
Etiology
- Genetic Factors: Strongest correlation is with Trisomy 21. Also associated with Ellis-van Creveld syndrome and Heterotaxy syndromes.
- Embryology: Result of the failure of the endocardial cushions to fuse during the 4th to 7th week of gestation. These cushions are responsible for forming the lower portion of the atrial septum, the upper portion of the ventricular septum, and the mitral and tricuspid valves.
Pathophysiology
- Shunting: Due to openings in both the atrial and ventricular septa, there is massive left-to-right shunting of blood.
- Valve Regurgitation: The common atrioventricular valve is often incompetent, leading to significant regurgitation of blood back into the atria during ventricular systole.
- Volume and Pressure Overload: The right ventricle and pulmonary circulation are subjected to both high pressure (from the VSD) and high volume (from the ASD and valve regurgitation).
- Pulmonary Vascular Disease: Rapid development of pulmonary hypertension occurs due to the excessive blood flow and high pressure delivered to the lungs.
- Heart Failure: The combination of volume overload and pressure overload leads to early-onset congestive heart failure (CHF).
Clinical Manifestations
Signs usually appear within the first 4 to 8 weeks of life as pulmonary vascular resistance drops.
- General: Tachypnea, poor feeding, diaphoresis during exertion (feeding), and failure to thrive.
- Respiratory: Frequent respiratory infections and persistent wheezing.
- Cyanosis: May be mild or absent initially, but can increase with crying or during episodes of heart failure.
- Cardiac Auscultation Features:
- Precordial Activity: Hyperactive precordium or a palpable thrill at the left lower sternal border.
- S2: Loud and narrowly split due to pulmonary hypertension.
- Murmurs: A harsh, holosystolic murmur heard best at the left lower sternal border (VSD component).
- An apical holosystolic murmur radiating to the axilla (mitral regurgitation component).
- A mid-diastolic rumble at the apex or LLSB due to increased flow across the common AV valve.
Diagnostic Evaluation
- Antenatal Echocardiography: Often diagnosed in utero between 18 and 22 weeks gestation, particularly in pregnancies screened for Trisomy 21.
- Echocardiogram (Postnatal): Primary diagnostic tool. It defines the size of the ASD and VSD, the anatomy of the common AV valve, and the degree of valvular regurgitation.
- Electrocardiogram (ECG): Classically shows a "superior" QRS axis (Left Axis Deviation between -30 and -150 degrees). Also reveals right ventricular hypertrophy (RVH), left ventricular hypertrophy (LVH), and first-degree AV block.
- Chest X-Ray (CXR): Shows significant cardiomegaly and increased pulmonary vascular markings (plethora).
Therapeutic Management
A. General Principles
The primary goal is to manage CHF symptoms and maintain growth until the infant is stable enough for surgical repair, which is typically performed between 3 and 6 months of age to prevent permanent pulmonary vascular damage.
B. Medical Management
- Diuretics: Furosemide (Lasix) to reduce pulmonary edema and systemic volume.
- Afterload Reducers: ACE inhibitors (Enalapril or Captopril) to decrease systemic vascular resistance, thereby reducing the left-to-right shunt and mitral regurgitation.
- Nutrition: High-calorie formula (24 to 30 kcal/oz) or nasogastric tube feedings to meet the high metabolic demands of the failing heart.
- Digoxin: Used to improve myocardial contractility and control heart rate.
C. Surgical Management
- Complete Repair: This is the standard procedure. It involves:
- Closing the ASD with a patch.
- Closing the VSD with a patch.
- Dividing the common AV valve into two separate functional valves (mitral and tricuspid) and repairing the "cleft" in the mitral valve.
- Palliative (Pulmonary Artery Banding): Rarely performed today; reserved for very small or unstable infants to restrict pulmonary blood flow until total repair is feasible.
Post-Operative Nursing Interventions
Post-Op Day 1 to 2:
- Arrhythmia Monitoring: High risk for heart block (due to sutures near the AV node). Maintain external pacemaker at the bedside.
- Hemodynamic Stability: Titrate vasoactive drips (Dopamine, Milrinone) to maintain cardiac output and reduce afterload.
- Pulmonary Hypertension Hypertensive Crisis: Maintain sedation and paralysis as needed; avoid hypoxia and acidosis which can trigger pulmonary artery spasms.
- Fluid Management: Strict I&O; monitor chest tube output for hemorrhage (notify if > 3 to 5 mL/kg/hr).
Post-Op Day 3 to 4:
- Weaning: Gradual weaning from mechanical ventilation. Monitor work of breathing and ABGs.
- Chest Tube Removal: Assist with removal once drainage decreases. Monitor for subsequent pleural effusion.
- Pacing: Continue to monitor for delayed AV block; assess the need for temporary pacing wires.
Post-Op Day 5 to Discharge:
- Nutrition: Transition from IV fluids to oral feedings. Monitor for fatigue during feeding.
- Infection Control: Monitor sternal incision and invasive line sites for erythema or purulent drainage.
- Parental Education: Instruct on sternal precautions (no lifting under the arms) and the need for lifelong endocarditis prophylaxis.
Nursing Diagnosis (Post-Op)
Post-Op Day 1 to 2:
- Decreased Cardiac Output related to surgical trauma, cardiopulmonary bypass effects, or heart block.
- Risk for Ineffective Cerebral/Systemic Tissue Perfusion related to low cardiac output or thromboembolism.
- Impaired Gas Exchange related to pulmonary hypertension or atelectasis.
Post-Op Day 3 to 5:
- Acute Pain related to sternotomy and chest tubes.
- Excess Fluid Volume related to the inflammatory response to bypass and renal compensation.
- Risk for Infection related to surgical incision and multiple invasive catheters.
Post-Op Day 6 to Discharge:
- Caregiver Role Strain related to the complexity of the child's care and future follow-up requirements.
- Imbalanced Nutrition: Less than Body Requirements related to high metabolic rate and post-op recovery.
Complications
- Heart Block: Permanent damage to the conduction system requiring a permanent pacemaker.
- Residual Valve Regurgitation: Specifically mitral valve regurgitation, which may require later valve replacement or re-repair.
- Subaortic Stenosis: Narrowing below the aortic valve due to the "gooseneck" deformity common in AVCD.
- Pulmonary Vascular Obstructive Disease: If surgery is delayed, permanent pulmonary hypertension (Eisenmenger syndrome) can occur.
Prognosis
- Without surgery, most infants with complete AVCD will die from CHF or pneumonia before age 2 or 3.
- Post-surgical survival rate is over 90%.
- Long-term survival is excellent, though 10% to 15% of patients may require a second operation within 10 years for mitral valve repair or replacement.
Patent ductus arteriosus
- Patent ductus arteriosus (PDA) is a common congenital heart defect where the ductus arteriosus, a fetal vascular connection between the main pulmonary artery and the descending aorta, fails to physiologically close after birth. In utero, this vessel shunts blood away from the non-functional lungs. Postnatally, its persistence results in a left-to-right shunt.
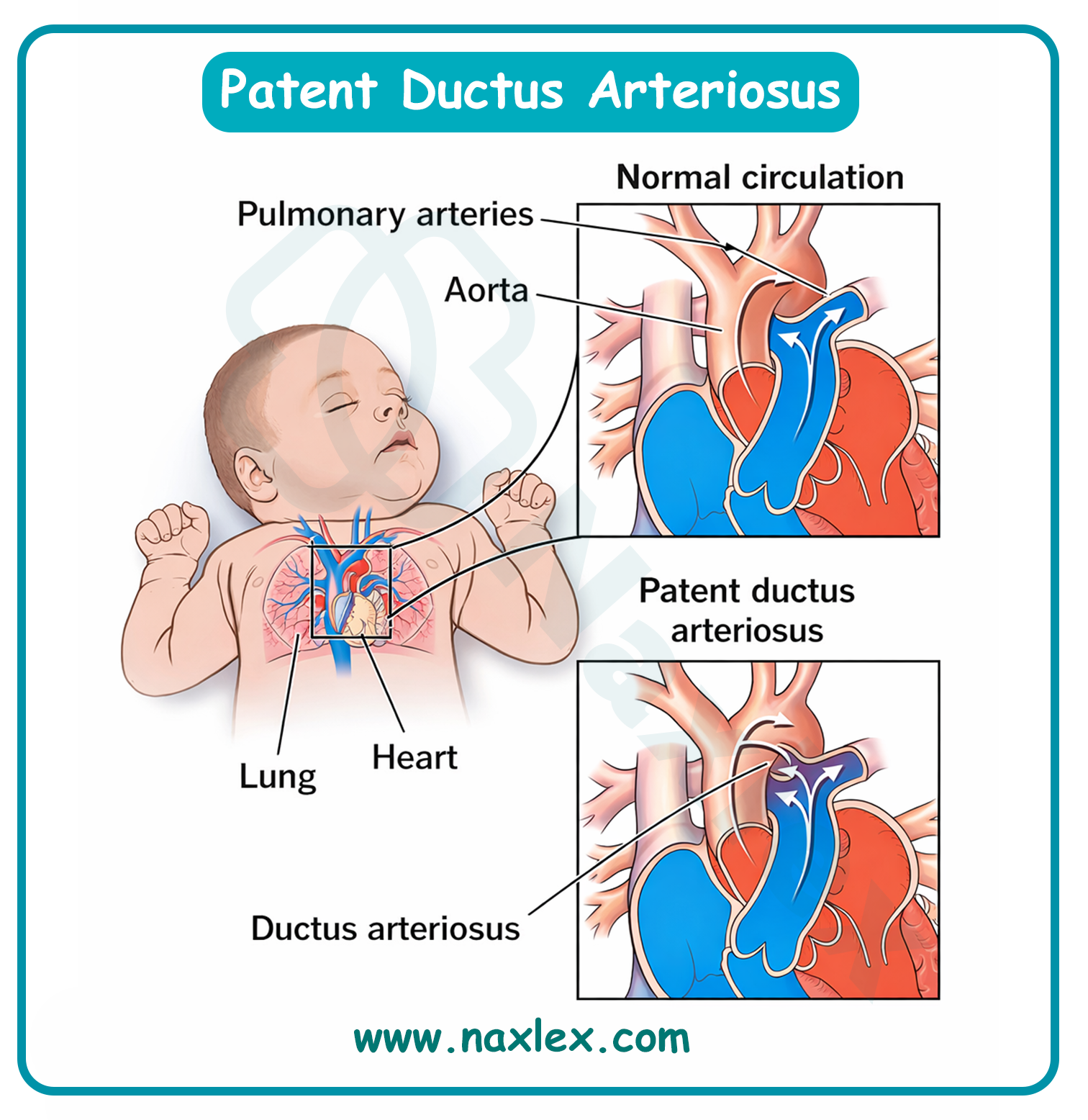
Epidemiology
- PDA accounts for approximately 5% to 10% of all congenital heart defects in full-term infants.
- There is a much higher incidence in preterm infants, occurring in up to 60% of those born at < 28 weeks gestation.
- It is twice as common in females as in males.
- Frequently associated with high-altitude births and maternal rubella infection.
Etiology
- Prematurity: The most significant risk factor. The immature ductus is less sensitive to oxygen and has higher circulating prostaglandin levels.
- Prostaglandin Levels: High levels of circulating prostaglandin E2 (PGE2) keep the ductus patent.
- Hypoxia: Low arterial oxygen tension (PaO2) prevents the muscular constriction required for closure.
- Genetic Factors: Associated with Trisomy 21 and Carpenter syndrome.
Pathophysiology
- Shunt Direction: After birth, systemic vascular resistance rises and pulmonary vascular resistance falls. Blood shunts from the high-pressure aorta into the low-pressure pulmonary artery.
- Pulmonary Overcirculation: The lungs receive extra blood flow, leading to pulmonary congestion and increased pulmonary venous return to the left atrium (LA).
- Left-Sided Overload: The LA and left ventricle (LV) must pump the normal cardiac output plus the shunted blood, leading to LA and LV dilation and hypertrophy.
- Steal Phenomenon: In large PDAs, blood is "stolen" from the systemic circulation during diastole, resulting in low diastolic blood pressure and decreased perfusion to distal organs (e.g., gut and kidneys).
Clinical Manifestations
- General: May be asymptomatic if small. Large shunts present with tachypnea, poor feeding, bounding peripheral pulses (e.g., palmar pulses), and widened pulse pressure.
- Cardiac Auscultation Features:
- Murmur: A classic, continuous, "machinery-like" murmur heard throughout systole and diastole.
- Location: Best heard at the left infraclavicular area or the upper left sternal border.
- S2: May be obscured by the loud machinery murmur.
- Mid-diastolic Rumble: Heard at the apex if the shunt is large, indicating increased flow across the mitral valve.
Diagnostic Evaluation
- Echocardiogram (ECHO): Gold standard. Confirms the presence of the ductus, measures its diameter, determines the direction of the shunt, and assesses for left-sided chamber enlargement.
- Chest X-Ray (CXR): In large PDAs, shows cardiomegaly (prominent LV/LA) and increased pulmonary vascular markings.
- Electrocardiogram (ECG): Often normal in small PDAs; may show Left Ventricular Hypertrophy (LVH) or biventricular hypertrophy in large shunts.
- Arterial Blood Gas (ABG): May show respiratory acidosis or hypoxemia if pulmonary edema is severe.
Therapeutic Management
A. General Principles
Management focuses on facilitating ductal closure while maintaining systemic perfusion and oxygenation, particularly in premature neonates.
B. Medical Management
- Fluid Restriction: Minimal to moderate fluid restriction to reduce pulmonary congestion.
- Diuretics: Furosemide to manage symptoms of congestive heart failure.
- Prostaglandin Inhibitors:
- Indomethacin: A non-selective COX inhibitor used primarily in preterm infants to trigger ductal constriction.
- Ibuprofen (IV): Often preferred over Indomethacin due to a lower risk of renal side effects and necrotizing enterocolitis (NEC).
- Monitoring: Nursing must monitor urine output (minimum 1 mL/kg/hr), serum creatinine, and platelet counts during therapy.
C. Surgical/Interventional Management
- Transcatheter Occlusion: Deployment of a coil or occluder device via cardiac catheterization. This is the treatment of choice for older infants and children.
- Surgical Ligation: Performed via a left posterolateral thoracotomy. The ductus is tied off (ligated) or clipped. This is usually reserved for preterm infants who fail medical therapy.
Post-Operative Nursing Interventions
Post-Op Day 1:
- Respiratory Care: Monitor mechanical ventilation or CPAP settings. Provide aggressive suctioning and monitor breath sounds for atelectasis.
- Blood Pressure Monitoring: Watch for "Post-Ligation Hypertension," a common transient rebound effect.
- Fluid Balance: Monitor I&O strictly. Check for peripheral edema.
- Neurological Check: Monitor for recurrent laryngeal nerve damage (check for hoarse cry or stridor).
Post-Op Day 2 to 3:
- Pain Management: Administer scheduled analgesics (e.g., Acetaminophen or IV opioids). Monitor for pain-related tachycardia.
- Wound Care: Assess thoracotomy incision for drainage, redness, or crepitus.
- Nutrition: Slowly reintroduce oral or gavage feedings as tolerated.
Nursing Diagnosis (Post-Op)
Post-Op Day 1:
- Ineffective Breathing Pattern related to surgical pain and thoracotomy.
- Decreased Cardiac Output related to changes in hemodynamics following ductal closure.
- Impaired Gas Exchange related to pulmonary edema or atelectasis.
Post-Op Day 2 to 3:
- Acute Pain related to surgical incision and chest tube (if present).
- Risk for Infection related to invasive procedures and surgical site.
- Interrupted Family Processes related to the stress of a child's hospitalization.
Complications
- Congestive Heart Failure (CHF): Due to chronic volume overload.
- Infective Endocarditis: The turbulent flow at the ductus increases the risk of bacterial colonization.
- Pulmonary Hypertension: Irreversible damage to pulmonary vasculature (Eisenmenger syndrome) if left untreated for years.
- Necrotizing Enterocolitis (NEC): Specifically in preterm infants due to "diastolic steal" reducing mesenteric perfusion.
Prognosis
- Treated PDA: Excellent. Most children have a normal life expectancy and no activity restrictions after closure.
- Untreated PDA: Risk of heart failure, bacterial endocarditis, and pulmonary hypertension increases with age.
- Surgical Success: Mortality for surgical ligation or transcatheter closure is < 1%.
Summary
- Congenital heart defects that increase pulmonary blood flow are primarily defined by structural openings that allow oxygenated blood to recirculate through the lungs.
- Under normal physiological conditions, the left side of the heart operates at a higher pressure than the right.
- When a defect exists, such as an atrial septal defect (ASD), ventricular septal defect (VSD), or patent ductus arteriosus (PDA), it creates a left-to-right shunt. This means blood is diverted from the systemic circulation back into the pulmonary system, leading to an over-circulation of the lungs and a volume overload on the heart.
- The primary consequence of this increased flow is pulmonary congestion. The lungs become flooded with excess fluid, which significantly increases the work of breathing and interferes with gas exchange.
- If these defects are left uncorrected, the chronic high-pressure flow can cause permanent damage to the pulmonary vasculature. This may eventually lead to Eisenmenger Syndrome, where pulmonary resistance becomes so high that the shunt reverses (right-to-left), resulting in systemic cyanosis.
- Clinically, these infants present with signs of congestive heart failure rather than the "blue" tint associated with cyanotic defects. Key nursing observations include tachypnea, tachycardia, and a characteristic "machine-like" or holosystolic murmur depending on the defect. Because the heart is working at an accelerated rate, these infants often demonstrate poor weight gain and exhaustion during feedings.
- Nursing management is centered on optimizing cardiac output and reducing pulmonary congestion. This involves administering diuretics to manage fluid volume, providing high-calorie nutrition to support growth, and monitoring for respiratory infections.
- Treatment varies from medical management with Indomethacin (for PDA closure) to surgical interventions like patching or transcatheter device closure, all aimed at restoring normal hemodynamic balance.
Naxlex
Videos
Login to View Video
Click here to loginTake Notes on Congenital heart diseases: disorders with decreased pulmonary blood flow
This filled cannot be empty
Join Naxlex Nursing for nursing questions & guides! Sign Up Now

