
Please set your exam date
Antepartum Diagnostic Testing
Study Questions
Practice Exercise 1
Which of the following statements accurately describes a key difference between prenatal screening tests and prenatal diagnostic tests?
Explanation
Prenatal testing is used to detect chromosomal and structural abnormalities in the fetus. Screening involves non-invasive methods like ultrasound and maternal serum markers that estimate risk but are not diagnostic. Diagnostic tests such as amniocentesis and chorionic villus sampling directly analyze fetal cells and DNA, confirming or excluding disease. Normal nuchal translucency is <3.5 mm, while maternal serum alpha-fetoprotein is normally 0.5–2.5 multiples of the median.
Rationale for correct answers
3. Screening tests assess the risk of fetal conditions, while diagnostic tests provide confirmation. Screening uses maternal serum and ultrasound, estimating probability. Diagnostic methods obtain fetal cells via invasive sampling, confirming chromosomal abnormalities with high sensitivity and specificity.
Rationale for incorrect answers
1. Screening tests are non-invasive and carry no direct miscarriage risk. Diagnostic tests, such as amniocentesis, are invasive and increase miscarriage risk by approximately 0.1–0.3%. Therefore, the statement reverses the actual risk profile.
2. Screening tests do not provide definitive diagnosis but only probability estimates. A positive screen result must be confirmed with a diagnostic test such as CVS or amniocentesis. Diagnostic tests provide direct chromosomal or genetic information, not just risk assessment.
4. Screening tests are performed earlier, typically in the first trimester at 11–14 weeks or second trimester at 15–20 weeks. Diagnostic tests, including CVS at 10–13 weeks and amniocentesis at 15–20 weeks, occur later or selectively. Hence, screening usually precedes diagnostic evaluation.
Take home points
- Screening tests estimate risk, diagnostic tests confirm disease.
- Screening is non-invasive, diagnostic is invasive and carries miscarriage risk.
- Screening occurs earlier, diagnostic tests are done after abnormal screen results.
- Screening requires follow-up if abnormal, diagnostic provides definitive confirmation.
A client expresses concern about the ethical implications of prenatal testing. The nurse’s response should be guided by which of the following ethical principles?
Explanation
Autonomy is the ethical principle that emphasizes the patient’s right to make informed choices regarding medical care without coercion. It requires full disclosure of information, including risks, benefits, and alternatives, enabling patients to make decisions based on their values. Normal decision-making capacity requires intact cognition, orientation, and comprehension.
Rationale for correct answers
3. Autonomy ensures that patients retain control over healthcare decisions, including whether to undergo prenatal testing. Nurses must provide unbiased, clear, and accurate information, respecting the patient’s choices even if they differ from provider preferences.
Rationale for incorrect answers
1. Paternalism removes decision-making power from the patient, placing it solely with the healthcare provider. This contradicts current ethical standards, as modern practice prioritizes patient-centered care and informed decision-making.
2. Beneficence involves acting in the patient’s best interests but does not override autonomy. Defining the “best” outcome from the provider’s perspective risks imposing values that may conflict with the patient’s own beliefs or cultural views.
4. Non-maleficence focuses on avoiding harm but only addresses physical outcomes. While important, it does not address ethical concerns about choice, consent, and respect for patient values, which are central in prenatal testing discussions.
Take home points
- Autonomy is the key principle guiding prenatal testing decisions.
- Paternalism conflicts with patient-centered ethical practice.
- Beneficence must balance with autonomy, not override it.
- Non-maleficence alone does not encompass the ethical complexity of prenatal testing.
The following maternal conditions may necessitate antepartum diagnostic testing to assess fetal well-being except:
Explanation
Antepartum fetal surveillance evaluates fetal well-being in pregnancies with increased risk of placental insufficiency, fetal hypoxia, growth restriction, or stillbirth. Testing includes non-stress tests, biophysical profiles, and Doppler velocimetry. Indications include hypertensive disorders, diabetes, thyroid dysfunction, and anemia. Normal fetal heart rate baseline is 110–160 bpm. Amniotic fluid index should be ≥5 cm. Umbilical artery systolic/diastolic ratio should be <3.0 in term gestation.
Rationale for correct answers
3. Mild asthma without hypoxia or exacerbations does not typically impair placental function or oxygen delivery to the fetus. Well-controlled asthma with normal maternal oxygen saturation (>95%) does not necessitate routine antepartum testing. No evidence of increased risk for fetal compromise in mild cases.
Rationale for incorrect answers
1. Gestational diabetes increases risk for macrosomia, polyhydramnios, and stillbirth due to altered glucose metabolism and placental dysfunction. Antepartum testing is indicated especially if insulin or oral hypoglycemics are used, or if poor glycemic control is present. Normal fasting glucose should be <95 mg/dL, 1-hour postprandial <140 mg/dL.
2. Iron-deficiency anemia with hemoglobin <11 g/dL in the first and third trimesters or <10.5 g/dL in the second trimester can impair oxygen delivery to the fetus. Severe anemia (<8 g/dL) is associated with fetal growth restriction and increased perinatal morbidity, warranting surveillance.
4. Hypothyroidism, especially if untreated, can lead to fetal neurodevelopmental delay, low birth weight, and placental insufficiency. TSH levels should be <2.5 mIU/L in the first trimester and <3.0 mIU/L in the second and third trimesters. Elevated TSH or low free T4 requires monitoring and fetal assessment.
Take home points
- Mild asthma without hypoxia does not require antepartum fetal testing.
- Gestational diabetes increases risk for fetal compromise and warrants surveillance.
- Iron-deficiency anemia can impair fetal oxygenation and growth.
- Hypothyroidism affects placental function and fetal development.
Which of the following are indications for recommending antepartum diagnostic testing? Select all that apply
Explanation
Antepartum diagnostic testing is recommended when there is increased risk of fetal chromosomal, structural, or genetic abnormalities. Indications include advanced maternal age (≥35 years), abnormal maternal serum or ultrasound results, and family history of inherited disorders such as cystic fibrosis. Diagnostic tests such as amniocentesis and chorionic villus sampling directly analyze fetal karyotype or DNA. Normal amniotic fluid volume is 5–25 cm by amniotic fluid index; abnormal values warrant closer evaluation.
Rationale for correct answers
1. Advanced maternal age (≥35 years) significantly increases the risk of chromosomal abnormalities such as trisomy 21. This is a standard indication for diagnostic testing such as amniocentesis.
3. A family history of cystic fibrosis, an autosomal recessive genetic disorder, is an indication for prenatal diagnostic testing to assess carrier status or confirm fetal genetic status.
4. Abnormal maternal serum screening results, such as elevated alpha-fetoprotein or abnormal quad screen values, require confirmatory diagnostic testing to determine the presence of chromosomal or structural abnormalities.
Rationale for incorrect answers
2. Normal first-trimester ultrasound findings are reassuring and do not warrant further diagnostic testing. Diagnostic testing is only indicated if abnormalities are detected or other risk factors are present.
5. Maternal history of seasonal allergies is not associated with fetal genetic or chromosomal abnormalities and does not indicate the need for antepartum diagnostic testing.
Take home points
- Antepartum diagnostic testing is indicated for advanced maternal age.
- Family history of genetic disorders such as cystic fibrosis requires confirmatory fetal testing.
- Abnormal serum screening results warrant diagnostic testing.
- Normal findings and benign maternal conditions do not require invasive testing.
Which of the following ethical principles guide nursing care in antepartum diagnostic testing? Select all that apply
Explanation
Ethical principles guide nursing care during antepartum diagnostic testing by ensuring respect, safety, and balanced decision-making. Autonomy ensures the woman’s right to make informed, voluntary choices. Beneficence requires acting in the best interest of both mother and fetus, promoting positive outcomes. Non-maleficence obligates avoidance of unnecessary harm, particularly with invasive procedures such as amniocentesis. Normal miscarriage risk from amniocentesis is approximately 0.1–0.3%, underscoring the importance of weighing risks against benefits.
Rationale for correct answers
1. Autonomy guides care by supporting the patient’s right to make independent decisions after receiving full, unbiased information.
2. Beneficence directs nurses to promote maternal-fetal well-being, balancing medical benefits against potential risks.
4. Non-maleficence ensures avoidance of unnecessary harm, requiring careful consideration of procedural risks before recommending testing.
Rationale for incorrect answers
3. Paternalism involves providers making decisions for the patient, which contradicts ethical standards of respecting autonomy and informed consent.
5. Utilitarianism focuses on outcomes that benefit the majority, not the individual patient. Nursing ethics prioritize patient-centered care, not population-based utility.
Take home points
- Autonomy ensures patient freedom in prenatal testing decisions.
- Beneficence balances interventions toward maternal and fetal well-being.
- Non-maleficence prevents unnecessary harm in invasive procedures.
- Paternalism and utilitarianism conflict with patient-centered ethical nursing practice.
Practice Exercise 2
Which prenatal screening test measures the fluid accumulation at the back of the fetal neck?
Explanation
Nuchal translucency ultrasound is a screening test performed in the first trimester to measure fluid-filled space at the fetal neck. Increased thickness (>3.5 mm at 11–14 weeks) is associated with chromosomal abnormalities such as trisomy 21, 18, and 13, as well as structural defects like congenital heart disease. Normal nuchal translucency is <3.5 mm. This non-invasive test, when combined with maternal serum markers, improves early detection of fetal anomalies.
Rationale for correct answers
2. Nuchal translucency ultrasound specifically measures the subcutaneous fluid accumulation at the back of the fetal neck between 11–14 weeks gestation. Increased measurements are linked to chromosomal and structural abnormalities.
Rationale for incorrect answers
1. The quadruple screen is a maternal serum screening performed between 15–20 weeks measuring AFP, hCG, estriol, and inhibin A. It does not involve ultrasound or direct fetal measurements.
3. Amniocentesis is a diagnostic test done at 15–20 weeks where amniotic fluid is sampled to analyze fetal karyotype, not neck fluid.
4. Chorionic villus sampling is performed at 10–13 weeks to obtain placental tissue for genetic analysis. It provides a definitive diagnosis but does not measure nuchal translucency.
Take home points
- Nuchal translucency ultrasound measures fluid at the fetal neck.
- Increased thickness (>3.5 mm) indicates higher risk of chromosomal and structural defects.
- Quad screen and serum markers assess biochemical risk but not neck fluid.
- Amniocentesis and CVS are diagnostic, not screening via ultrasound measurement.
A nurse is educating a patient about maternal serum screening. Which protein is measured in this test?
Explanation
Alpha-fetoprotein (AFP) is a glycoprotein produced by the fetal liver, yolk sac, and gastrointestinal tract. It normally enters maternal circulation and is measurable in maternal serum between 15–20 weeks. Normal maternal serum AFP values are 0.5–2.5 multiples of the median (MoM). Abnormally elevated AFP may indicate open neural tube defects or abdominal wall defects, while low AFP may indicate trisomy 21 or 18. Maternal serum screening combines AFP with other analytes for more accurate risk assessment.
Rationale for correct answers
1. Alpha-fetoprotein is the protein measured in maternal serum screening. Its levels provide important information about risk for neural tube defects and chromosomal abnormalities.
Rationale for incorrect answers
2. Pregnancy-associated plasma protein-A (PAPP-A) is used in first-trimester screening, not the standard second-trimester maternal serum screen. Low levels may indicate increased risk of trisomy 21.
3. Estriol (unconjugated estriol, uE3) is a hormone included in the quadruple screen but is not classified as a protein. It reflects placental and fetal adrenal function.
4. Inhibin-A is a glycoprotein hormone also included in the quadruple screen, but it is not the main protein measured to detect neural tube defects.
Take home points
- AFP is the primary protein measured in maternal serum screening.
- Elevated AFP suggests neural tube or abdominal wall defects.
- Low AFP is associated with trisomy 21 and 18.
- Other analytes like estriol and inhibin-A are hormones, not proteins.
The nurse is explaining the components of the first-trimester combined screening to a client. Which two maternal serum markers are typically included in this screening?
Explanation
First-trimester combined screening evaluates risk for chromosomal abnormalities such as trisomy 21 and 18 between 11–14 weeks. It integrates nuchal translucency ultrasound with two maternal serum markers: pregnancy-associated plasma protein-A (PAPP-A) and free β-human chorionic gonadotropin (free β-hCG). Low PAPP-A (<0.5 MoM) and high free β-hCG (>2.0 MoM) are associated with trisomy 21, while both low PAPP-A and low β-hCG suggest trisomy 18. Normal PAPP-A and β-hCG levels fall within 0.5–2.0 multiples of the median (MoM).
Rationale for correct answers
2. PAPP-A and free β-hCG are the maternal serum markers included in first-trimester combined screening. When combined with nuchal translucency measurement, detection rates for trisomy 21 increase significantly.
Rationale for incorrect answers
1. Alpha-fetoprotein (AFP) and unconjugated estriol (uE3) are measured later, in second-trimester maternal serum screening (triple or quadruple screen), not in the first trimester.
3. Inhibin-A and AFP are both components of the quadruple screen done in the second trimester, not part of first-trimester combined screening.
4. Unconjugated estriol and inhibin-A are also part of second-trimester screening panels and not used in the first-trimester combined screening.
Take home points
- First-trimester combined screening includes PAPP-A and free β-hCG.
- Low PAPP-A and high free β-hCG suggest trisomy 21.
- Both low PAPP-A and β-hCG suggest trisomy 18.
- AFP, estriol, and inhibin-A are measured in second-trimester screening.
Which of the following are components of the quadruple screen? Select all that apply
Explanation
Quadruple screen is a second-trimester maternal serum screening test performed between 15–20 weeks to estimate risk of fetal chromosomal and structural abnormalities. It measures alpha-fetoprotein (AFP), human chorionic gonadotropin (hCG), unconjugated estriol (uE3), and inhibin-A. Normal AFP levels are 0.5–2.5 MoM, hCG is normally within 0.5–2.0 MoM, estriol reflects placental-fetal steroid production, and inhibin-A is elevated in trisomy 21. Abnormal combinations of these markers indicate increased risk of trisomy 21, trisomy 18, or neural tube defects.
Rationale for correct answers
1. Alpha-fetoprotein is included; elevated levels suggest neural tube or abdominal wall defects, while low levels suggest trisomy 21 or 18.
2. Human chorionic gonadotropin is measured; elevated levels are associated with trisomy 21, while low levels may indicate trisomy 18.
3. Estriol (unconjugated) is included; low levels suggest trisomy 21 and 18 due to impaired placental and fetal steroid metabolism.
5. Inhibin-A is part of the quadruple screen; elevated levels are linked to trisomy 21.
Rationale for incorrect answers
4. Pregnancy-associated plasma protein-A (PAPP-A) is not part of the quadruple screen. It is measured in first-trimester combined screening along with free β-hCG.
Take home points
- Quadruple screen includes AFP, hCG, estriol, and inhibin-A.
- Elevated AFP suggests neural tube defects; low AFP suggests trisomy 21 or 18.
- High hCG and inhibin-A with low AFP and estriol suggest trisomy 21.
- PAPP-A belongs to first-trimester combined screening, not quad screen.
Which of the following are considered advantages of Non-Invasive Prenatal Testing (NIPT) over traditional serum screening methods? Select all that apply
Explanation
Non-invasive prenatal testing (NIPT) analyzes cell-free fetal DNA fragments in maternal blood, usually available from 10 weeks gestation. It has higher detection rates and lower false-positive rates for trisomy 21, 18, and 13 compared to traditional serum screening. It is non-invasive, requiring only a maternal blood sample, thus avoiding procedure-related miscarriage risk. NIPT can also screen for sex chromosome aneuploidies such as Turner syndrome (45,X) or Klinefelter syndrome (47,XXY). However, it is still a screening test, not diagnostic, and abnormal results require confirmation with invasive testing.
Rationale for correct answers
1. NIPT offers higher sensitivity and specificity (>99% for trisomy 21) compared to serum screening, which has lower detection rates.
2. NIPT can be performed as early as 10 weeks gestation, whereas some traditional serum screenings begin at 11–14 weeks or later.
3. NIPT is non-invasive, requiring only maternal venipuncture, eliminating risks associated with procedures like amniocentesis.
5. NIPT can screen for sex chromosome abnormalities, including monosomy X and extra sex chromosomes, which traditional serum screening cannot detect.
Rationale for incorrect answer
4. NIPT does not provide definitive diagnostic results. It is highly accurate but remains a screening test. Positive results must be confirmed with diagnostic methods such as CVS or amniocentesis.
Take home points
- NIPT has higher accuracy than traditional serum screening.
- It can be done earlier (≥10 weeks) and is non-invasive.
- NIPT also screens for sex chromosome abnormalities.
- Abnormal results require confirmatory diagnostic testing.
Practice Exercise 3
Which diagnostic test is performed by sampling placental tissue to analyze fetal DNA?
Explanation
Chorionic villus sampling is a prenatal diagnostic procedure used for early detection of chromosomal abnormalities and genetic disorders. It involves sampling of placental tissue which contains fetal DNA. It is usually performed between 10–13 weeks of gestation. Normal fetal karyotype is 46 chromosomes. Fetal heart rate should remain 110–160 beats/min after the procedure to indicate stability.
Rationale for correct answers
3. Chorionic villus sampling directly obtains placental villi containing fetal DNA, making it the test described in the question. It is indicated for early detection of genetic disorders such as trisomy 21. The stem specifies placental tissue analysis for fetal DNA, which matches this procedure exactly.
Rationale for incorrect answers
1. Amniocentesis involves sampling of amniotic fluid, not placental tissue. It is performed after 15 weeks and is used for chromosomal analysis, neural tube defect detection, and genetic studies. It does not involve chorionic villi and is therefore not the test described.
2. Cordocentesis, also called percutaneous umbilical blood sampling, involves obtaining fetal blood from the umbilical vein under ultrasound guidance, usually after 18 weeks. It is used for rapid karyotyping, detection of fetal anemia, and certain infections. It does not analyze placental tissue, so it does not fit.
4. Nuchal translucency ultrasound measures the fluid-filled space at the back of the fetal neck between 11–14 weeks. Increased thickness is associated with chromosomal abnormalities such as trisomy 21. This method uses ultrasound imaging only, not tissue or DNA sampling, so it is not correct.
Take home points
- Chorionic villus sampling obtains placental tissue for fetal DNA analysis between 10–13 weeks.
- Amniocentesis uses amniotic fluid and is performed later, after 15 weeks.
- Cordocentesis samples umbilical blood after 18 weeks for hematologic and genetic assessment.
- Nuchal translucency ultrasound measures neck fluid, does not involve DNA or tissue analysis.
A nurse is counseling a patient about the risks of amniocentesis. Which complication should the nurse mention?
Explanation
Amniocentesis is an invasive prenatal diagnostic procedure performed after 15 weeks to obtain amniotic fluid for fetal karyotype and biochemical analysis. It can detect chromosomal abnormalities, neural tube defects, and genetic disorders. Normal amniotic fluid volume is 500–1200 mL at term. Fetal heart rate should remain 110–160 beats/min after the procedure. The most significant risk is miscarriage, with an incidence of 0.5–1%.
Rationale for correct answers
2. Risk of miscarriage (0.5–1%) is the primary complication of amniocentesis. The procedure involves puncturing the amniotic sac, which can disrupt membranes or cause infection leading to pregnancy loss. This percentage is widely accepted as the main quantified risk associated with the test.
Rationale for incorrect answers
1. Increased risk of preterm labor is not a typical complication of amniocentesis when performed in the second trimester. Preterm labor is more strongly associated with uterine or cervical conditions and not directly linked to the sampling of fluid.
3. Development of gestational diabetes is unrelated to amniocentesis. Gestational diabetes results from insulin resistance mediated by placental hormones such as human placental lactogen. An invasive diagnostic test does not alter glucose metabolism.
4. Fetal growth restriction is not caused by amniocentesis. Growth restriction results from placental insufficiency, maternal hypertension, smoking, or infection. Amniocentesis does not alter nutrient supply or placental function, so it is not a complication.
Take home points
- Amniocentesis is performed after 15 weeks to analyze amniotic fluid.
- The main risk is miscarriage with an incidence of 0.5–1%.
- Preterm labor, gestational diabetes, and growth restriction are not direct complications.
- Fetal monitoring after the procedure is essential to ensure stability.
The nurse is educating a client about the risks of Chorionic Villus Sampling (CVS). Which of the following is a potential complication specific to CVS compared to amniocentesis, particularly if performed too early?
Explanation
Chorionic villus sampling is a prenatal diagnostic test that involves obtaining placental tissue containing fetal DNA between 10–13 weeks of gestation. It allows early detection of chromosomal abnormalities and genetic disorders. Normal fetal heart rate should remain 110–160 beats/min post-procedure. If performed before 10 weeks, a specific complication is limb reduction defects, likely due to disruption of vascular supply during early limb development.
Rationale for correct answers
2. Limb reduction defects are a complication specific to CVS if performed too early, especially before 10 weeks. Early disruption of embryonic vascular supply may impair proper limb development, leading to hypoplasia or amputation-type defects. This risk is not associated with amniocentesis.
Rationale for incorrect answers
1. Fetal bradycardia can occur transiently with many invasive procedures, including amniocentesis and cordocentesis, but it is not specific to CVS. It generally resolves without lasting consequences and is not the hallmark complication linked to CVS timing.
3. Maternal infection, such as chorioamnionitis, is a rare complication of both CVS and amniocentesis. It results from invasive instrumentation but is not unique to CVS nor tied to gestational timing.
4. Preterm labor is not a typical risk associated with CVS because it is performed in the first trimester. Preterm labor complications occur later in pregnancy, and procedures like amniocentesis or trauma are more relevant contributors.
Take home points
- CVS is performed between 10–13 weeks for early genetic diagnosis.
- If done before 10 weeks, it increases the risk of limb reduction defects.
- Amniocentesis does not carry this specific risk.
- Infection and fluid leakage are possible but nonspecific to CVS.
Which of the following are potential complications of amniocentesis? Select all that apply
Explanation
Amniocentesis is an invasive prenatal procedure performed after 15 weeks to obtain amniotic fluid for chromosomal and biochemical analysis. It can detect aneuploidies, single-gene disorders, and neural tube defects via amniotic fluid alpha-fetoprotein (normal maternal serum AFP at 15–20 weeks: 10–150 ng/mL). Normal amniotic fluid at term measures 500–1200 mL. Complications arise from puncture of membranes and disturbance of the intrauterine environment, the most significant being miscarriage (0.5–1%).
Rationale for correct answers
1. Miscarriage is the most significant complication, occurring in 0.5–1% of cases. It results from rupture of membranes, bleeding, or infection following the procedure.
2. Infection, such as chorioamnionitis, may occur due to introduction of pathogens during needle insertion. It is rare but serious and can threaten both mother and fetus.
4. Cramping is a common minor complication due to uterine irritation from the needle puncture. It usually resolves without intervention but must be monitored.
Rationale for incorrect answers
3. Gestational diabetes is unrelated to amniocentesis. It develops from placental hormone–induced insulin resistance, not from invasive procedures.
5. Maternal hypertension is not a recognized complication of amniocentesis. Hypertensive disorders in pregnancy are linked to placental dysfunction, not diagnostic sampling.
Take home points
- Amniocentesis is performed after 15 weeks for genetic and biochemical testing.
- Main risks: miscarriage (0.5–1%), infection, and cramping.
- Gestational diabetes and maternal hypertension are unrelated conditions.
- Monitoring maternal vitals and fetal heart rate after the procedure is essential.
Which of the following are components of the biophysical profile (BPP)? Select all that apply
Explanation
Biophysical profile is a prenatal assessment tool that evaluates fetal well-being using real-time ultrasound and a non-stress test. It measures acute and chronic markers of hypoxia. Components scored include fetal breathing movements, body/limb movements, tone, amniotic fluid volume, and non-stress test. A normal score is 8–10. Normal amniotic fluid index (AFI) is 8–24 cm; normal single deepest pocket is 2–8 cm.
Rationale for correct answers
1. Fetal breathing movements are assessed by ultrasound; at least 1 episode of rhythmic breathing lasting ≥30 seconds in 30 minutes is normal.
3. Fetal movement is part of the BPP; ≥3 discrete body or limb movements within 30 minutes indicates normal activity.
4. Amniotic fluid volume reflects chronic uteroplacental function. A single vertical pocket ≥2 cm or AFI ≥5 cm is normal.
5. Non-stress test evaluates fetal heart rate accelerations with movement. Normal is ≥2 accelerations of ≥15 bpm lasting ≥15 seconds in 20 minutes.
Rationale for incorrect answer
2. Maternal blood pressure is not part of the BPP. It is assessed separately in maternal monitoring, particularly in preeclampsia evaluation, but it is not a fetal biophysical parameter.
Take home points
- The BPP assesses fetal well-being using 5 parameters: breathing, movement, tone, amniotic fluid, and NST.
- A score of 8–10 is normal; 6 is equivocal; ≤4 suggests fetal compromise.
- Maternal vital signs, including blood pressure, are not included in BPP.
- BPP helps differentiate acute vs chronic fetal hypoxia and guides delivery timing.
Practice Exercise 4
A nurse is educating a patient about the anomaly ultrasound. What is typically assessed during this test?
Explanation
Anomaly ultrasound is a mid-trimester scan performed between 18–22 weeks of gestation to evaluate fetal anatomy and detect structural malformations. It systematically examines fetal brain, heart, kidneys, limbs, and spine. Normal amniotic fluid is essential for fetal development; amniotic fluid index (AFI) normally ranges 8–24 cm, and single deepest pocket ranges 2–8 cm. Abnormalities in volume may indicate oligohydramnios or polyhydramnios.
Rationale for correct answers
2. Amniotic fluid volume is typically assessed during the anomaly ultrasound. Measurement ensures adequate fetal environment and screens for conditions like oligohydramnios (AFI <5 cm) and polyhydramnios (AFI >24 cm). This is part of the standard anomaly survey.
Rationale for incorrect answers
1. Fetal blood type cannot be determined by ultrasound. It requires laboratory analysis using maternal or fetal samples (e.g., cell-free DNA, cord blood, or amniocentesis).
3. Maternal hormone levels are not evaluated during ultrasound. Hormone levels such as hCG, estriol, and inhibin A are assessed through serum screening, not imaging.
4. Placental blood flow is not routinely measured in a standard anomaly scan. It requires Doppler ultrasound, typically reserved for high-risk pregnancies with suspected placental insufficiency.
Take home points
- Anomaly ultrasound at 18–22 weeks evaluates fetal anatomy and growth.
- Amniotic fluid volume is routinely assessed during this scan.
- Fetal blood type and maternal hormones cannot be detected by ultrasound.
- Doppler studies of placental blood flow are specialized, not part of standard anomaly scan.
Which of the following fetal structures is specifically assessed during an anatomy ultrasound for potential neural tube defects?
Explanation
Neural tube defects are congenital malformations resulting from incomplete closure of the neural tube during embryogenesis, usually by day 28 post-conception. Defects include spina bifida, meningocele, and myelomeningocele. The spinal column is specifically assessed during the anatomy ultrasound at 18–22 weeks. Normal findings include intact vertebral arches and overlying skin. Maternal serum alpha-fetoprotein (normal 10–150 ng/mL at 15–20 weeks) is often elevated in open neural tube defects.
Rationale for correct answers
3. The spinal column is specifically evaluated during the anatomy ultrasound to detect neural tube defects. Ultrasound looks for discontinuity in vertebral arches, abnormal curvature, or associated cranial signs such as “lemon” or “banana” signs.
Rationale for incorrect answers
1. The four chambers of the heart are assessed during the anatomy scan but this evaluates cardiac malformations, not neural tube defects.
2. Kidneys and bladder are checked to assess urinary tract development and amniotic fluid production, not neural tube closure.
4. Stomach and bowel are assessed to evaluate gastrointestinal patency and situs, not defects of the neural tube.
Take home points
- Neural tube defects result from failed closure of the neural tube by day 28.
- The spinal column is the key structure assessed for these defects on anatomy scan.
- Other structures (heart, kidneys, bowel) are evaluated but for different systems.
- Maternal serum AFP elevation supports diagnosis of open neural tube defects.
Which specialized ultrasound assessment is used to evaluate the risk of preterm labor?
Explanation
Preterm labor is defined as onset of regular uterine contractions with cervical changes before 37 completed weeks of gestation. Risk prediction relies on assessment of cervical length via transvaginal ultrasound. A normal mid-trimester cervical length is ≥25 mm; shorter measurements significantly increase the risk of preterm birth. Additional biochemical markers include fetal fibronectin, but ultrasound cervical length is the standard structural measure.
Rationale for correct answers
3. Cervical length assessment is the specialized ultrasound used to evaluate risk of preterm labor. A cervical length <25 mm before 24 weeks indicates increased risk of preterm birth. It directly measures structural changes in the cervix that precede labor.
Rationale for incorrect answers
1. Doppler ultrasound evaluates blood flow in uteroplacental and fetal vessels. It is used for conditions like intrauterine growth restriction and preeclampsia, not preterm labor risk.
2. Fetal echocardiography assesses structural and functional abnormalities of the fetal heart. It is indicated when congenital heart disease is suspected, not for prediction of preterm delivery.
4. Amniotic fluid index measures fluid volume to assess chronic placental function and fetal well-being. It helps detect oligohydramnios or polyhydramnios but does not predict preterm labor.
Take home points
- Preterm labor is defined as cervical changes with contractions before 37 weeks.
- Cervical length <25 mm before 24 weeks predicts increased risk of preterm delivery.
- Doppler ultrasound and fetal echocardiography assess other conditions, not preterm birth risk.
- Amniotic fluid index reflects uteroplacental function, not cervical readiness for labor.
Which of the following are assessed during an anomaly ultrasound? Select all that apply
Explanation
Anomaly ultrasound is a detailed mid-trimester scan done at 18–22 weeks to evaluate fetal anatomy, placental position, and intrauterine environment. It checks structural integrity of the fetus, placenta, cord, and fluid volume. Normal amniotic fluid index (AFI) ranges 8–24 cm, with a single deepest pocket of 2–8 cm. The exam screens for congenital malformations, multiple gestations, and growth patterns.
Rationale for correct answers
1. Fetal heart structure is assessed, including the four-chamber view and outflow tracts, to detect congenital cardiac defects.
2. Placental position is evaluated to identify placenta previa, accreta spectrum risk, or abnormal insertion sites.
4. Amniotic fluid volume is measured to detect oligohydramnios (AFI <5 cm) or polyhydramnios (AFI >24 cm), which affect fetal development.
Rationale for incorrect answers
3. Maternal blood pressure is not assessed by ultrasound. It is measured clinically as part of maternal monitoring, especially in preeclampsia screening.
5. Fetal blood type cannot be determined by ultrasound. It requires maternal blood tests for cell-free fetal DNA or invasive testing like amniocentesis.
Take home points
- Anomaly ultrasound evaluates fetal anatomy, placental position, and amniotic fluid volume.
- Normal AFI is 8–24 cm; <5 cm indicates oligohydramnios, >24 cm indicates polyhydramnios.
- Maternal blood pressure is measured clinically, not by ultrasound.
- Fetal blood type requires genetic or laboratory testing, not imaging.
Which of the following are indications for performing a specialized ultrasound assessment? Select all that apply
Explanation
Specialized ultrasound is performed when there is a higher risk of fetal abnormality, placental insufficiency, or atypical growth patterns. It involves detailed imaging such as fetal echocardiography, Doppler velocimetry, or targeted organ scans. Normal umbilical artery Doppler shows end-diastolic flow; absent or reversed flow suggests pathology. Amniotic fluid index is normally 8–24 cm, and abnormalities prompt advanced assessment.
Rationale for correct answers
1. Suspected intrauterine growth restriction is an indication because specialized ultrasound with Doppler studies evaluates uteroplacental blood flow and fetal well-being.
3. Family history of congenital heart defects requires fetal echocardiography, a specialized ultrasound, to detect structural cardiac abnormalities.
5. Abnormal amniotic fluid volume (oligohydramnios <5 cm AFI or polyhydramnios >24 cm AFI) requires detailed ultrasound to identify underlying causes such as renal anomalies or gastrointestinal obstruction.
Rationale for incorrect answers
2. Maternal history of seasonal allergies is not an indication for specialized ultrasound. Allergies do not impact fetal structural development or hemodynamics.
4. Routine first-trimester screening involves nuchal translucency and serum markers. It is standard care, not a specialized ultrasound, unless abnormalities are detected.
Take home points
- Specialized ultrasound is reserved for suspected abnormalities or high-risk pregnancies.
- Indications include suspected IUGR, abnormal amniotic fluid, or family history of congenital anomalies.
- Routine screenings are not specialized ultrasounds.
- Seasonal allergies have no effect on fetal imaging needs.
Practice Exercise 5
What is the primary purpose of carrier screening in prenatal care?
Explanation
Carrier screening is a genetic test performed before or during pregnancy to identify if parents carry mutations associated with inherited disorders. It detects autosomal recessive or X-linked conditions where carriers are asymptomatic. When both parents carry the same pathogenic variant, the risk of an affected offspring is 25%. Normal human karyotype contains 46 chromosomes (46,XX or 46,XY). Carrier screening panels commonly test for disorders such as cystic fibrosis, spinal muscular atrophy, and hemoglobinopathies.
Rationale for correct answers
2. Carrier screening identifies parental genetic mutations associated with specific heritable diseases. If both parents carry the same mutation, there is a 25% chance of passing the disorder to the fetus. This allows informed reproductive decision-making and counseling, which is the primary purpose of carrier screening.
Rationale for incorrect answers
1. Carrier screening does not diagnose fetal chromosomal abnormalities. Fetal chromosomal abnormalities such as trisomy 21, 18, or 13 are detected using diagnostic tests like chorionic villus sampling or amniocentesis, or screening tools such as nuchal translucency and non-invasive prenatal testing.
3. Fetal growth restriction is assessed through ultrasound biometry, such as estimated fetal weight, abdominal circumference, and head circumference, not through carrier screening. Doppler velocimetry and serial growth scans are also used for monitoring growth restriction.
4. Evaluation of amniotic fluid volume is done through ultrasound using the amniotic fluid index (normal 8–24 cm) or the single deepest pocket (normal 2–8 cm). Carrier screening cannot provide any information about amniotic fluid volume.
Take home points
- Carrier screening identifies parental mutations that can cause autosomal recessive or X-linked disorders.
- It does not detect chromosomal abnormalities in the fetus.
- Growth restriction is monitored through ultrasound, not genetic carrier testing.
- Amniotic fluid abnormalities are detected by ultrasound indices, not carrier screening.
A pregnant client is referred for genetic counseling. Which of the following is a primary role of the genetic counselor?
Explanation
Rationale for correct answers
3. The genetic counselor interprets genetic test results and provides non-directive counseling. Their primary role is to explain findings, clarify recurrence risks, and offer available reproductive or management options while respecting patient autonomy.
Rationale for incorrect answers
1. Genetic counselors do not perform invasive procedures such as amniocentesis or chorionic villus sampling. These procedures are carried out by physicians trained in maternal-fetal medicine or specialized obstetric care.
2. Genetic counselors do not provide definitive diagnoses. They explain test results and probabilities, but definitive diagnosis comes from laboratory confirmation or physician evaluation of genetic and phenotypic data.
4. Genetic counselors do not recommend specific reproductive choices. They maintain a non-directive approach, ensuring clients understand risks and options but allowing families to make independent decisions.
Take home points
- Genetic counseling is focused on interpretation of results and risk communication.
- Counselors provide non-directive support, not prescriptive advice.
- Invasive diagnostic procedures are performed by physicians, not counselors.
- Diagnosis of genetic disorders relies on laboratory and clinical confirmation, not solely counseling.
What is the chance of a fetus inheriting an autosomal recessive disorder if both parents are carriers?
Explanation
Autosomal recessive inheritance occurs when both parents carry one mutated allele of the same gene. Each child has a 25% chance of inheriting both mutated alleles (affected), a 50% chance of inheriting one mutated and one normal allele (carrier), and a 25% chance of inheriting two normal alleles (unaffected). The mutation must be present in both copies of the gene for the disorder to manifest. Disorders commonly inherited in this manner include cystic fibrosis, sickle cell anemia, and Tay-Sachs disease.
Rationale for correct answers
2. When both parents are carriers of the same autosomal recessive gene mutation, the probability of the fetus inheriting both defective alleles and expressing the disease is 25%. This is based on Mendelian genetics with a Punnett square cross of heterozygous parents.
Rationale for incorrect answers
1. A 10% chance is incorrect because Mendelian inheritance patterns follow 25%, 50%, or 0% probabilities, not 10%. Such a figure has no biological basis in autosomal recessive inheritance.
3. A 50% chance refers to the probability of the fetus being a carrier, not affected. Half of the offspring inherit one normal allele and one mutated allele but remain asymptomatic carriers.
4. A 75% chance is incorrect because 75% of the offspring are either unaffected or carriers. Only 25% inherit both mutated alleles and present with the disease.
Take home points
- Autosomal recessive inheritance requires both mutated alleles for disease expression.
- Each pregnancy has a 25% affected, 50% carrier, and 25% unaffected probability.
- Carriers are asymptomatic but can transmit the mutation to offspring.
- Disorders include cystic fibrosis, sickle cell disease, and Tay-Sachs disease.
Which of the following are purposes of carrier screening? Select all that apply
Explanation
Carrier screening is a genetic test that identifies if prospective parents carry mutations associated with inherited conditions. It focuses on autosomal recessive and X-linked disorders, where carriers are typically asymptomatic but can transmit the disease. Each pregnancy with two carriers has a 25% risk of an affected child. Disorders commonly screened include cystic fibrosis, spinal muscular atrophy, and hemoglobinopathies. Normal chromosomal complement is 46 (46,XX or 46,XY), but carrier screening does not detect chromosomal abnormalities, growth restriction, or fetal sex.
Rationale for correct answers
1. Carrier screening identifies parental genetic mutations associated with specific diseases. This allows couples to understand their reproductive risks and consider further testing or counseling.
3. Carrier screening assesses the risk of inherited conditions by determining if both parents carry the same mutation. If they do, the risk of having an affected child is 25%.
Rationale for incorrect answers
2. Carrier screening does not diagnose fetal chromosomal abnormalities. Chromosomal abnormalities such as trisomy 21 or 18 are detected by karyotyping, chorionic villus sampling, amniocentesis, or non-invasive prenatal testing.
4. Fetal growth is evaluated by ultrasound using biometric parameters like abdominal circumference, head circumference, and femur length. Carrier screening provides no information on growth.
5. Fetal sex can be determined by ultrasound after 14 weeks or through cell-free DNA testing, but not by carrier screening. Carrier testing focuses on parental mutations, not fetal chromosomal sex determination.
Take home points
- Carrier screening identifies parental mutations that can cause autosomal recessive or X-linked disorders.
- It assesses the risk of inherited disease but does not provide a diagnosis in the fetus.
- Chromosomal abnormalities, fetal growth, and sex determination are assessed by other modalities, not carrier screening.
- Carrier screening is most useful before or early in pregnancy for reproductive planning.
Which of the following are indications for referring a patient to genetic counseling? Select all that apply
Explanation
Genetic counseling is indicated when there is an increased risk of inherited or chromosomal abnormalities in pregnancy. Advanced maternal age (≥35 years at delivery) raises the risk of aneuploidy, such as trisomy 21, with risk increasing from 1 in 1,480 at age 20 to 1 in 85 at age 40. A family history of conditions like cystic fibrosis suggests increased genetic risk. Positive carrier screening results require counseling to explain inheritance and recurrence risk. Normal ultrasound or unrelated conditions like seasonal allergies are not indications.
Rationale for correct answers
1. Advanced maternal age increases the risk of chromosomal abnormalities, especially trisomy 21, 18, and 13, warranting referral for genetic counseling.
3. A family history of cystic fibrosis indicates a possible inherited risk, making genetic counseling essential for assessing recurrence and testing options.
4. Positive carrier screening results require genetic counseling to explain autosomal recessive or X-linked inheritance and reproductive risk.
Rationale for incorrect answers
2. Normal first-trimester ultrasound findings do not increase risk and do not require referral. Ultrasound is a reassuring finding rather than an indication for counseling.
5. Seasonal allergies are not genetically inherited in a Mendelian pattern and do not pose risks that require prenatal genetic counseling.
Take home points
- Genetic counseling is indicated for advanced maternal age due to increased risk of chromosomal abnormalities.
- A family history of genetic disorders like cystic fibrosis warrants referral.
- Positive carrier screening requires counseling to interpret inheritance and recurrence risks.
- Normal ultrasounds and conditions like seasonal allergies are not indications.
Practice Exercise 6
What is a key nursing responsibility before a patient undergoes amniocentesis?
Explanation
Amniocentesis is an invasive prenatal diagnostic procedure in which a needle is inserted into the amniotic sac to obtain fluid for genetic, biochemical, or maturity testing. It is typically performed between 15–20 weeks gestation. Risks include infection, miscarriage, and leakage of amniotic fluid. Normal amniotic fluid volume ranges from 8–24 cm by amniotic fluid index (AFI). Because the procedure carries risks, informed consent must be obtained before proceeding.
Rationale for correct answers
2. Ensuring informed consent is obtained is the key nursing responsibility before amniocentesis. The nurse verifies that the patient understands the purpose, risks, and benefits of the procedure and has signed the consent form before it is performed.
Rationale for incorrect answers
1. Pain medication is not typically required for amniocentesis. The procedure causes mild discomfort similar to venipuncture, and pain management is not a primary nursing responsibility.
3. Checking maternal blood glucose levels is not related to amniocentesis. Blood glucose monitoring is indicated for diabetic mothers but is not specific to this procedure.
4. A fetal stress test is not performed before amniocentesis. Instead, ultrasound guidance is used during the procedure to identify a safe pocket of fluid and avoid fetal injury.
Take home points
- Amniocentesis is an invasive diagnostic procedure requiring informed consent.
- The nurse ensures the patient understands risks and benefits before the procedure.
- Pain control and blood glucose monitoring are not routine pre-procedure responsibilities.
- Ultrasound, not fetal stress testing, guides needle placement during amniocentesis.
What is a key nursing role during prenatal testing?
Explanation
Prenatal testing includes screening and diagnostic procedures to evaluate fetal health, detect chromosomal or genetic abnormalities, and monitor pregnancy complications. Tests include ultrasound, maternal serum screening, cell-free DNA, chorionic villus sampling, and amniocentesis. Normal fetal heart rate is 110–160 beats per minute, often monitored during testing for reassurance. A key nursing role is to provide emotional support, helping patients manage anxiety and understand the process, while physicians and specialists perform and interpret the procedures.
Rationale for correct answers
2. Providing emotional support is a key nursing role during prenatal testing. Nurses help patients cope with stress, explain the procedures in simple terms, and offer reassurance throughout testing.
Rationale for incorrect answers
1. Nurses do not perform the ultrasound procedure. Sonographers or physicians are trained to perform and interpret obstetric ultrasounds.
3. Nurses do not analyze genetic test results. Laboratory geneticists and physicians interpret results, while nurses may reinforce explanations and provide support.
4. Nurses do not prescribe follow-up diagnostic tests. Ordering or prescribing tests is a physician’s responsibility, while the nurse facilitates communication and coordination of care.
Take home points
- Nurses play a supportive role in prenatal testing by reducing anxiety and offering reassurance.
- Performing, interpreting, or prescribing tests are physician and specialist responsibilities.
- Patient understanding and comfort are central to the nursing role.
- Emotional support improves patient experience and cooperation during testing.
A nurse is monitoring a patient post-amniocentesis. Which complication should the nurse assess for?
Explanation
Amniocentesis is an invasive diagnostic procedure performed between 15–20 weeks of gestation to obtain amniotic fluid for analysis. Risks include infection, miscarriage, and trauma to the fetus or placenta. A major complication is rupture of membranes leading to amniotic fluid leakage, which can result in oligohydramnios and fetal compromise. Normal amniotic fluid volume is 8–24 cm by amniotic fluid index (AFI). Nurses must closely monitor for maternal and fetal complications after the procedure.
Rationale for correct answers
2. Fluid leakage from the vagina indicates rupture of membranes and possible amniotic fluid loss, a known complication of amniocentesis. This finding requires immediate evaluation and intervention to prevent infection and preterm labor.
Rationale for incorrect answers
1. Elevated blood pressure is not a typical complication of amniocentesis. Hypertension in pregnancy is more associated with preeclampsia, not the procedure itself.
3. Increased fetal heart rate is not a common complication of amniocentesis. The nurse monitors fetal heart rate for bradycardia or decelerations, but tachycardia is not directly caused by the procedure.
4. Maternal weight gain is unrelated to amniocentesis. Weight gain occurs gradually during pregnancy and is not linked to immediate post-procedure complications.
Take home points
- Amniocentesis carries risks such as rupture of membranes and amniotic fluid leakage.
- Fluid leakage is a major complication requiring urgent follow-up.
- Elevated maternal blood pressure and weight gain are not associated with the procedure.
- Fetal monitoring post-procedure focuses on bradycardia or distress, not tachycardia.
Which of the following are nursing responsibilities post-amniocentesis? Select all that apply
Explanation
Amniocentesis is an invasive prenatal diagnostic test performed under ultrasound guidance to withdraw amniotic fluid for genetic or biochemical analysis. Normal amniotic fluid index (AFI) ranges from 8–24 cm. Post-procedure, the nurse must monitor for complications such as rupture of membranes, infection, and preterm labor. Nursing care focuses on maternal and fetal monitoring, recognition of complications, and providing education about danger signs to report.
Rationale for correct answers
1. Monitoring for fluid leakage is essential to detect rupture of membranes or persistent amniotic fluid loss, a potential complication of the procedure.
3. Monitoring fetal heart rate ensures fetal well-being and detects early signs of distress, such as bradycardia or decelerations.
5. Providing post-procedure instructions is a critical nursing role. Patients must be educated to report vaginal bleeding, contractions, fever, or fluid leakage immediately.
Rationale for incorrect answers
2. Administering pain medication is not routinely required post-amniocentesis. The procedure causes only mild discomfort, and analgesics are not standard post-procedure care.
4. Checking maternal blood glucose is not related to amniocentesis. This is specific to diabetic care, not post-procedure monitoring after amniotic fluid sampling.
Take home points
- Post-amniocentesis nursing care focuses on maternal and fetal safety.
- Fluid leakage, bleeding, or contractions are danger signs requiring immediate attention.
- Fetal heart rate monitoring ensures detection of early fetal distress.
- Patient education prevents delayed recognition of complications.
Which of the following are key components of patient education for antepartum diagnostic testing? Select all that apply
Explanation
Antepartum diagnostic testing includes invasive and non-invasive procedures performed during pregnancy to detect chromosomal, genetic, or structural abnormalities in the fetus. Examples are ultrasound, amniocentesis, and chorionic villus sampling. Normal fetal heart rate is 110–160 beats per minute, often monitored during testing. Patient education must cover procedure purpose, associated risks, available alternatives, and the potential emotional impact of results, as testing outcomes may affect pregnancy decisions. Nurses support understanding but do not independently perform or interpret these tests.
Rationale for correct answers
1. Explaining the purpose and risks ensures informed decision-making. Patients must understand why the test is done and the possible complications.
3. Discussing potential emotional impacts helps patients prepare for stress or anxiety related to abnormal results and future decisions.
4. Providing information on test alternatives allows patients to make informed choices, such as choosing between screening and diagnostic tests.
Rationale for incorrect answers
2. Nurses do not independently perform antepartum diagnostic procedures like amniocentesis or chorionic villus sampling. These require specialized physician training.
5. Interpreting genetic test results is done by genetic counselors, laboratory specialists, or physicians. Nurses can clarify results in simpler language but not provide formal interpretation.
Take home points
- Patient education for antepartum testing focuses on purpose, risks, alternatives, and emotional implications.
- Nurses do not independently perform or interpret diagnostic procedures.
- Emotional support is crucial due to anxiety associated with genetic testing.
- Informed consent is based on thorough patient education and understanding.
Comprehensive Questions
A client presents with polyhydramnios on ultrasound. This finding might prompt further evaluation for which of the following fetal conditions?
Explanation
Polyhydramnios is an excess of amniotic fluid with an amniotic fluid index (AFI) >24 cm or deepest vertical pocket >8 cm. It occurs in 1–2% of pregnancies and is associated with impaired fetal swallowing, maternal diabetes, or congenital anomalies. Normal AFI is 8–24 cm. Since the fetus continuously swallows and recycles amniotic fluid, structural gastrointestinal abnormalities such as esophageal atresia can lead to polyhydramnios due to impaired swallowing.
Rationale for correct answers
2. Esophageal atresia prevents the fetus from swallowing and absorbing amniotic fluid. This leads to accumulation, resulting in polyhydramnios, which is a common prenatal ultrasound finding in such cases.
Rationale for incorrect answers
1. Renal agenesis results in oligohydramnios, not polyhydramnios. Absence of fetal kidneys prevents urine production, reducing amniotic fluid volume.
3. Oligohydramnios is the opposite of polyhydramnios, defined as AFI <5 cm or single deepest pocket <2 cm. It is not a condition associated with increased amniotic fluid.
4. Fetal growth restriction may be associated with oligohydramnios due to placental insufficiency, not polyhydramnios.
Take home points
- Polyhydramnios is defined as AFI >24 cm or deepest vertical pocket >8 cm.
- It is commonly associated with impaired fetal swallowing, such as in esophageal atresia.
- Renal agenesis and growth restriction are linked with oligohydramnios, not polyhydramnios.
- Maternal diabetes is another important cause of polyhydramnios.
Which of the following "soft markers" identified on an anatomy ultrasound might suggest an increased risk for chromosomal abnormalities, potentially warranting further genetic counseling?
Explanation
Soft markers are subtle ultrasound findings that are not structural anomalies themselves but are associated with increased risk of chromosomal abnormalities, particularly trisomy 21. These include echogenic intracardiac focus (EIF), choroid plexus cysts, echogenic bowel, and nuchal fold thickening. EIF represents a small calcification in a papillary muscle of the heart and is seen in up to 5% of normal pregnancies, but its presence slightly raises the risk for aneuploidy. Normal fetal anatomy findings, such as a four-chamber heart, visible stomach bubble, and bilateral kidneys, are reassuring and not considered markers.
Rationale for correct answers
3. Echogenic intracardiac focus is a soft marker for chromosomal abnormalities, especially trisomy 21. While often benign, it may prompt referral for further genetic counseling depending on associated risk factors.
Rationale for incorrect answers
1. Presence of a four-chamber heart is a normal finding. It indicates appropriate cardiac development and does not suggest aneuploidy.
2. Normal bilateral kidneys confirm renal development and normal amniotic fluid production. This finding is not associated with chromosomal abnormalities.
4. A full stomach bubble is a normal ultrasound finding indicating fetal swallowing, which rules out gastrointestinal obstruction. It does not increase chromosomal abnormality risk.
Take home points
- Soft markers are subtle ultrasound findings linked to higher aneuploidy risk.
- EIF is one of the most common soft markers, associated with trisomy 21.
- Normal anatomical findings like heart chambers, kidneys, and stomach bubble are not markers.
- Further genetic counseling is considered when soft markers are present, especially with abnormal screening results.
A genetic counselor is discussing the potential for genetic discrimination with a client considering prenatal testing. Which act in the United States helps to protect individuals from discrimination based on genetic information in health insurance and employment?
Explanation
Genetic discrimination is the unfair treatment of individuals based on genetic information from testing or family history. Genetic testing can reveal risks for conditions such as cystic fibrosis, sickle cell disease, and Huntington’s disease. DNA mutations may predispose to disease but not guarantee occurrence. Genetic privacy laws aim to prevent misuse. Normal human karyotype is 46 chromosomes (44 autosomes and 2 sex chromosomes). Hemoglobin reference range is 13–17 g/dL for males and 12–15 g/dL for females.
Rationale for correct answers
3. The Genetic Information Nondiscrimination Act (GINA) specifically prohibits health insurers and employers from using genetic information for eligibility, premium setting, or hiring decisions. This law was enacted in 2008 to directly address genetic discrimination.
Rationale for incorrect answers
1. HIPAA protects patient health information confidentiality but does not specifically prevent discrimination based on genetic testing results. It focuses on privacy and security standards for health records.
2. The Americans with Disabilities Act (ADA) prevents discrimination against individuals with physical or mental disabilities but does not cover genetic predispositions in individuals not yet symptomatic.
4. The Affordable Care Act (ACA) prevents insurance denial due to preexisting conditions but does not specifically regulate employer or insurer use of genetic test results for predictive purposes.
Take home points
- GINA is the primary U.S. law preventing misuse of genetic information in health insurance and employment.
- HIPAA regulates privacy of health information but not genetic discrimination.
- ADA applies to existing disabilities, not genetic predisposition.
- ACA covers preexisting conditions but does not directly address genetic information misuse.
Which of the following is a common indication for recommending genetic counseling?
Explanation
Genetic counseling is a process that provides individuals and families with information on genetic risks, inheritance, and disease prevention. Chromosomal abnormalities such as trisomy 21, trisomy 18, and trisomy 13 are frequent causes of adverse outcomes. Karyotype analysis identifies chromosomal changes; normal human karyotype is 46 chromosomes. Pedigree assessment and detailed family history are critical in identifying risk. Normal maternal serum alpha-fetoprotein (MSAFP) is 16–95 ng/mL in the second trimester. Normal nuchal translucency measurement is <3.0 mm at 11–13 weeks gestation.
Rationale for correct answers
3. A previous child with a chromosomal abnormality is a strong indication for genetic counseling, as recurrence risk increases and parents require guidance on testing options, inheritance risk, and preventive strategies.
Rationale for incorrect answers
1. Maternal age 25 years is below the advanced maternal age threshold of 35 years, where the risk of nondisjunction and chromosomal abnormalities such as trisomy 21 significantly rises. Without family history, this does not warrant genetic counseling.
2. Normal first-trimester combined screening (nuchal translucency, free beta-hCG, and PAPP-A) indicates low risk for aneuploidy, and therefore does not necessitate referral for genetic counseling unless other risk factors are present.
4. A routine, uncomplicated pregnancy without risk factors does not require genetic counseling, as the likelihood of chromosomal abnormalities or inherited disease is not elevated.
Take home points
- Previous child with a chromosomal abnormality is a key indication for genetic counseling.
- Advanced maternal age ≥35 years increases risk of aneuploidy.
- Normal prenatal screening reduces need for specialized counseling.
- Uncomplicated pregnancies without risk factors do not require genetic counseling.
Which factor increases the likelihood of recommending diagnostic prenatal testing?
Explanation
Diagnostic prenatal testing such as chorionic villus sampling and amniocentesis is performed to directly examine fetal chromosomes or DNA. Advanced maternal age increases the risk of meiotic nondisjunction, leading to trisomy 21, trisomy 18, and trisomy 13. The risk of Down syndrome is 1 in 365 at age 35, and rises to 1 in 100 at age 40. Aneuploidy risk is the main determinant for offering invasive testing. Ultrasound markers such as nuchal translucency >3.0 mm or structural anomalies are also important triggers. Normal maternal fasting blood glucose is 70–99 mg/dL, and normal BMI is 18.5–24.9 kg/m², which do not directly impact the indication for invasive genetic testing.
Rationale for correct answer
1. Maternal age ≥35 years increases risk of fetal chromosomal abnormalities due to oocyte nondisjunction. This makes invasive diagnostic testing such as amniocentesis or CVS medically indicated.
Rationale for incorrect answers
2. History of gestational diabetes increases maternal risk for type 2 diabetes and macrosomia in the fetus, but does not elevate the risk for chromosomal abnormalities, so it is not an indication for diagnostic prenatal testing.
3. Previous preterm delivery raises concern for maternal or uterine factors such as infection, cervical insufficiency, or preeclampsia, but does not alter the chromosomal risk for the fetus, making diagnostic genetic testing unnecessary.
4. Maternal obesity increases risk of gestational diabetes, hypertensive disorders, and neural tube defects due to altered folate metabolism, but it is not in itself an indication for invasive prenatal genetic testing unless combined with abnormal screening.
Take home points
- Maternal age ≥35 years is a major risk factor for fetal chromosomal abnormalities.
- Invasive diagnostic tests are indicated when aneuploidy risk is elevated.
- Conditions like gestational diabetes or preterm delivery do not increase genetic abnormality risk.
- Maternal obesity increases obstetric risks but not direct chromosomal abnormality risk.
A client declines all prenatal screening and diagnostic tests. How should the nurse respond to this decision?
Explanation
Autonomy is the ethical principle that supports a client’s right to make informed decisions about their own care. Informed consent requires that clients receive accurate, balanced information about risks, benefits, and alternatives before deciding. Nonmaleficence obligates the nurse to avoid coercion or harm, while beneficence ensures care is provided respectfully. Documentation of the client’s choice ensures legal and ethical accountability. Normal fetal heart rate is 110–160 bpm, and normal amniotic fluid index is 8–24 cm, but these physiological parameters are not impacted by the client’s decision to decline testing.
Rationale for correct answer
3. Documenting the client’s decision and continuing respectful, non-judgmental care respects autonomy and maintains the therapeutic relationship while ensuring legal and ethical standards are met.
Rationale for incorrect answers
1. Attempting to persuade undermines client autonomy and may be perceived as coercive. Nurses are obligated to provide information but not pressure clients into testing.
2. Telling the client they are putting their baby at unnecessary risk is judgmental, undermines trust, and violates the principle of nonmaleficence. It may also cause undue guilt or anxiety.
4. Referring the client to another healthcare provider solely because they declined testing constitutes abandonment and violates professional responsibility to provide ongoing care.
Take home points
- Autonomy allows clients to decline prenatal screening and diagnostic testing.
- Nurses must provide balanced information without coercion.
- Documentation of decisions protects client rights and nurse accountability.
- Non-judgmental support maintains therapeutic trust and professional ethics.
Which of the following conditions is most commonly associated with elevated levels of Alpha-Fetoprotein (AFP) in the amniotic fluid?
Explanation
Alpha-fetoprotein (AFP) is a glycoprotein produced by the fetal yolk sac and liver, measurable in maternal serum and amniotic fluid. Neural tube defects such as anencephaly and spina bifida cause leakage of AFP into amniotic fluid, leading to elevated levels. Amniotic fluid AFP normally decreases after 13–15 weeks, while maternal serum AFP peaks at 16–18 weeks. Normal maternal serum AFP at 16–18 weeks is 16–95 ng/mL. Open defects allow direct transfer of fetal proteins into amniotic fluid, which explains the rise.
Rationale for correct answer
3. Anencephaly is an open neural tube defect where absence of cranial vault leads to marked leakage of AFP into amniotic fluid, making it strongly associated with elevated AFP.
Rationale for incorrect answers
1. Down syndrome (trisomy 21) is associated with decreased maternal serum and amniotic fluid AFP due to reduced fetal protein production, not elevation.
2. Trisomy 18 is also associated with decreased AFP, along with multiple structural anomalies, but not with elevations.
4. Cystic fibrosis affects chloride transport and mucus viscosity, not AFP metabolism or leakage, and does not elevate amniotic fluid AFP.
Take home points
- Elevated AFP in amniotic fluid is most strongly linked with neural tube defects.
- Down syndrome and trisomy 18 typically lower AFP levels.
- Cystic fibrosis does not impact AFP concentrations.
- Open fetal defects allow direct leakage of fetal proteins into amniotic fluid.
In the context of prenatal testing, what does the term “non-directiveness” imply in genetic counseling?
Explanation
Non-directiveness in genetic counseling is an ethical approach emphasizing autonomy and informed decision-making. Counselors provide accurate information on risks, inheritance patterns, and testing options without influencing or pressuring the client. Patient-centered support helps clients weigh benefits and risks while making personal choices. Normal risk of trisomy 21 at maternal age 30 is 1 in 900, and at 35 is 1 in 365. Genetic testing may include serum markers, amniocentesis, or chorionic villus sampling, but non-directiveness ensures the client chooses based on values, not counselor bias.
Rationale for correct answer
3. The counselor provides unbiased information about genetic risks, testing options, and outcomes while supporting the patient’s autonomous decisions, ensuring informed consent and respect for personal values.
Rationale for incorrect answers
1. Telling the patient what decision to make violates autonomy and the core principle of non-directiveness, creating coercion rather than informed choice.
2. Providing information only about positive test results is incomplete and biased, undermining informed decision-making and the ethical obligation to present full information.
4. Focusing on preventing the birth of a child with a disability imposes a value judgment and directive counseling, directly contradicting non-directiveness principles.
Take home points
- Non-directiveness supports patient autonomy in genetic counseling decisions.
- Counselors provide unbiased information on risks, benefits, and alternatives.
- The approach avoids coercion or directive advice.
- Ethical counseling respects personal values and informed consent.
What is the primary source of fetal cell-free DNA (cfDNA) found in the maternal bloodstream for NIPT?
Explanation
Cell-free fetal DNA (cfDNA) in maternal blood originates primarily from apoptotic placental trophoblasts, which release fragmented fetal DNA into the maternal circulation. Non-invasive prenatal testing (NIPT) analyzes these fragments to detect aneuploidies such as trisomy 21, 18, and 13. Fetal fraction typically ranges from 4–20% of total cfDNA, increasing with gestational age and maternal weight. Normal maternal plasma cfDNA clearance occurs within hours after delivery. cfDNA is not derived from fetal red blood cells, which are rapidly cleared, or fetal neuronal cells, which do not release detectable DNA into maternal circulation.
Rationale for correct answer
4. Apoptotic placental trophoblasts release fetal DNA fragments into maternal blood, providing the source of cfDNA for NIPT, allowing non-invasive detection of chromosomal abnormalities.
Rationale for incorrect answers
1. Fetal red blood cells are present in extremely low quantities in maternal blood and are rapidly cleared; they do not contribute meaningfully to cfDNA.
2. Maternal placental trophoblasts are maternal in origin and do not release fetal DNA fragments, so they are not the source of cfDNA used in NIPT.
3. Fetal neuronal cells do not release DNA into maternal circulation in detectable amounts, making them irrelevant for cfDNA analysis.
Take home points
- cfDNA in maternal blood is derived from apoptotic placental trophoblasts.
- NIPT analyzes cfDNA to detect trisomy 21, 18, and 13.
- Fetal fraction ranges 4–20%, increasing with gestational age.
- Fetal red blood cells and neuronal cells do not provide usable cfDNA.
A pregnant client with a history of a previous child with Trisomy 13 is considering genetic testing for her current pregnancy. Which prenatal diagnostic test would offer the most rapid definitive diagnosis in the early second trimester?
Explanation
Trisomy 13 (Patau syndrome) is a chromosomal disorder caused by an extra copy of chromosome 13, leading to severe congenital anomalies such as cleft lip/palate, holoprosencephaly, and congenital heart defects. Prenatal diagnosis relies on direct chromosomal analysis. Amniocentesis performed at 15–20 weeks gestation samples amniotic fluid containing fetal cells, which can undergo fluorescence in situ hybridization (FISH) for rapid detection of aneuploidy. Normal fetal karyotype is 46,XX or 46,XY. Serum AFP is 16–95 ng/mL at 16–18 weeks, and normal nuchal translucency is <3.0 mm; these are screening, not diagnostic, measures.
Rationale for correct answer
3. Amniocentesis with FISH allows direct analysis of fetal chromosomes from amniotic fluid cells, providing a rapid and definitive diagnosis of trisomy 13 in the early second trimester.
Rationale for incorrect answers
1. Maternal serum alpha-fetoprotein (MSAFP) is a screening tool for neural tube defects and abdominal wall defects, not for chromosomal aneuploidies like trisomy 13.
2. Nuchal translucency (NT) scan is an early first-trimester screening method indicating risk for trisomy 21 and 18, but it cannot provide a definitive diagnosis for trisomy 13.
4. Non-invasive prenatal testing (NIPT) can detect trisomy 13 with high sensitivity, but it is still considered a screening test and may require confirmatory diagnostic testing for a definitive result.
Take home points
- Trisomy 13 results from an extra copy of chromosome 13, causing severe congenital anomalies.
- Amniocentesis with FISH provides definitive early second-trimester diagnosis.
- Screening tests like MSAFP, NT scan, and NIPT assess risk but are not definitive.
- Rapid and accurate chromosomal analysis is critical when there is a history of previous aneuploidy.
Which of the following conditions can be detected or strongly suggested by analysis of amniotic fluid obtained during amniocentesis? Select all that apply
Explanation
Amniocentesis is a prenatal diagnostic procedure used to evaluate fetal health, detect genetic abnormalities, and assess biochemical markers. It involves transabdominal aspiration of amniotic fluid, typically performed between 15 and 20 weeks gestation. Normal amniotic fluid alpha-fetoprotein (AFP) levels are <2.0 multiples of the median (MoM); ΔOD450 values <0.15 indicate no significant hemolysis. Fetal cells in the fluid allow karyotyping and molecular analysis.
Rationale for correct answers
1. Amniotic fluid spectrophotometry (ΔOD450) detects bilirubin levels, which correlate with fetal hemolysis severity in Rh isoimmunization. ΔOD450 >0.25 suggests moderate to severe anemia due to red cell destruction.
2. Elevated AFP in amniotic fluid (>2.5 MoM) and positive acetylcholinesterase confirm open neural tube defects like spina bifida or anencephaly. These markers are released directly into the fluid from exposed neural tissue.
3. Fetal cells obtained during amniocentesis allow chromosomal analysis via karyotyping or microarray. Trisomies such as 21, 18, and 13 are reliably detected through cytogenetic evaluation.
4. DNA extracted from fetal cells enables targeted mutation analysis for monogenic disorders. Cystic fibrosis is confirmed by identifying CFTR gene mutations, especially ΔF508, using PCR or sequencing.
Rationale for incorrect answer
5. Confined placental mosaicism refers to chromosomal abnormalities present only in placental tissue, not in fetal cells. Amniocentesis samples fetal-derived cells, so placental mosaicism may be missed. Chorionic villus sampling is preferred for placental cytogenetics.
Take home points
- Amniocentesis enables biochemical and genetic evaluation of fetal health.
- AFP and acetylcholinesterase are key markers for neural tube defects.
- ΔOD450 spectrophotometry assesses fetal anemia severity in Rh disease.
- Confined placental mosaicism requires chorionic villus sampling, not amniocentesis.
Which of the following are valid reasons for a referral to genetic counseling during pregnancy? Select all that apply
Explanation
Genetic counseling is a specialized process that provides risk assessment, education, and support for families with potential heritable conditions. It integrates family history, screening results, and diagnostic findings to guide reproductive decisions. Referral is indicated when maternal age ≥35 years, abnormal ultrasound findings, or known familial mutations are present. First-trimester combined screening includes nuchal translucency, PAPP-A, and free β-hCG; normal values reduce risk but do not eliminate it.
Rationale for correct answers
1. Maternal age ≥35 years increases the risk of chromosomal aneuploidies, especially trisomy 21. This threshold warrants counseling to discuss screening and diagnostic options.
2. A prior child with a confirmed genetic disorder significantly elevates recurrence risk. Counseling helps assess inheritance patterns and guide prenatal testing.
4. Soft markers (e.g., echogenic intracardiac focus, choroid plexus cysts) may be associated with chromosomal abnormalities. Their presence on anatomy scan prompts further evaluation and counseling.
5. A family history of inherited conditions (e.g., cystic fibrosis, Tay-Sachs disease) necessitates counseling to assess carrier status and risk to the fetus.
Rationale for incorrect answers
3. Normal first-trimester combined screening results indicate low risk for common aneuploidies. In the absence of other risk factors, this does not warrant referral. Screening is not diagnostic and cannot detect all genetic conditions, but normal results reduce the likelihood of referral unless other concerns arise.
Take home points
- Maternal age ≥35 years increases risk for chromosomal abnormalities.
- Soft markers on ultrasound may suggest underlying genetic conditions.
- Family history and prior affected children are strong indications for counseling.
- Normal screening results reduce risk but do not eliminate need for further evaluation if other factors exist.
During an anatomy ultrasound, the sonographer assesses various fetal systems. Which of the following are key systems evaluated during this comprehensive scan? Select all that apply
Explanation
Fetal anatomy ultrasound is a detailed structural survey performed between 18 and 22 weeks gestation to evaluate organ development, detect congenital anomalies, and guide perinatal management. It assesses fetal biometry, placental location, and amniotic fluid volume. Normal biparietal diameter ranges from 42–60 mm at 20 weeks; femur length averages 32 mm. Key systems visualized include the brain, heart, kidneys, spine, and gastrointestinal tract. Hormonal glands like the thyroid and adrenal are not routinely assessed unless clinically indicated.
Rationale for correct answers
1. The central nervous system is evaluated via visualization of the cerebral ventricles, cerebellum, cisterna magna, and spine. Abnormalities like ventriculomegaly or spina bifida are detectable.
2. The cardiac system is assessed through four-chamber and outflow tract views. Structural defects such as ventricular septal defects or transposition of the great arteries can be identified.
3. The gastrointestinal system is evaluated by visualizing the stomach bubble and bowel echogenicity. Findings like duodenal atresia or echogenic bowel may suggest underlying pathology.
4. The renal system is assessed by identifying both kidneys and bladder. Renal agenesis, hydronephrosis, or multicystic dysplastic kidneys are detectable.
Rationale for incorrect answer
5. The endocrine system is not a standard component of routine anatomy scans. Glands such as the thyroid, pancreas, and adrenal are not consistently visualized unless there is a specific indication. Functional endocrine assessment requires biochemical testing, not sonographic imaging.
Take home points
- Anatomy ultrasound evaluates structural development, not endocrine function.
- CNS, cardiac, renal, and GI systems are core components of fetal anatomy scans.
- Endocrine glands are not routinely assessed unless clinically indicated.
- Ultrasound findings guide further genetic testing and perinatal planning.
Which of the following conditions may be associated with an abnormally low level of Pregnancy-Associated Plasma Protein-A (PAPP-A) in the first-trimester maternal serum screening? Select all that apply
Explanation
Low Pregnancy-Associated Plasma Protein-A (PAPP-A) PAPP-A is a glycoprotein produced by the placenta and trophoblasts, essential for fetal growth and placental function. It modulates insulin-like growth factor (IGF) bioavailability. Low levels in first-trimester maternal serum (<0.4 multiples of the median [MoM]) are associated with chromosomal abnormalities, placental insufficiency, and adverse pregnancy outcomes. Normal range is 0.5–2.0 MoM. It is measured as part of the combined screening with free β-hCG and nuchal translucency.
Rationale for correct answers
1. Low PAPP-A is a recognized marker in first-trimester screening for Down syndrome (trisomy 21). It reflects impaired trophoblastic function and abnormal placentation.
2. Trisomy 18 is strongly associated with low PAPP-A due to severe placental dysfunction and poor fetal development. It is often accompanied by low β-hCG and increased nuchal translucency.
3. Preeclampsia is linked to low PAPP-A due to early placental insufficiency and abnormal trophoblast invasion. It predicts increased risk for hypertensive disorders of pregnancy.
5. Fetal growth restriction (FGR) correlates with low PAPP-A, reflecting compromised placental nutrient transfer and impaired IGF regulation. It is a marker of poor fetal prognosis.
Rationale for incorrect answer
4. Neural tube defects are not associated with low PAPP-A. They are detected by elevated alpha-fetoprotein (AFP) in maternal serum or amniotic fluid. PAPP-A levels do not reflect neural tube integrity or closure defects.
Take home points
- Low PAPP-A (<0.4 MoM) suggests placental dysfunction and adverse outcomes.
- Associated conditions include trisomy 21, trisomy 18, preeclampsia, and fetal growth restriction.
- Neural tube defects are linked to elevated AFP, not PAPP-A.
The ethical principle of autonomy in prenatal testing emphasizes which of the following? Select all that apply
Explanation
Autonomy is a foundational bioethical principle that upholds the individual’s right to make informed decisions about their healthcare. In prenatal testing, it mandates that pregnant individuals receive complete, unbiased information and are supported in making choices that align with their values. Autonomy requires freedom from coercion and emphasizes non-directive counseling. It does not prioritize provider preferences or impose normative judgments about testing decisions.
Rationale for correct answers
2. Autonomy guarantees the pregnant individual’s right to accept or decline any test. This includes invasive procedures like amniocentesis or noninvasive screening, regardless of provider recommendation.
3. Complete and unbiased information is essential for informed consent. Autonomy requires that risks, benefits, and limitations of all testing options be clearly communicated without persuasion.
4. Non-directive counseling supports autonomous decision-making by presenting options neutrally, allowing individuals to choose based on personal values rather than provider influence.
5. Autonomy includes the right to make decisions free from coercion, pressure, or manipulation. This protects the integrity of informed consent and respects individual agency.
Rationale for incorrect answers
1. The healthcare provider’s right to recommend the “best” test reflects beneficence, not autonomy. While providers may offer guidance, autonomy prioritizes the patient’s values and choices over professional judgment. Recommendations must be balanced with respect for the individual’s decision-making authority.
Take home points
- Autonomy centers the pregnant individual’s right to informed, voluntary decision-making.
- Non-directive counseling and unbiased information are essential to ethical prenatal care.
- Provider recommendations must not override patient autonomy.
- Coercion undermines informed consent and violates ethical standards.
Exams on Antepartum Diagnostic Testing
Custom Exams
Login to Create a Quiz
Click here to loginLessons
 Naxlex
Just Now
Naxlex
Just Now
- Objectives
- Introduction
- Overview Of Antepartum Testing
- Practice Exercise 1
- Screening Tests
- Practice Exercise 2
- Diagnostic Tests
- Practice Exercise 3
- Ultrasound Examinations In Antepartum Care
- Practice Exercise 4
- Carrier Screening And Genetic Counseling
- Practice Exercise 5
- Nursing Considerations In Antepartum Diagnostic Testing
- Practice Exercise 6
- Summary
- Comprehensive Questions
Notes Highlighting is available once you sign in. Login Here.
Objectives
- Understand the Classification and Indications for Antepartum Testing: Describe the differences between screening and diagnostic tests, including their purposes, methodologies, and clinical indications, to guide appropriate patient care decisions.
- Evaluate Ethical and Cultural Considerations in Prenatal Testing: Analyze the ethical principles, such as autonomy and non-maleficence, and cultural factors that influence patient decision-making in antepartum diagnostic testing.
- Comprehend Screening Test Methodologies and Interpretations: Explain the scientific basis, procedural details, and clinical significance of screening tests, including nuchal translucency ultrasound, maternal serum screening, non-invasive prenatal testing (NIPT), blood type and Rh factor screening, and rubella titer testing.
- Analyze Diagnostic Test Procedures and Risks: Detail the procedural techniques, gestational timing, and potential complications of diagnostic tests such as chorionic villus sampling (CVS), amniocentesis, cordocentesis, and biophysical profile (BPP) to ensure safe patient management.
- Assess Ultrasound Applications in Antepartum Care: Describe the role of ultrasound examinations, including anomaly ultrasound and specialized assessments, in evaluating fetal anatomy and well-being during pregnancy.
- Understand Carrier Screening and Genetic Counseling: Elucidate the principles of carrier screening and the role of genetic counseling in supporting informed patient choices regarding prenatal testing.
- Apply Nursing Considerations in Antepartum Testing: Identify key nursing responsibilities, including patient education, informed consent, procedural preparation, monitoring, emotional support, and post-procedure care, to optimize patient outcomes.
- Synthesize Knowledge Through Practice and Comprehensive Questions: Demonstrate mastery of antepartum diagnostic testing concepts by answering targeted practice questions and comprehensive questions that reinforce clinical and theoretical knowledge.
Overview Of Antepartum Testing
Antepartum testing encompasses a spectrum of screening and diagnostic procedures utilized during pregnancy to evaluate fetal health, detect congenital anomalies, and identify maternal conditions that may influence pregnancy outcomes. These tests are integral to prenatal care, enabling healthcare providers to assess risks, implement timely interventions, and facilitate informed decision-making for expectant parents. The tests vary in invasiveness, timing, and purpose, ranging from non-invasive screening modalities to invasive diagnostic procedures that provide definitive results. Understanding the classification, indications, and ethical considerations of these tests is critical for nurses to deliver evidence-based care and support patients effectively.
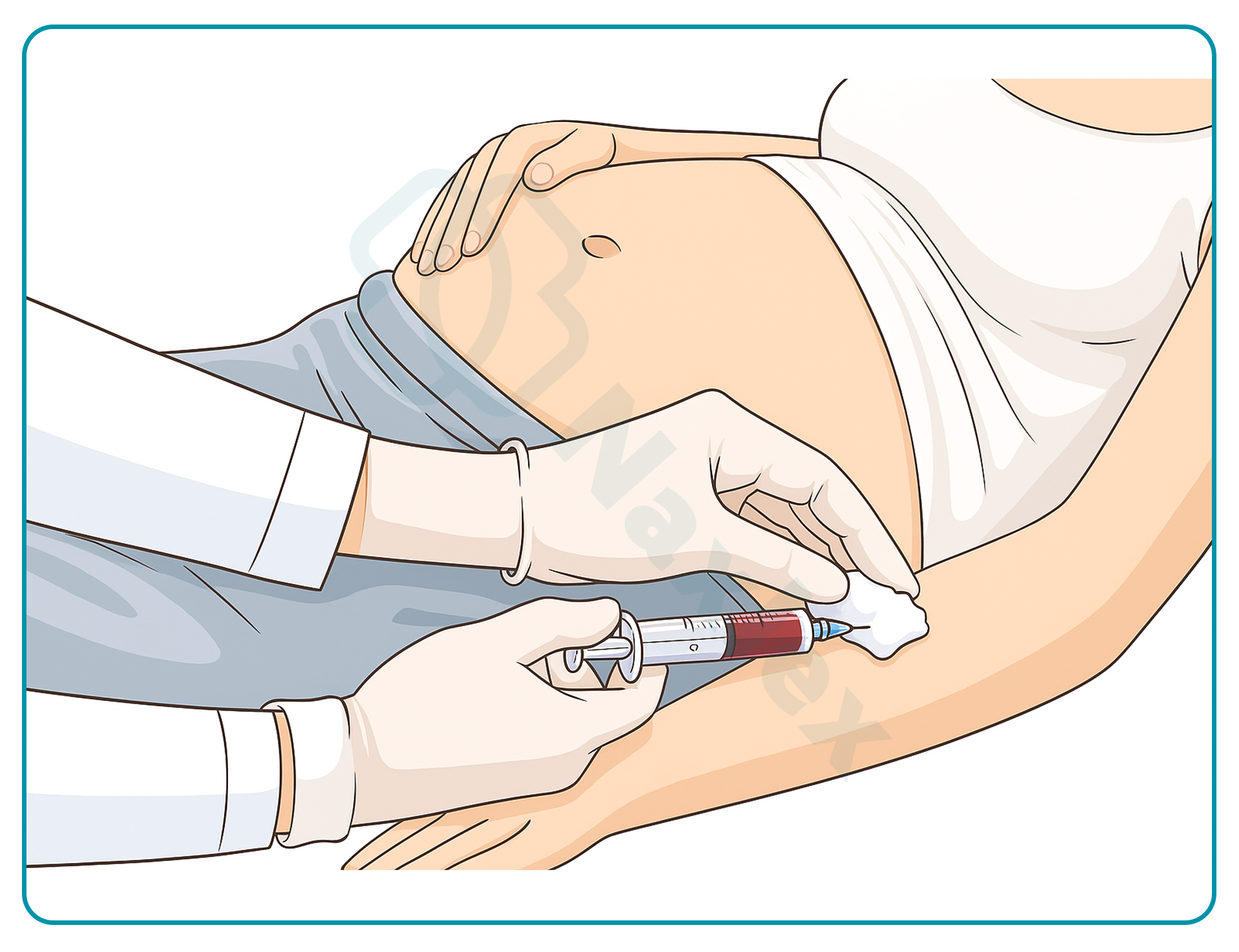
1.1 Classification of Prenatal Tests
Prenatal tests are broadly categorized into screening and diagnostic tests, each serving distinct purposes in assessing fetal health. Screening tests estimate the risk of specific conditions without providing a definitive diagnosis, while diagnostic tests confirm the presence or absence of abnormalities. The classification is based on the test’s methodology, invasiveness, and clinical utility.
- Screening Tests:
- Screening tests are non-invasive procedures designed to identify pregnancies at increased risk for congenital anomalies, such as chromosomal abnormalities or structural defects.
- These tests include nuchal translucency ultrasound, maternal serum screening (first-trimester combined screening and second-trimester quadruple screen), non-invasive prenatal testing (NIPT), blood type and Rh factor screening, and rubella titer testing.
- Results are reported as risk ratios (e.g., 1:100 for Down syndrome), indicating the likelihood of a condition but requiring diagnostic confirmation for definitive results.
- Screening tests are typically offered to all pregnant individuals, regardless of risk factors, due to their non-invasive nature and low risk to the fetus.
- Diagnostic Tests:
- Diagnostic tests are invasive procedures that provide definitive confirmation of fetal conditions, such as chromosomal aneuploidies or genetic disorders.
- Examples include chorionic villus sampling (CVS), amniocentesis, cordocentesis (percutaneous umbilical blood sampling), and biophysical profile (BPP).
- These tests carry a small risk of complications, such as miscarriage or infection, and are typically reserved for high-risk pregnancies or following abnormal screening results.
- Diagnostic tests analyze fetal cells, amniotic fluid, or fetal blood to provide precise genetic or physiological information.
Nursing Insights: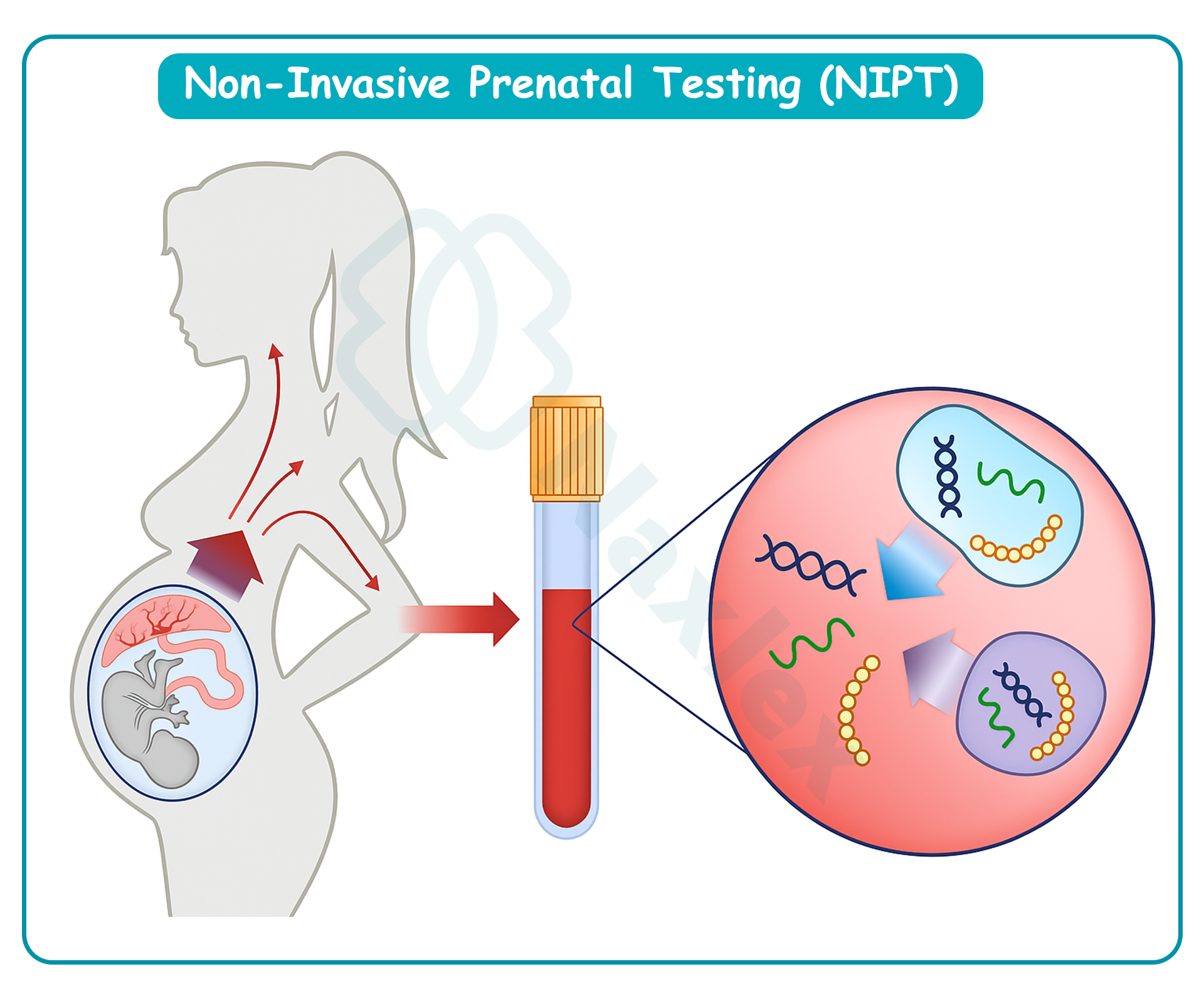
- Nurses must differentiate between screening and diagnostic tests when educating patients to ensure accurate expectations about test outcomes.
- Screening test results are reported as probabilities, not diagnoses, and nurses must clarify this distinction to prevent patient misunderstanding.
- Diagnostic tests require informed consent due to their invasive nature and associated risks, and nurses must ensure patients understand these risks before proceeding.
- Nurses should assess patient understanding of the purpose and limitations of each test type to facilitate informed decision-making.
1.2 Indications for Antepartum Testing
Antepartum diagnostic testing is indicated based on maternal, fetal, or familial risk factors that increase the likelihood of adverse pregnancy outcomes. These indications guide healthcare providers in recommending appropriate tests to optimize maternal and fetal health.
- Maternal Risk Factors:
- Advanced maternal age (≥35 years at delivery) increases the risk of chromosomal abnormalities, such as Down syndrome (trisomy 21), prompting recommendations for screening or diagnostic testing.
- Maternal medical conditions, such as diabetes or hypertension, may necessitate specialized testing to assess fetal well-being.
- Exposure to teratogens (e.g., medications, infections, or environmental toxins) during pregnancy warrants further evaluation to detect potential fetal anomalies.
- Fetal Risk Factors:
- Abnormal findings on routine ultrasound, such as increased nuchal translucency or soft markers (e.g., echogenic intracardiac focus), indicate a need for further diagnostic testing.
- Suspected fetal growth restriction or abnormal amniotic fluid volume (e.g., polyhydramnios or oligohydramnios) may prompt tests like the biophysical profile to assess fetal well-being.
- Fetal anemia, often due to Rh isoimmunization, requires diagnostic procedures like cordocentesis to confirm and guide management.
- Familial Risk Factors:
- A family history of genetic disorders, such as cystic fibrosis or sickle cell disease, necessitates carrier screening to assess the risk of inherited conditions.
- A previous pregnancy affected by a chromosomal abnormality or congenital anomaly increases the likelihood of recommending diagnostic testing in subsequent pregnancies.
- Screening Test Results:
- Abnormal results from screening tests, such as elevated maternal serum alpha-fetoprotein (MSAFP) or high-risk NIPT results, indicate the need for diagnostic testing to confirm findings.
- False-positive or inconclusive screening results may require follow-up diagnostic procedures to clarify the fetal status.
Nursing Insights:
- Nurses must obtain a comprehensive maternal and family history to identify risk factors that may warrant specific prenatal tests.
- Patients with abnormal screening results should be referred promptly for genetic counseling to discuss the implications and options for diagnostic testing.
- Nurses should monitor patients with high-risk indications closely, coordinating with the healthcare team to ensure timely testing and intervention.
- Clear communication of test indications helps patients understand the rationale for recommended procedures, enhancing compliance and trust.
1.3 Ethical and Cultural Considerations in Testing
Antepartum diagnostic testing raises significant ethical and cultural considerations that influence patient decision-making and nursing care. Ethical principles, such as autonomy, beneficence, and non-maleficence, guide the approach to prenatal testing, while cultural beliefs shape patient preferences and acceptance of testing.
- Ethical Principles:
- Autonomy: Pregnant individuals have the right to accept or decline prenatal testing without coercion, and nurses must respect their decisions.
- Beneficence: Testing aims to promote the well-being of the mother and fetus by identifying conditions that may benefit from early intervention.
- Non-maleficence: Nurses must ensure that the risks of testing, such as miscarriage from invasive procedures, are minimized and clearly communicated.
- Justice: Access to prenatal testing should be equitable, with nurses advocating for patients who face barriers due to socioeconomic or systemic factors.
- Cultural Considerations:
- Cultural beliefs about pregnancy, disability, or medical interventions may influence a patient’s willingness to undergo testing.
- Some cultures may view prenatal testing as interfering with natural processes, necessitating sensitive counseling to align care with patient values.
- Language barriers or differing family dynamics (e.g., involvement of extended family in decision-making) require nurses to adapt communication strategies.
- Genetic Discrimination:
- The Genetic Information Nondiscrimination Act (GINA) in the United States protects individuals from discrimination based on genetic test results in health insurance and employment.
- Nurses must educate patients about protections under GINA to alleviate fears of genetic discrimination when considering testing.
- Non-Directive Counseling:
- Genetic counseling and nursing education must remain non-directive, providing unbiased information to support patient decision-making without influencing choices.
- Nurses should avoid imposing personal beliefs or assumptions about the “best” course of action, particularly regarding decisions about pregnancy continuation.
Nursing Insights:
- Nurses must provide culturally sensitive education, using interpreters if needed, to ensure patients understand testing options in the context of their beliefs.
- Informed consent discussions should include ethical implications, such as the potential psychological impact of test results, to prepare patients for decision-making.
- Nurses should advocate for equitable access to testing, connecting patients with resources to overcome financial or logistical barriers.
- Documentation of a patient’s decision to decline testing is essential to respect autonomy and ensure continuity of non-judgmental care.
Screening Tests
Screening tests are non-invasive procedures designed to assess the risk of fetal anomalies, such as chromosomal abnormalities or structural defects, without posing significant risk to the mother or fetus. These tests are typically offered to all pregnant individuals and provide probabilistic results that may necessitate further diagnostic evaluation. Key screening tests include nuchal translucency ultrasound, maternal serum screening, and non-invasive prenatal testing (NIPT).
1.1 Nuchal Translucency Ultrasound
The nuchal translucency (NT) ultrasound is a first-trimester screening test performed between 11 and 14 weeks of gestation to assess the risk of chromosomal abnormalities, particularly Down syndrome (trisomy 21) and trisomy 18. The test measures the fluid-filled space at the back of the fetal neck, which is often increased in fetuses with chromosomal or structural anomalies.
- Procedure:
- The NT ultrasound is performed transabdominally using high-frequency sound waves to visualize the fetal neck.
- The measurement of nuchal translucency is taken in the sagittal plane, with the fetus in a neutral position, and requires precise crown-rump length (CRL) measurement (45–84 mm) for accuracy.
- The test is often combined with maternal serum markers (first-trimester combined screening) to enhance detection rates.
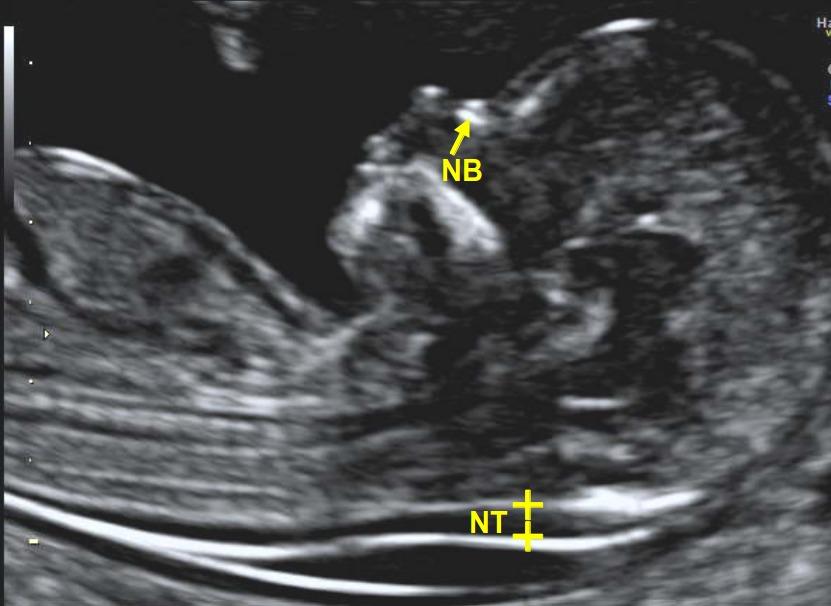
- Clinical Significance:
- An increased NT measurement (>3 mm) is associated with an elevated risk of chromosomal abnormalities, congenital heart defects, and other syndromes.
- The test has a detection rate of approximately 70–80% for Down syndrome when performed alone, increasing to 85–90% when combined with serum markers.
- Results are reported as a risk ratio, adjusted for maternal age and gestational age.
- Limitations:
- The NT ultrasound is a screening tool, not a diagnostic test, and requires follow-up with invasive procedures (e.g., CVS or amniocentesis) for confirmation.
- False-positive results may occur, particularly in low-risk populations, necessitating careful counseling to avoid undue anxiety.
- The accuracy of the test depends on the sonographer’s expertise and adherence to standardized measurement protocols.
Nursing Insights:
- Nurses must ensure patients maintain a full bladder for transabdominal NT ultrasound to optimize visualization of the fetus.
- Patients should be educated that an increased NT measurement does not confirm a diagnosis but indicates a need for further testing.
- Nurses should assess for patient anxiety related to NT results and provide emotional support, referring to genetic counseling as needed.
- Documentation of NT results and patient education is critical for continuity of care and follow-up planning.
1.2 Maternal Serum Screening
Maternal serum screening involves analyzing maternal blood samples to measure specific biomarkers associated with fetal anomalies. These tests are performed in the first or second trimester and provide risk assessments for chromosomal abnormalities and neural tube defects.
1.2.1 First-Trimester Combined Screening
The first-trimester combined screening integrates the NT ultrasound with maternal serum markers to assess the risk of chromosomal abnormalities, primarily Down syndrome and trisomy 18, between 11 and 14 weeks of gestation.
- Components:
- Nuchal Translucency Measurement: As described, measures fluid accumulation at the fetal neck.
- Maternal Serum Markers:
- Pregnancy-associated plasma protein-A (PAPP-A): Low levels are associated with Down syndrome, trisomy 18, preeclampsia, and fetal growth restriction.
- Free beta-human chorionic gonadotropin (free β-hCG): Elevated levels are associated with Down syndrome, while low levels may indicate trisomy 18.
- The combination of NT and serum markers provides a detection rate of approximately 85–90% for Down syndrome with a 5% false-positive rate.
- Procedure:
- A maternal blood sample is collected concurrently with the NT ultrasound, typically between 11 and 13 weeks.
- Results are adjusted for maternal age, weight, ethnicity, and gestational age to generate a risk ratio.
- Clinical Significance:
- Abnormal results (e.g., low PAPP-A or high free β-hCG) indicate an increased risk of chromosomal abnormalities or adverse pregnancy outcomes.
- The test is highly sensitive but not diagnostic, requiring follow-up with CVS or amniocentesis for confirmation.
- Limitations:
- The test’s accuracy depends on precise gestational dating and maternal factors, such as body mass index, which may affect serum marker levels.
- False-positive results can lead to unnecessary anxiety and invasive testing, necessitating thorough patient counseling.
Nursing Insights:
- Nurses must verify accurate gestational dating before maternal serum screening to ensure reliable results.
- Patients should be informed that first-trimester screening results are risk estimates, not definitive diagnoses, to manage expectations.
- Nurses should coordinate with the laboratory to ensure timely blood sample collection and result reporting.
- Emotional support is critical for patients receiving abnormal results, with referrals to genetic counseling for further discussion.
1.2.2 Second-Trimester Quadruple Screen
The quadruple screen, performed between 15 and 22 weeks of gestation, measures four maternal serum markers to assess the risk of Down syndrome, trisomy 18, and open neural tube defects, such as spina bifida or anencephaly.
- Components:
- Alpha-Fetoprotein (AFP): Elevated levels indicate a risk of neural tube defects, while low levels are associated with Down syndrome.
- Human Chorionic Gonadotropin (hCG): High levels are associated with Down syndrome, while low levels may indicate trisomy 18.
- Unconjugated Estriol (uE3): Low levels are associated with Down syndrome and trisomy 18.
- Inhibin-A: Elevated levels are associated with Down syndrome.
- Procedure:
- A maternal blood sample is collected, typically between 16 and 18 weeks for optimal accuracy.
- Results are reported as a risk ratio, adjusted for maternal age, weight, and gestational age.
- Clinical Significance:
- The quadruple screen has a detection rate of approximately 80% for Down syndrome and 60–70% for neural tube defects, with a 5% false-positive rate.
- Abnormal results prompt further evaluation with diagnostic tests, such as amniocentesis or detailed ultrasound.
- Limitations:
- The test does not detect all congenital anomalies, and false-positive results may occur, particularly in multifetal pregnancies or inaccurate gestational dating.
- Certain maternal conditions, such as diabetes, may affect AFP levels, requiring adjusted interpretation.
Nursing Insights:
- Nurses must educate patients about the timing of the quadruple screen to ensure it is performed within the optimal gestational window.
- Patients with abnormal AFP levels should be referred for a detailed ultrasound to assess for neural tube defects or other anomalies.
- Nurses should clarify that the quadruple screen assesses risk, not diagnosis, to prevent misinterpretation of results.
- Coordination with the healthcare team is essential to ensure follow-up testing is arranged promptly for abnormal results.
1.3 Non-Invasive Prenatal Testing (NIPT)
Non-invasive prenatal testing (NIPT) is a highly sensitive screening test that analyzes cell-free fetal DNA (cfDNA) in maternal blood to assess the risk of chromosomal aneuploidies, such as Down syndrome, trisomy 18, and trisomy 13, as early as 10 weeks of gestation.
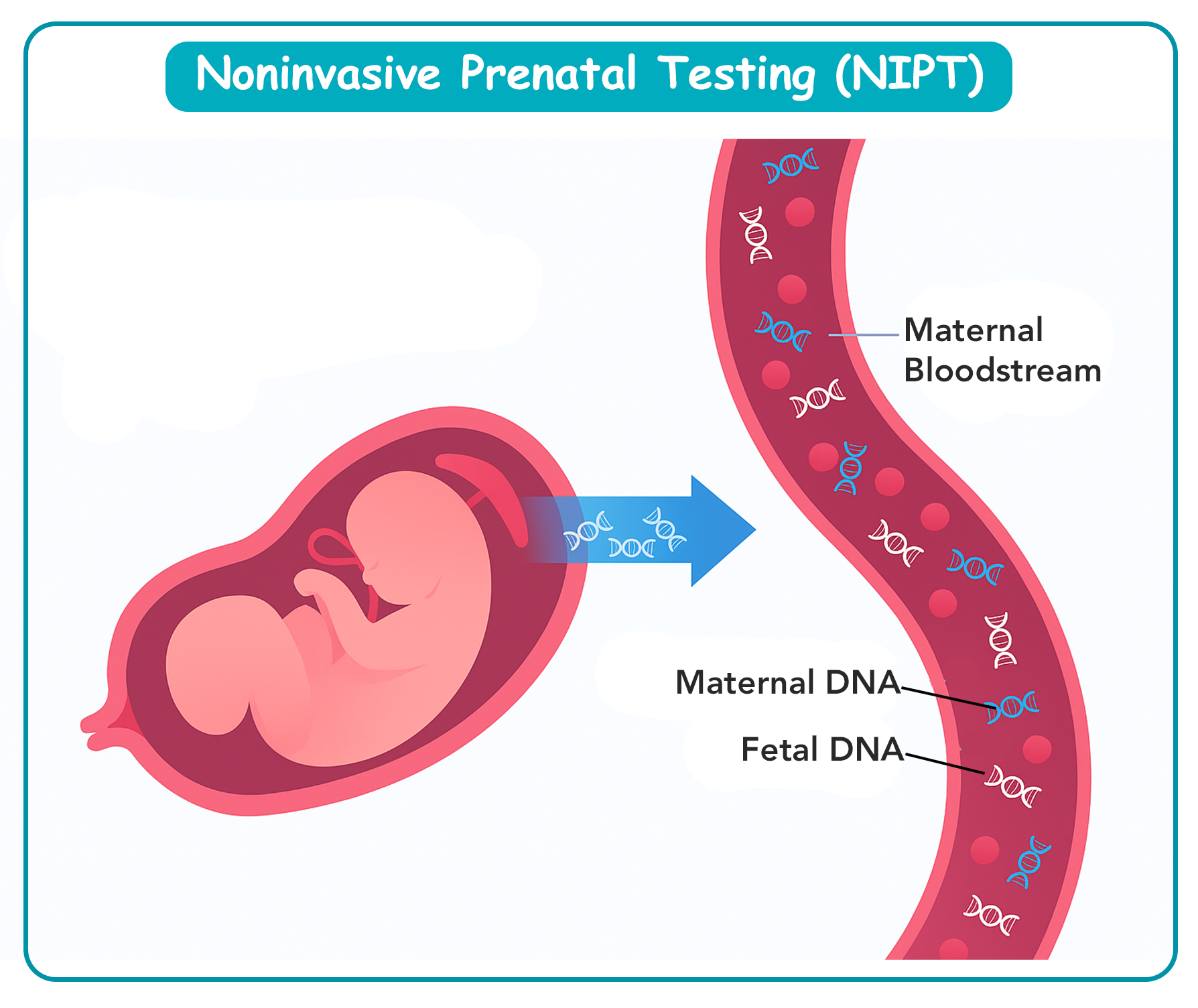
- Procedure:
- A maternal blood sample is collected, and cfDNA, originating primarily from apoptotic placental trophoblasts, is analyzed using next-generation sequencing or microarray technology.
- The test evaluates the proportion of fetal DNA (fetal fraction) to ensure sufficient material for accurate analysis (typically >4% fetal fraction).
- Clinical Significance:
- NIPT has a detection rate of >99% for Down syndrome, with a false-positive rate of <0.1%, making it more accurate than traditional serum screening.
- The test can also screen for sex chromosome aneuploidies (e.g., Turner syndrome) and determine fetal sex.
- NIPT may include panels for microdeletions or microduplications, though sensitivity for these conditions varies.
- Limitations:
- NIPT is a screening test, not diagnostic, and positive results require confirmation with CVS or amniocentesis due to the possibility of confined placental mosaicism.
- A low fetal fraction, often seen in early gestation or maternal obesity, may result in a “no call” result, necessitating re-draw or alternative testing.
- NIPT does not screen for structural anomalies, such as neural tube defects, requiring complementary ultrasound evaluation.
- Indications:
- NIPT is recommended for high-risk pregnancies, such as those with advanced maternal age, abnormal ultrasound findings, or a history of chromosomal abnormalities.
- It is increasingly offered to low-risk populations due to its high sensitivity and non-invasive nature.
Nursing Insights:
- Nurses must explain that NIPT is a screening test, and positive results require diagnostic confirmation to avoid premature decision-making.
- Patients should be informed that NIPT does not replace the need for anomaly ultrasound to assess structural defects.
- Nurses should assess for maternal factors, such as obesity, that may affect fetal fraction and test reliability.
- Providing emotional support is critical for patients awaiting NIPT results, as the test’s high sensitivity may heighten anxiety about potential abnormalities.
Diagnostic Tests
Diagnostic tests in antepartum care are invasive procedures that provide definitive confirmation of fetal conditions, such as chromosomal abnormalities, genetic disorders, or physiological impairments. Unlike screening tests, which estimate risk, diagnostic tests analyze fetal cells, amniotic fluid, or blood to yield precise results. These procedures, including chorionic villus sampling (CVS), amniocentesis, cordocentesis, and biophysical profile (BPP), are typically reserved for high-risk pregnancies due to their associated risks. Nurses must understand the indications, procedures, risks, and nursing responsibilities to ensure patient safety and informed decision-making.
1.1 Chorionic Villus Sampling (CVS)
Chorionic villus sampling (CVS) is an invasive diagnostic test performed between 10 and 13 weeks of gestation to analyze fetal DNA for chromosomal abnormalities and genetic disorders. The procedure involves sampling placental villi, which share the fetus’s genetic makeup, to provide early and definitive results.
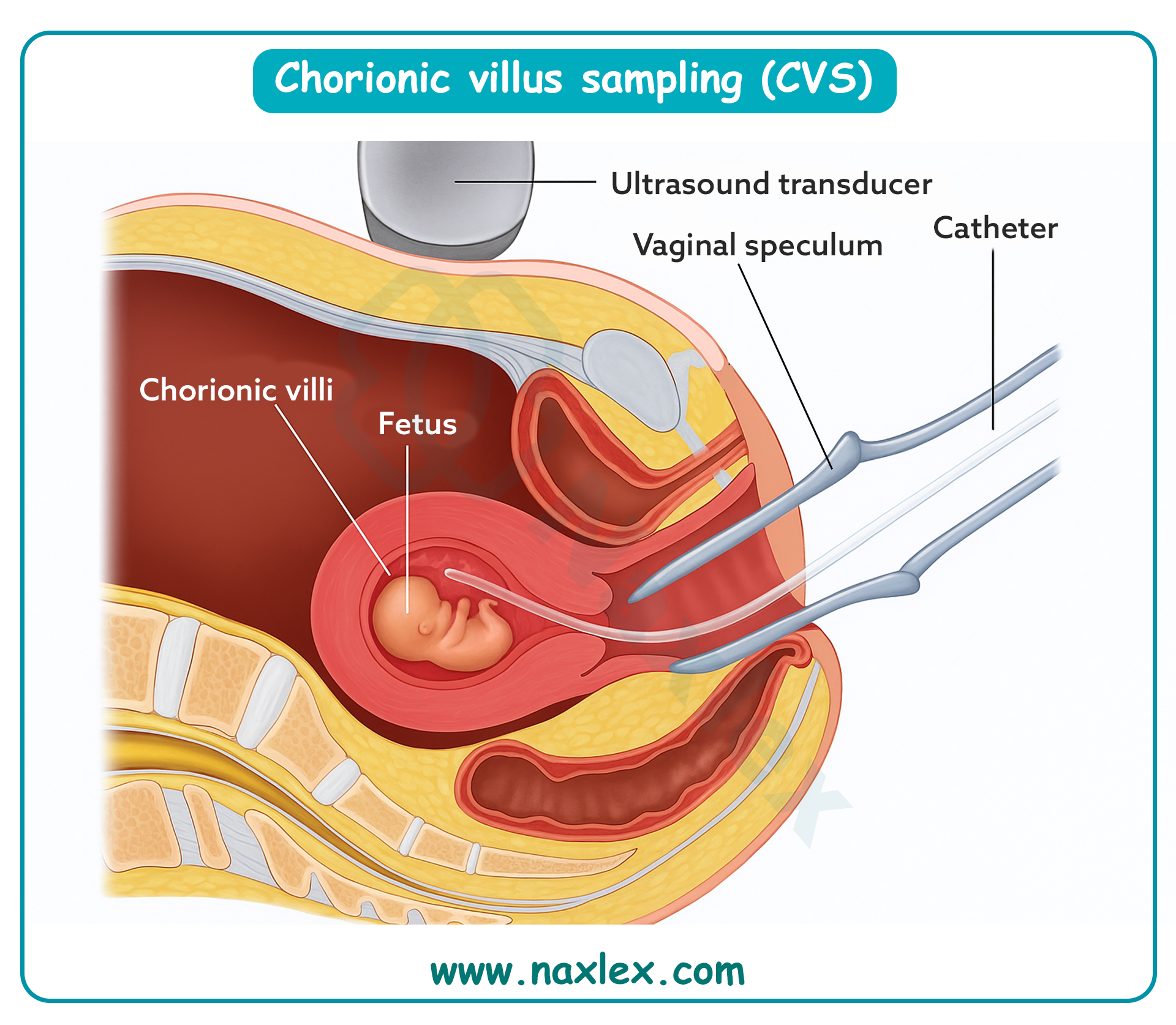
- Procedure:
- CVS is performed either transcervically or transabdominally under ultrasound guidance to ensure accurate sampling.
- In the transcervical approach, a catheter is inserted through the cervix to aspirate villi from the placenta.
- In the transabdominal approach, a needle is inserted through the maternal abdomen into the placenta to collect villi.
- The sampled tissue is analyzed using karyotyping, fluorescence in situ hybridization (FISH), or polymerase chain reaction (PCR) to detect conditions such as Down syndrome, trisomy 18, or cystic fibrosis.
- Indications:
- Abnormal first-trimester screening results (e.g., increased nuchal translucency or abnormal maternal serum markers).
- Family history of genetic disorders or known parental carrier status for conditions like sickle cell disease.
- Advanced maternal age (≥35 years) or previous pregnancy with chromosomal abnormalities.
- Risks and Complications:
- Miscarriage risk of approximately 0.5–1%, slightly higher than amniocentesis due to earlier gestational timing.
- Vaginal bleeding or spotting, particularly with the transcervical approach.
- Infection, including chorioamnionitis, though rare.
- Limb reduction defects, a rare complication if performed before 10 weeks gestation.
- Confined placental mosaicism, where genetic abnormalities are limited to the placenta, may lead to false-positive results.
- Advantages:
- Early diagnosis (first trimester) allows for earlier decision-making compared to amniocentesis.
- Provides definitive genetic information, unlike screening tests.
- Limitations:
- Does not assess neural tube defects, requiring additional testing (e.g., amniocentesis or ultrasound).
- Risk of confined placental mosaicism may necessitate confirmatory amniocentesis.
Nursing Insights:
- Nurses must assess for contraindications, such as active vaginal infections or cervical incompetence, before CVS to minimize complications.
- Patients should be educated about the miscarriage risk and the need for follow-up testing if results indicate mosaicism.
- Post-procedure monitoring includes assessing for vaginal bleeding, cramping, or signs of infection, with immediate reporting to the healthcare provider.
- Nurses should provide emotional support, as the early timing of CVS may heighten patient anxiety about pregnancy outcomes.
1.2 Amniocentesis
Amniocentesis is an invasive diagnostic procedure typically performed between 15 and 20 weeks of gestation to analyze amniotic fluid for fetal chromosomal abnormalities, genetic disorders, and neural tube defects.
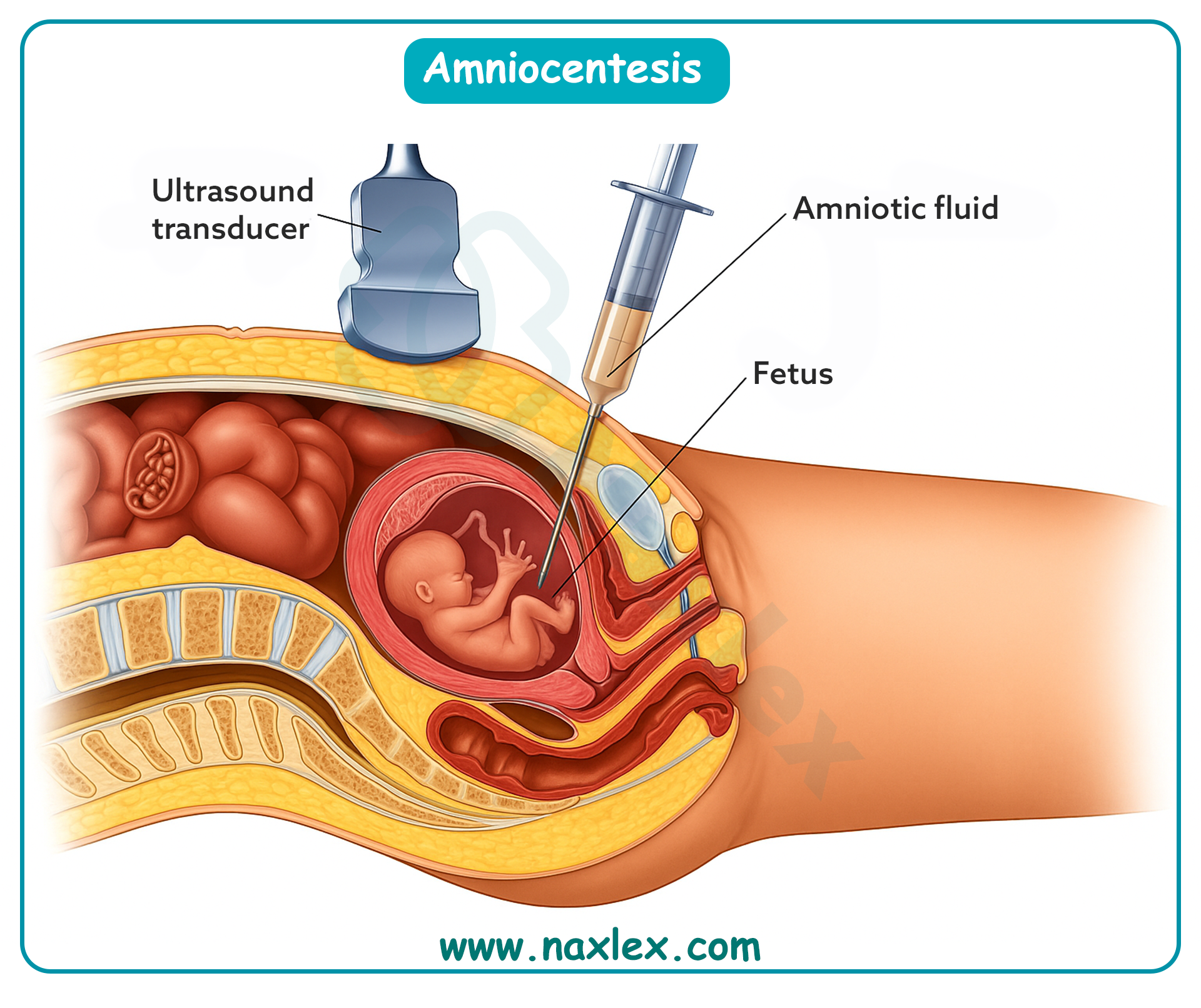
- Procedure:
- A needle is inserted transabdominally under ultrasound guidance to aspirate 15–30 mL of amniotic fluid containing fetal cells and biochemical markers.
- The fluid is analyzed for karyotyping, FISH, or specific gene mutation testing to diagnose conditions such as Down syndrome or spina bifida.
- Amniotic fluid alpha-fetoprotein (AFP) levels are measured to detect neural tube defects.
- Indications:
- Abnormal maternal serum screening results (e.g., elevated AFP or high-risk quadruple screen).
- Advanced maternal age or family history of genetic disorders.
- Ultrasound findings suggestive of structural anomalies requiring genetic confirmation.
- Risks and Complications:
- Miscarriage risk of approximately 0.5–1%, lower than CVS due to later gestational timing.
- Leakage of amniotic fluid, which may lead to oligohydramnios or preterm labor.
- Infection, including chorioamnionitis, though rare with sterile technique.
- Cramping or temporary fetal bradycardia during the procedure.
- Rh sensitization in Rh-negative mothers, necessitating Rh immunoglobulin (RhoGAM) administration.
- Advantages:
- Provides definitive diagnosis for a wide range of conditions, including neural tube defects, unlike CVS.
- Lower miscarriage risk compared to CVS and cordocentesis.
- Limitations:
- Performed later than CVS, delaying diagnosis and decision-making.
- Does not assess fetal physiological status (e.g., anemia), requiring additional tests like cordocentesis.
Nursing Insights:
- Nurses must ensure informed consent is obtained, clearly explaining the procedure’s risks, including miscarriage and infection.
- Patients should be instructed to maintain a full bladder for transabdominal ultrasound guidance during amniocentesis to optimize visualization.
- Post-procedure monitoring includes assessing for fluid leakage, uterine cramping, or fever, with instructions to report these symptoms immediately.
- Nurses should administer RhoGAM to Rh-negative patients post-procedure to prevent Rh isoimmunization.
1.3 Cordocentesis (Percutaneous Umbilical Blood Sampling)
Cordocentesis, also known as percutaneous umbilical blood sampling (PUBS), is an invasive diagnostic procedure performed after 18 weeks of gestation to obtain fetal blood from the umbilical cord for analysis of hematologic, infectious, or genetic conditions.
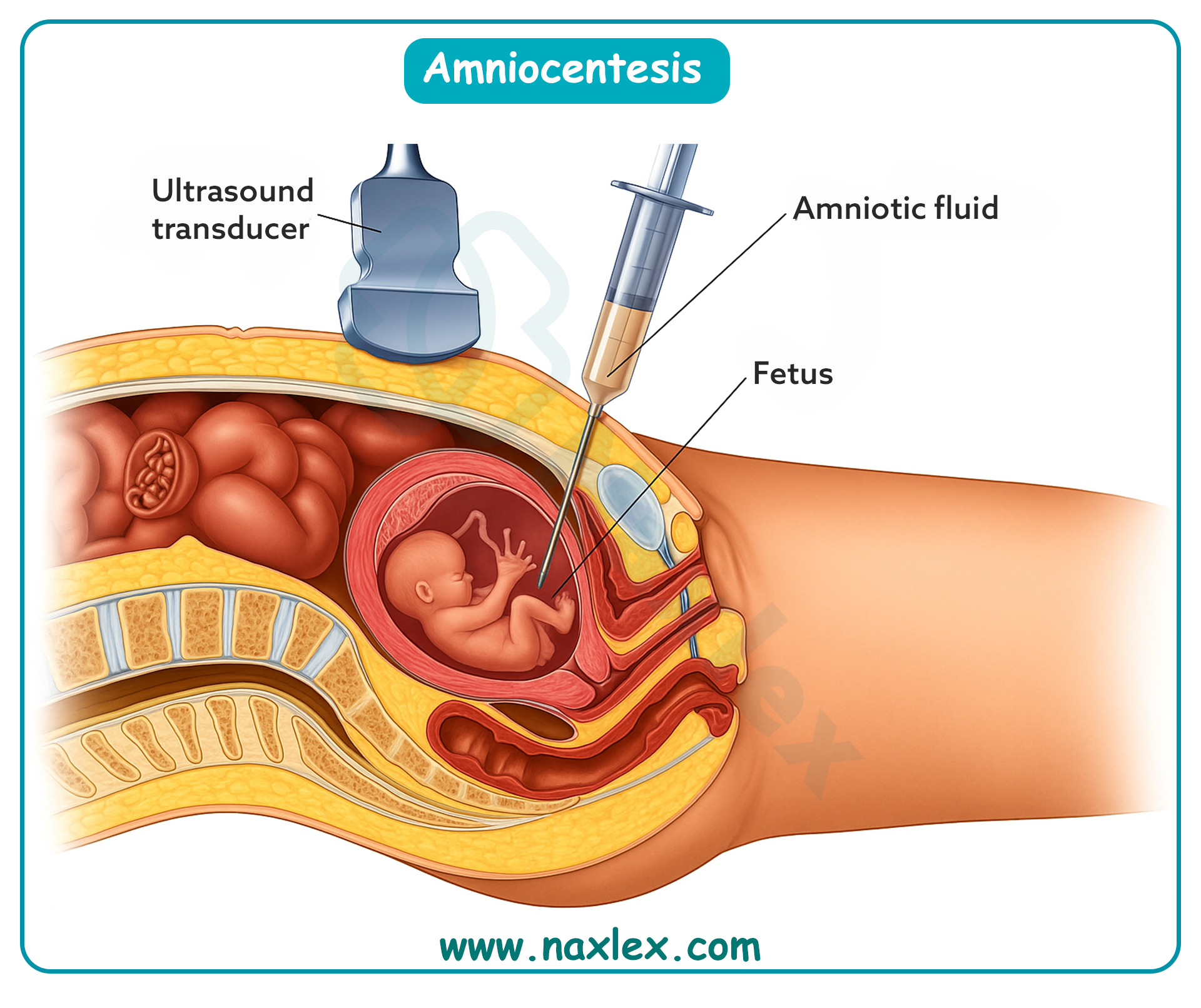
- Procedure:
- A needle is inserted transabdominally under ultrasound guidance into the umbilical vein to aspirate fetal blood.
- The blood sample is analyzed for hemoglobin levels, blood type, infection markers (e.g., toxoplasmosis), or genetic abnormalities.
- Indications:
- Diagnosis of fetal anemia, particularly due to Rh isoimmunization or parvovirus B19 infection.
- Detection of fetal infections or metabolic disorders.
- Confirmation of genetic disorders when CVS or amniocentesis results are inconclusive.
- Risks and Complications:
- Highest miscarriage risk among diagnostic tests (1–2%), due to the procedure’s complexity and later gestational timing.
- Fetal bradycardia or hemorrhage at the puncture site.
- Preterm labor or premature rupture of membranes.
- Maternal-fetal hemorrhage, increasing the risk of Rh sensitization.
- Advantages:
- Provides direct access to fetal blood, enabling diagnosis of conditions not detectable by CVS or amniocentesis (e.g., fetal anemia).
- Rapid results for urgent conditions, such as severe fetal anemia requiring intrauterine transfusion.
- Limitations:
- Highly specialized procedure requiring experienced operators, limiting availability.
- Higher risk profile compared to other diagnostic tests, restricting its use to specific indications.
Nursing Insights:
- Nurses must monitor fetal heart rate during and after cordocentesis to detect bradycardia or distress, reporting abnormalities immediately.
- Patients should be educated about the higher miscarriage risk and the need for post-procedure rest to minimize complications.
- Nurses should assess for signs of preterm labor, such as uterine contractions or vaginal bleeding, post-procedure.
- Coordination with the blood bank is essential for Rh-negative patients to ensure timely RhoGAM administration.
1.4 Biophysical Profile (BPP)
The biophysical profile (BPP) is a non-invasive diagnostic test that combines ultrasound and non-stress testing to assess fetal well-being, typically performed in the third trimester or late second trimester when concerns about fetal health arise.
- Procedure:
- The BPP evaluates five parameters, each scored 0 or 2 (total score out of 10):
- Fetal breathing movements: Presence of at least one episode of rhythmic breathing lasting ≥30 seconds within 30 minutes.
- Fetal movement: At least three discrete body or limb movements within 30 minutes.
- Fetal tone: At least one episode of extension and flexion of a limb or trunk.
- Amniotic fluid volume: A single vertical pocket of amniotic fluid ≥2 cm or an amniotic fluid index (AFI) ≥5 cm.
- Non-stress test (NST): Reactive test with at least two fetal heart rate accelerations (≥15 bpm lasting ≥15 seconds) within 20–40 minutes.
- The ultrasound components are assessed using transabdominal ultrasound, while the NST uses external fetal monitoring.
- The BPP evaluates five parameters, each scored 0 or 2 (total score out of 10):
- Indications:
- Suspected fetal compromise, such as decreased fetal movement or intrauterine growth restriction (IUGR).
- Maternal conditions, such as gestational diabetes or hypertension, that may affect fetal well-being.
- Post-term pregnancy or abnormal amniotic fluid volume (e.g., oligohydramnios or polyhydramnios).
- Scoring and Interpretation:
- A score of 8–10 indicates normal fetal well-being, typically requiring no immediate intervention.
- A score of 6 is equivocal, often prompting repeat testing within 24 hours or further evaluation.
- A score of 0–4 indicates potential fetal compromise, necessitating urgent intervention, such as delivery or additional testing.
- Advantages:
- Non-invasive, posing no direct risk to the mother or fetus.
- Provides comprehensive assessment of fetal health across multiple parameters.
- Limitations:
- Requires skilled sonographers and interpreters for accurate results.
- Results may be influenced by fetal sleep cycles or maternal medications, requiring repeat testing.
Nursing Insights:
- Nurses must prepare patients for the BPP by explaining the procedure and ensuring a comfortable environment for the NST and ultrasound.
- Patients should be instructed to report decreased fetal movement immediately, as this may prompt a BPP to assess fetal well-being.
- Nurses should monitor for maternal discomfort during the NST and assist with positioning to optimize fetal heart rate tracing.
- Abnormal BPP results (score ≤6) require immediate notification of the healthcare provider for further management, such as delivery planning.
Ultrasound Examinations In Antepartum Care
Ultrasound examinations are critical non-invasive tools in antepartum care, utilizing high-frequency sound waves to visualize fetal anatomy, assess placental function, and monitor fetal well-being. These examinations, including the anomaly ultrasound and specialized assessments, provide essential information for detecting structural anomalies and guiding clinical management.
1.1 Anomaly Ultrasound (Level II Ultrasound)
The anomaly ultrasound, also known as a Level II ultrasound, is a detailed ultrasound examination performed between 18 and 22 weeks of gestation to assess fetal anatomical structures and identify potential congenital anomalies.
- Procedure:
- Performed transabdominally using high-resolution ultrasound to visualize fetal organs and structures.
- Key systems evaluated include:
- Central nervous system (e.g., brain ventricles, cerebellum, spinal column for neural tube defects).
- Cardiac system (e.g., four-chamber heart view, outflow tracts).
- Gastrointestinal system (e.g., stomach, bowel).
- Renal system (e.g., kidneys, bladder).
- Skeletal system (e.g., limbs, spine).
- Placental position and amniotic fluid volume (e.g., amniotic fluid index or deepest vertical pocket).
- Indications:
- Routine assessment in all pregnancies to screen for structural anomalies.
- Follow-up for abnormal screening test results, such as elevated AFP or high-risk NIPT.
- Maternal risk factors, such as diabetes or teratogen exposure, increasing the likelihood of congenital anomalies.
- Clinical Significance:
- Detects major structural anomalies, such as spina bifida, congenital heart defects, or cleft lip/palate.
- Identifies “soft markers” (e.g., echogenic intracardiac focus, choroid plexus cysts) that may suggest an increased risk of chromosomal abnormalities, prompting further testing.
- Assesses amniotic fluid volume and placental position to guide pregnancy management.
- Limitations:
- Not all anomalies are detectable, particularly minor defects or those developing later in pregnancy.
- Maternal obesity or fetal positioning may limit visualization, requiring repeat imaging.
- Soft markers may lead to unnecessary anxiety or testing if not interpreted in context.
Nursing Insights:
- Nurses must instruct patients to maintain a full bladder for the anomaly ultrasound to enhance visualization of fetal structures.
- Patients should be educated that the anomaly ultrasound assesses anatomy, not chromosomal status, to clarify its purpose.
- Nurses should provide emotional support for patients receiving abnormal findings, facilitating referrals to genetic counseling or maternal-fetal medicine.
- Documentation of ultrasound findings and patient education is essential for coordinating follow-up care.
1.2 Specialized Ultrasound Assessments
Specialized ultrasound assessments are targeted evaluations performed to address specific clinical concerns, such as fetal growth, amniotic fluid abnormalities, or placental function, often in high-risk pregnancies.
- Types of Specialized Ultrasounds:
- Doppler Ultrasound:
- Measures blood flow in the umbilical artery, middle cerebral artery, or ductus venosus to assess placental function and fetal oxygenation.
- Indicated for suspected intrauterine growth restriction (IUGR) or preeclampsia.
- Abnormal findings, such as absent or reversed end-diastolic flow, suggest fetal compromise requiring urgent intervention.
- Fetal Echocardiography:
- Detailed assessment of the fetal heart to detect congenital heart defects.
- Indicated for family history of heart defects, maternal diabetes, or abnormal findings on anomaly ultrasound.
- Performed by specialized sonographers, typically between 18 and 24 weeks.
- Doppler Ultrasound:
-
- Cervical Length Assessment:
- Measures cervical length transvaginally to assess the risk of preterm labor.
- Indicated for patients with a history of preterm birth or cervical incompetence.
- A cervical length <25 mm before 24 weeks is associated with increased preterm delivery risk.
- Amniotic Fluid Assessment:
- Evaluates amniotic fluid volume using the amniotic fluid index (AFI) or deepest vertical pocket.
- Polyhydramnios (AFI >24 cm) may indicate fetal anomalies (e.g., esophageal atresia), while oligohydramnios (AFI <5 cm) may suggest renal agenesis or IUGR.
- Cervical Length Assessment:
- Clinical Significance:
- Specialized ultrasounds guide management decisions, such as timing of delivery or intrauterine interventions.
- Provide critical data for high-risk pregnancies, improving maternal and fetal outcomes.
- Limitations:
- Require specialized equipment and trained personnel, limiting availability in some settings.
- Interpretation may be affected by maternal or fetal factors, such as obesity or fetal positioning.
Nursing Insights:
- Nurses must prepare patients for specialized ultrasounds by explaining the specific purpose (e.g., Doppler for blood flow, echocardiography for heart defects).
- Patients undergoing transvaginal ultrasound for cervical length should be informed about the procedure’s discomfort and the need for an empty bladder.
- Nurses should monitor patients with abnormal specialized ultrasound findings for signs of distress and coordinate with the healthcare team for timely intervention.
- Emotional support is critical for patients receiving concerning results, such as abnormal Doppler findings, to alleviate anxiety and facilitate coping.
Carrier Screening And Genetic Counseling
Carrier screening and genetic counseling are integral components of antepartum care, aimed at identifying risks for inherited genetic disorders and supporting informed decision-making. Carrier screening assesses parental risk for transmitting genetic conditions, while genetic counseling provides education and guidance to help patients navigate complex testing and reproductive choices. These processes are critical for optimizing maternal and fetal outcomes, particularly in high-risk populations.
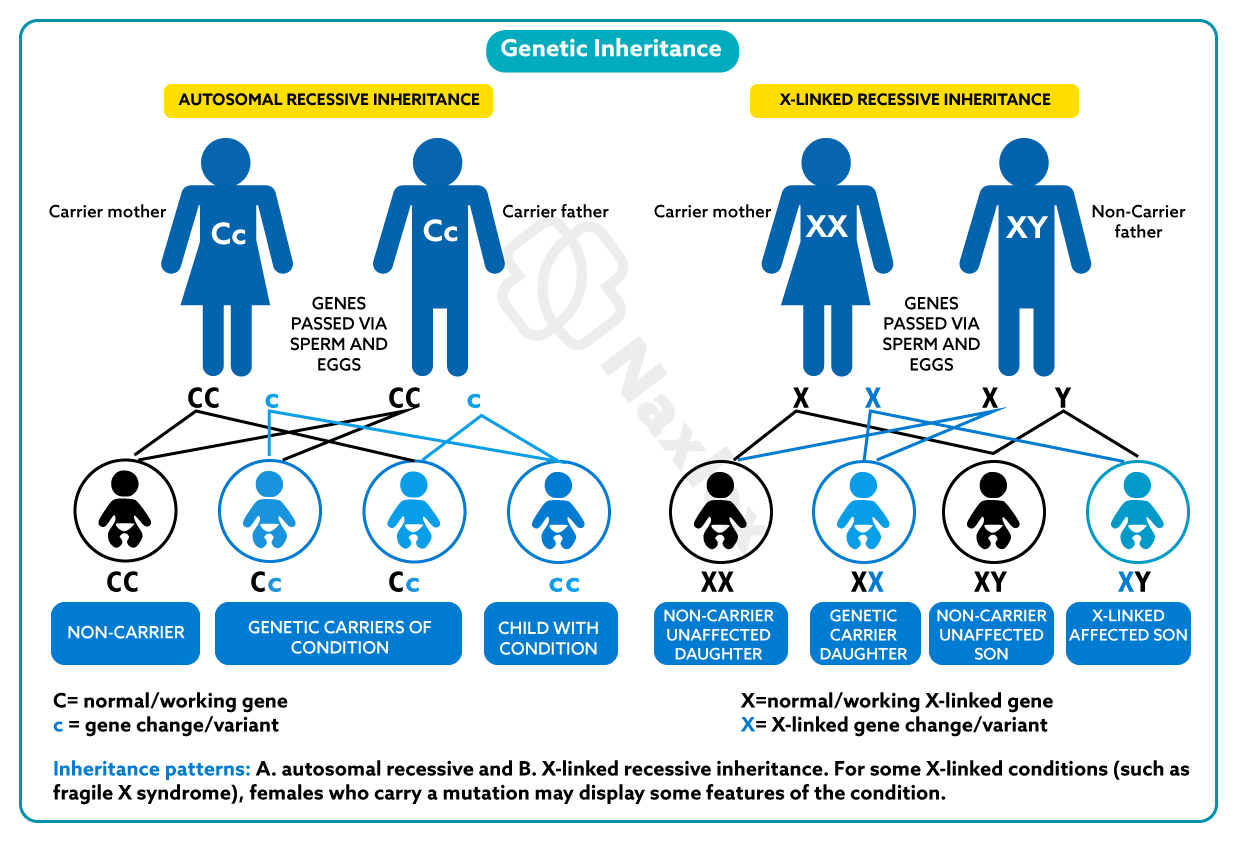
1.1 Principles of Carrier Screening
Carrier screening is a genetic test offered to prospective or expectant parents to identify asymptomatic carriers of recessive or X-linked genetic disorders that could be passed to their offspring. The screening analyzes parental DNA to detect mutations associated with specific conditions, enabling risk assessment for the fetus.
- Purpose and Scope:
- Identifies carriers of autosomal recessive disorders (e.g., cystic fibrosis, sickle cell disease, Tay-Sachs disease) or X-linked disorders (e.g., fragile X syndrome, Duchenne muscular dystrophy).
- Determines the probability of the fetus inheriting a genetic condition, particularly when both parents are carriers (25% risk for autosomal recessive disorders).
- Offered to all pregnant individuals or those planning pregnancy, with expanded panels for high-risk ethnic groups (e.g., Ashkenazi Jewish populations for Tay-Sachs).
- Procedure:
- Involves collecting a maternal and/or paternal blood or saliva sample for DNA analysis via polymerase chain reaction (PCR) or next-generation sequencing.
- Targeted screening focuses on specific disorders based on ethnicity or family history, while expanded carrier screening tests for hundreds of conditions simultaneously.
- Results are reported as carrier status (positive or negative) for specific mutations, with risk calculations if both parents are carriers.
- Indications:
- Family history of a known genetic disorder.
- Ethnic predisposition to certain conditions (e.g., sickle cell anemia in African ancestry, thalassemia in Mediterranean or Asian populations).
- Abnormal prenatal screening results prompting further genetic evaluation.
- Couples planning pregnancy to inform reproductive decisions, such as preimplantation genetic diagnosis.
- Genetic Risk Assessment:
- If both parents are carriers of the same autosomal recessive disorder, there is a 25% chance the fetus will be affected, a 50% chance of being a carrier, and a 25% chance of being unaffected.
- For X-linked disorders, maternal carrier status increases the risk for male offspring (50% chance of being affected) and female offspring (50% chance of being carriers).
- Limitations:
- Carrier screening does not detect all possible mutations for a given disorder, particularly rare variants.
- Negative results reduce but do not eliminate the risk of being a carrier due to limitations in testing sensitivity.
- Expanded carrier screening may identify variants of uncertain significance, requiring genetic counseling for interpretation.
- Ethical Considerations:
- Patients must be informed of the potential for unexpected findings, such as non-paternity or consanguinity.
- The Genetic Information Nondiscrimination Act (GINA) protects against discrimination based on genetic test results in health insurance and employment.
Nursing Insights:
- Nurses must collect a detailed family history to identify candidates for targeted carrier screening based on ethnicity or known genetic disorders.
- Patients should be educated that a negative carrier screening result does not guarantee an unaffected fetus, as some mutations may not be detected.
- Nurses should facilitate referrals to genetic counseling for patients with positive carrier screening results to discuss implications and options.
- Documentation of carrier screening results and patient education is essential for coordinating follow-up care and ensuring informed consent for further testing.
1.2 Role of Genetic Counseling
Genetic counseling is a specialized service provided by trained professionals to educate patients about genetic risks, interpret test results, and support autonomous decision-making regarding prenatal testing and reproductive options. It bridges clinical genetics and patient-centered care, addressing both medical and psychosocial needs.
- Purpose and Process:
- Provides non-directive counseling, presenting unbiased information to empower patients to make informed choices without coercion.
- Interprets complex genetic test results, including carrier screening, NIPT, CVS, or amniocentesis, in the context of the patient’s medical and family history.
- Discusses reproductive options, such as diagnostic testing, preimplantation genetic diagnosis, or pregnancy management, based on test outcomes.
- Components of Genetic Counseling:
- Risk Assessment: Evaluates the likelihood of genetic disorders based on family history, ethnicity, and screening results.
- Education: Explains the nature, inheritance patterns, and implications of genetic conditions in understandable terms.
- Decision Support: Assists patients in weighing the benefits, risks, and limitations of testing or interventions, respecting their values and preferences.
- Psychosocial Support: Addresses emotional responses, such as anxiety or grief, related to genetic findings or reproductive decisions.
- Indications for Referral:
- Positive carrier screening results indicating a risk for an affected fetus.
- Abnormal prenatal screening or diagnostic test results (e.g., high-risk NIPT or ultrasound anomalies).
- Family history of genetic disorders or previous affected pregnancies.
- Advanced maternal age or consanguinity increasing genetic risk.
- Ethical Principles:
- Autonomy: Patients have the right to accept or decline testing or counseling without judgment.
- Non-directiveness: Counselors provide information without influencing decisions, ensuring patient-driven choices.
- Confidentiality: Genetic information is protected under HIPAA and GINA, requiring secure handling of results.
- Cultural Sensitivity:
- Counselors adapt communication to align with patients’ cultural beliefs, which may influence attitudes toward genetic testing or disability.
- Language barriers or family dynamics (e.g., involvement of extended family) are addressed to ensure inclusive decision-making.
- Limitations:
- Genetic counseling cannot eliminate uncertainty, as some test results (e.g., variants of uncertain significance) may lack clear implications.
- Access to counseling may be limited in underserved areas, requiring nurses to advocate for resources.
Nursing Insights:
- Nurses must refer patients to genetic counseling promptly when indicated, ensuring timely access to specialized support.
- Patients should be prepared for genetic counseling by explaining its non-directive nature and the potential for emotional discussions.
- Nurses should assess patients’ understanding of genetic counseling outcomes and provide follow-up support to address questions or concerns.
- Collaboration with genetic counselors ensures continuity of care, particularly for patients pursuing diagnostic testing or reproductive planning.
Nursing Considerations In Antepartum Diagnostic Testing
Nursing considerations in antepartum diagnostic testing encompass a range of responsibilities, including patient education, procedural preparation, emotional support, and post-procedure care. Nurses play a pivotal role in ensuring patient safety, informed decision-making, and holistic care throughout the testing process.
1.1 Patient Education and Informed Consent
Patient education and informed consent are critical to ensuring that pregnant individuals understand the purpose, risks, benefits, and alternatives of antepartum diagnostic tests. Nurses facilitate comprehension and autonomy through clear, evidence-based communication.
- Patient Education:
- Nurses explain the purpose of each test (e.g., screening vs. diagnostic, specific conditions assessed) in lay terms to enhance understanding.
- Education includes procedural details, such as preparation requirements, duration, and expected sensations (e.g., discomfort during CVS).
- Patients are informed about test limitations, such as the inability of NIPT to detect structural anomalies or the risk of false positives in screening tests.
- Informed Consent:
- Informed consent is required for invasive procedures (e.g., CVS, amniocentesis, cordocentesis) due to their associated risks, such as miscarriage.
- Consent discussions include a detailed explanation of risks, benefits, alternatives, and the right to decline testing.
- Nurses verify that patients understand the information and document consent in the medical record.
- Cultural and Linguistic Considerations:
- Education is tailored to patients’ cultural beliefs, addressing potential concerns about testing or disability.
- Interpreters are used for non-English-speaking patients to ensure accurate communication and comprehension.
- Documentation:
- Nurses document all education provided, questions asked, and patient responses to ensure legal and clinical accountability.
- Refusal of testing is documented without judgment, respecting patient autonomy.
Nursing Insights:
- Nurses must use teach-back methods to confirm patient understanding of test purposes and risks before obtaining consent.
- Patients should be provided with written materials in their preferred language to reinforce verbal education.
- Nurses should clarify that screening tests provide risk estimates, not diagnoses, to prevent misinterpretation of results.
- Documentation of informed consent discussions is essential to protect against legal liability and ensure continuity of care.
1.2 Procedural Preparation and Monitoring
Nurses are responsible for preparing patients for antepartum diagnostic tests and monitoring for complications during procedures to ensure safety and optimize outcomes.
- Pre-Procedure Preparation:
- Verify patient identity, procedure type, and gestational age to ensure appropriateness of the test.
- Assess for contraindications, such as active infections or bleeding disorders, particularly for invasive procedures.
- Instruct patients on specific preparations, such as maintaining a full bladder for transabdominal ultrasound or fasting for certain procedures (if required).
- Administer Rh immunoglobulin (RhoGAM) to Rh-negative patients before invasive procedures to prevent Rh sensitization.
- Intra-Procedure Monitoring:
- Monitor maternal vital signs and fetal heart rate during invasive procedures to detect distress or complications.
- Assist with patient positioning to optimize ultrasound visualization and procedural access.
- Provide reassurance and explain sensations (e.g., pressure during amniocentesis) to reduce anxiety during the procedure.
- Equipment and Environment:
- Ensure sterile equipment and a clean environment for invasive procedures to minimize infection risk.
- Coordinate with the ultrasound technologist or physician to ensure all necessary supplies are available.
Nursing Insights:
- Nurses must confirm the patient’s Rh status and administer RhoGAM as needed before invasive procedures to prevent isoimmunization.
- Monitoring fetal heart rate during procedures like cordocentesis is critical to detect bradycardia, requiring immediate intervention.
- Nurses should ensure a calm and supportive environment during procedures to reduce patient stress and enhance cooperation.
- Pre-procedure checklists are used to verify all preparations, reducing the risk of errors or complications.
1.3 Emotional and Psychosocial Support
Antepartum diagnostic testing can evoke significant emotional and psychosocial stress due to the potential for abnormal findings or complex decision-making. Nurses provide holistic support to address these needs.
- Emotional Assessment:
- Assess patients for anxiety, fear, or grief related to testing, particularly after abnormal screening results or invasive procedures.
- Identify patients with a history of pregnancy loss or infertility, as they may experience heightened emotional distress.
- Support Strategies:
- Offer active listening and empathy, validating patients’ feelings without judgment.
- Provide a safe space for patients to express concerns about test outcomes or reproductive decisions.
- Facilitate partner or family involvement in discussions, respecting patient preferences for privacy or support.
- Referral to Resources:
- Refer patients to genetic counselors, social workers, or mental health professionals for additional support.
- Connect patients with support groups for those facing similar diagnoses or decisions.
- Cultural Sensitivity:
- Acknowledge cultural beliefs that may influence attitudes toward testing, disability, or pregnancy termination.
- Use culturally appropriate communication to build trust and ensure patient comfort.
Nursing Insights:
- Nurses must assess patients’ emotional state before and after testing, documenting concerns to guide holistic care.
- Providing anticipatory guidance about potential test outcomes helps prepare patients for difficult decisions.
- Nurses should offer private settings for discussing sensitive results to protect patient confidentiality and emotional well-being.
- Collaboration with interdisciplinary teams, such as social workers, ensures comprehensive support for patients with complex needs.
1.4 Post-Procedure Care and Complication Management
Post-procedure care is essential to monitor for complications, promote recovery, and provide ongoing education following antepartum diagnostic tests, particularly invasive procedures.
- Post-Procedure Monitoring:
- Assess for complications, such as vaginal bleeding, fluid leakage, cramping, or fever, after invasive procedures like CVS or amniocentesis.
- Monitor fetal heart rate and maternal vital signs post-procedure to detect distress or infection.
- Evaluate for signs of preterm labor, such as uterine contractions, particularly after cordocentesis.
- Patient Instructions:
- Instruct patients to rest for 24–48 hours post-procedure and avoid strenuous activity to minimize complication risks.
- Educate patients to report symptoms like fever, severe cramping, or fluid leakage immediately to the healthcare provider.
- Provide written post-procedure instructions, including emergency contact information.
- Complication Management:
- Administer RhoGAM to Rh-negative patients post-procedure to prevent Rh isoimmunization.
- Manage minor complications, such as cramping, with comfort measures (e.g., positioning, hydration) and report severe symptoms promptly.
- Coordinate with the healthcare team for urgent interventions if complications, such as chorioamnionitis or fetal distress, occur.
- Follow-Up Care:
- Schedule follow-up appointments to review test results and discuss implications with the patient.
- Ensure patients understand the need for additional testing or interventions based on results.
Nursing Insights:
- Nurses must monitor for signs of infection or amniotic fluid leakage post-procedure, reporting abnormalities immediately to prevent adverse outcomes.
- Patients should be educated to avoid heavy lifting or sexual activity for a specified period post-procedure to reduce miscarriage risk.
- Nurses should confirm administration of RhoGAM in Rh-negative patients and document it in the medical record.
- Clear post-procedure instructions enhance patient compliance and early detection of complications, improving safety.
Summary
- Antepartum testing encompasses screening and diagnostic procedures to assess fetal health, detect congenital anomalies, and identify maternal conditions affecting pregnancy outcomes.
- Screening tests, such as nuchal translucency ultrasound, maternal serum screening, non-invasive prenatal testing (NIPT), blood type and Rh factor screening, and rubella titer testing, estimate the risk of fetal anomalies without posing significant risk.
- Diagnostic tests, including chorionic villus sampling (CVS), amniocentesis, cordocentesis, and biophysical profile (BPP), provide definitive confirmation of fetal conditions but carry risks like miscarriage or infection.
- Ultrasound examinations, such as the anomaly ultrasound and specialized assessments (e.g., Doppler, fetal echocardiography), evaluate fetal anatomy and physiological function, guiding clinical management.
- Carrier screening identifies parental genetic mutations to assess the risk of inherited disorders, with a 25% chance of an affected fetus if both parents are carriers of an autosomal recessive condition.
- Genetic counseling provides non-directive support, interpreting test results and discussing reproductive options while respecting patient autonomy and cultural beliefs.
- Nursing responsibilities include patient education, obtaining informed consent, preparing for procedures, monitoring for complications, and providing emotional support to reduce anxiety and promote informed decision-making.
- Ethical principles, such as autonomy, beneficence, and non-maleficence, guide antepartum testing, ensuring respect for patient choices and equitable access to care.
- Post-procedure care involves monitoring for complications, such as fluid leakage or preterm labor, and administering RhoGAM to Rh-negative patients to prevent isoimmunization.
- Nurses play a critical role in coordinating interdisciplinary care, advocating for patients, and documenting all aspects of testing to ensure safety and continuity of care.
Naxlex
Videos
Login to View Video
Click here to loginTake Notes on Antepartum Diagnostic Testing
This filled cannot be empty
Join Naxlex Nursing for nursing questions & guides! Sign Up Now

