
Please set your exam date
Infections (TORCH)
Study Questions
Practice Exercise 1
A nurse is educating a pregnant client about preventing toxoplasmosis.
Which of the following instructions should the nurse include?
Explanation
Toxoplasmosis is caused by the obligate intracellular protozoan Toxoplasma gondii, which undergoes its sexual cycle exclusively within feline intestinal tracts. Transmission occurs through ingestion of oocysts found in contaminated soil or feces, or bradyzoites in undercooked meat, potentially leading to congenital infection and fetal neurological sequelae.
Rationale for correct answer
1. Felines are the definitive hosts for the parasite, shedding infectious oocysts in their feces. Pregnant clients must avoid handling cat litter to prevent accidental fecal-oral transmission of the pathogen. This precaution reduces the risk of transplacental infection, which can cause fetal hydrocephalus or chorioretinitis.
Rationale for incorrect answers
2. The MMR vaccine contains live-attenuated viruses for measles, mumps, and rubella, which are contraindicated during pregnancy. Administration of live vaccines poses a theoretical risk of vertical transmission to the developing fetus. Healthcare providers must ensure clients receive these immunizations at least 1 month prior to conception or during the postpartum period.
3. Toxoplasmosis is not transmitted via respiratory droplets, as it is a parasitic infection rather than a viral respiratory pathogen. Airborne or droplet precautions are relevant for diseases like influenza, varicella, or pertussis. Implementing these measures does not mitigate the risk of acquiring Toxoplasma gondii, which requires ingestion of the organism.
4. Mosquito-borne transmission is characteristic of pathogens like Zika virus, malaria, or West Nile virus. Insect repellent is an effective intervention for preventing these specific vector-borne illnesses but is ineffective against toxoplasmosis. Primary prevention for toxoplasmosis focuses on hygiene and dietary practices rather than avoiding hematophagous arthropods.
Test-taking strategy
- Identify the Pathogen: Recognize that toxoplasmosis is a parasitic infection with a specific life cycle involving felines.
- Determine Transmission Route: Evaluate the options based on how the parasite enters the human host, which is primarily via the fecal-oral route or ingestion of contaminated tissue.
- Eliminate Vector and Droplet Options: Rule out choice 3 (droplets) and choice 4 (mosquitoes) as they do not align with the known pathophysiology of Toxoplasma gondii.
- Evaluate Contraindications: Recognize that choice 2 involves a live vaccine, which is a standard contraindication in pregnancy, regardless of the infection being discussed.
- Select the Specific Intervention: Choose choice 1 because it directly addresses the definitive host (cats) and the most common environmental source of oocysts for pregnant women.
Take home points
- Pregnant women should avoid cleaning cat litter boxes or use gloves and wash hands thoroughly if contact is unavoidable.
- Consumption of raw or undercooked meat should be strictly avoided to prevent the ingestion of tissue cysts containing bradyzoites.
- All garden produce must be washed meticulously, and soil contact should be minimized, as oocysts can survive in the environment for months.
- Primary maternal infection during pregnancy can lead to severe fetal complications including intracranial calcifications, microcephaly, and blindness.
A nurse is assessing a newborn whose mother had acute toxoplasmosis during pregnancy.
Which of the following findings should the nurse expect?
Explanation
Congenital toxoplasmosis occurs via transplacental transmission of Toxoplasma gondii tachyzoites, primarily when maternal primary infection occurs during gestation. This parasitic invasion target the central nervous system and eyes, resulting in the classic Sabin triad of clinical manifestations. Intracranial pressure often increases due to obstructive hydrocephalus, and surviving neonates frequently exhibit permanent neurological sequelae including seizures or developmental delays.
Rationale for correct answer
1. The classic presentation of congenital toxoplasmosis consists of the Sabin triad. Infiltrative parasitic growth within the fetal brain causes intracranial calcifications and obstruction of cerebrospinal fluid pathways leading to hydrocephalus. Inflammation of the posterior eye segment results in chorioretinitis, which is the most frequent clinical finding. These specific pathological changes occur because the tachyzoites have a high affinity for neural and ocular tissues.
Rationale for incorrect answers
2. Limb hypoplasia and localized cutaneous scarring are hallmark manifestations of congenital varicella syndrome. This condition occurs following maternal infection with Varicella-Zoster virus, typically between 8 and 20 weeks of gestation. While microcephaly can occur in various TORCH infections, it is not part of the primary diagnostic triad for toxoplasmosis. These integumentary and musculoskeletal defects differentiate varicella from the primarily neuro-ocular presentation of toxoplasmosis.
3. The presence of cataracts, sensorineural deafness, and congenital heart defects, specifically patent ductus arteriosus, defines the Gregg triad. This clinical constellation is pathognomonic for congenital rubella syndrome, which results from maternal infection with the Rubella virus. Unlike toxoplasmosis, rubella frequently causes cardiac malformations and profound hearing loss. These defects are structural and occur during organogenesis in the first trimester.
4. Hepatosplenomegaly, a maculopapular desquamating rash, and snuffles characterize congenital syphilis. Snuffles refers to highly infectious, persistent rhinitis caused by Treponema pallidum invading the nasal mucosa. While both syphilis and toxoplasmosis can cause enlargement of the liver and spleen, the respiratory and specific dermatological findings are unique to syphilis. Syphilis requires immediate treatment with parenteral Penicillin G to prevent late-stage bone and dental deformities.
Test-taking strategy
- Analyze the Question Stem: Focus on the specific pathogen, Toxoplasma gondii, and its impact on the newborn.
- Recall TORCH Syndromes: Differentiate between the classic triads associated with different congenital infections to isolate the correct clinical picture.
- Identify Pathognomonic Signs: Recognize that chorioretinitis and hydrocephalus are uniquely linked as a primary triad in toxoplasmosis.
- Use Process of Elimination:
- Rule out choice 2 by associating limb hypoplasia with Varicella-Zoster.
- Rule out choice 3 by associating cataracts and heart defects with Rubella.
- Rule out choice 4 by associating snuffles and specific rashes with Syphilis.
- Synthesize the Findings: Confirm that choice 1 matches the neuro-ocular focus of the Sabin triad, confirming it as the expected finding for this specific parasitic infection.
Take home points
- Congenital toxoplasmosis is characterized by the classic Sabin triad of chorioretinitis, hydrocephalus, and diffuse intracranial calcifications.
- The severity of fetal infection is inversely proportional to gestational age at the time of maternal transmission, though transmission risk increases later in pregnancy.
- Diagnosis in the neonate is often confirmed through serological testing for IgM antibodies or polymerase chain reaction testing of cerebrospinal fluid.
- This condition must be clinically distinguished from congenital rubella, which presents with cataracts and cardiac defects, and congenital syphilis, which presents with snuffles.
A nurse is caring for a pregnant client diagnosed with acute toxoplasmosis.
Which medication is typically prescribed to reduce placental transmission?
Explanation
Toxoplasmosis pharmacotherapy targets the inhibition of parasitic replication to mitigate vertical transmission and fetal sequelae. Acute maternal infection necessitates macrolide antibiotics to sequester the organism within the maternal compartment and prevent penetration of the placental barrier. If fetal infection is confirmed via amniocentesis, the regimen transitions to synergistic antifolates to target tachyzoite proliferation.
Rationale for correct answer
1. The primary objective in managing acute gestational toxoplasmosis without confirmed fetal infection is preventing vertical transmission using spiramycin. This macrolide antibiotic concentrates within the placenta, creating a protective barrier against the passage of Toxoplasma gondii. It effectively reduces the rate of transmission by approximately 60% if administered immediately following maternal seroconversion.
Rationale for incorrect answers
2. The antiviral agent acyclovir serves as a nucleoside analogue specifically targeting the DNA polymerase of Herpesviridae members. It is the standard of care for maternal herpes simplex virus or varicella-zoster virus infections to prevent neonatal transmission. This medication has no inhibitory effect on the biochemical pathways or the structural integrity of protozoan parasites.
3. The bactericidal action of penicillin G involves the inhibition of cell wall peptidoglycan synthesis through binding to specific proteins. It remains the gold standard for treating maternal Treponema pallidum infections to prevent congenital syphilis and associated skeletal deformities. Because Toxoplasma gondii lacks a bacterial cell wall, this antibiotic provides no therapeutic benefit for parasitic infections.
4. The drug ganciclovir is a potent antiviral primarily utilized for life-threatening or sight-threatening cytomegalovirus infections in immunocompromised individuals. During pregnancy, its use is limited due to potential teratogenicity and significant hematologic toxicity affecting both mother and fetus. It does not possess the pharmacological properties required to treat infections caused by obligate intracellular protozoa.
Test-taking strategy
- Assess the Pathogen: Identify that toxoplasmosis is a parasitic infection, requiring an antimicrobial agent effective against protozoa rather than viruses or bacteria.
- Determine the Clinical Goal: Distinguish between preventing transmission to the fetus (prophylaxis) and treating an already infected fetus (active treatment).
- Evaluate Pharmacological Classes:
- Categorize spiramycin as the specific macrolide used for placental sequestration in toxoplasmosis.
- Identify acyclovir and ganciclovir as antivirals, making them inappropriate for parasitic pathogens.
- Identify penicillin G as a beta-lactam antibiotic, which is ineffective against parasites.
- Select the Standard of Care: Choose the intervention that aligns with international protocols for maternal seroconversion during pregnancy to achieve the best fetal outcomes.
Take home points
- Spiramycin is the drug of choice for preventing the transmission of toxoplasmosis from an acutely infected mother to the fetus.
- If fetal infection is confirmed via PCR of the amniotic fluid, the treatment shifts to pyrimethamine, sulfadiazine, and folinic acid.
- Spiramycin is utilized because it accumulates in high concentrations within placental tissue without crossing into the fetal circulation in therapeutic amounts.
- Healthcare providers must differentiate toxoplasmosis treatment from other TORCH infections like syphilis (penicillin) or herpes (acyclovir).
A nurse is educating a pregnant client on measures to prevent toxoplasmosis.
Which of the following should the nurse include?
Explanation
Toxoplasmosis is a parasitic zoonosis caused by the obligate intracellular coccidian Toxoplasma gondii, which transitions through various life stages including infectious tachyzoites and environmentally resilient oocysts. Maternal infection frequently occurs via the ingestion of contaminated bradyzoites sequestered in striated muscle tissue of livestock or through accidental fecal-oral contact with soil. Acute lymphadenopathy and flu-like symptoms may manifest in the mother, though many cases remain subclinical despite the high risk of vertical transmission.
Rationale for correct answers
1. Thermal processing is essential to denature the proteins and ensure the destruction of tissue cysts containing bradyzoites. Achieving an internal temperature of ≥71 degrees C (160 degrees F) effectively inactivates the parasite within ground meats and poultry. This rigorous temperature control is a primary defense mechanism against foodborne parasitic acquisition during the gestational period.
2. Mechanical friction and surfactants used during hand hygiene physically remove pathogens from the integumentary surface after processing potentially infected animal tissues. Proper sanitation after handling raw meat prevents the accidental transfer of the organism to the oral mucosa. This behavioral intervention is a cornerstone of primary prevention strategies for non-immune pregnant individuals to avoid seroconversion.
5. Oocysts shed by felines can remain viable and infectious in the environment for several months or years. Wearing gloves during gardening or any activity involving soil contact prevents direct exposure to these hardy parasitic stages. Subsequent handwashing further ensures that any pathogens adhering to the skin do not lead to accidental ingestion or infection.
Rationale for incorrect answers
3. Consumption of unpasteurized dairy products significantly increases the risk of acquiring several zoonotic pathogens, including Toxoplasma gondii and Listeria monocytogenes. Pasteurization utilizes heat to eliminate harmful microbes while maintaining the nutritional profile of the milk. Recommending raw milk is dangerous and contraindicated because it bypasses a critical safety step designed to protect the maternal immune system.
4. Sharing utensils with toddlers is a common transmission route for cytomegalovirus (CMV), which is shed in the saliva and urine of young children. While CMV is a member of the TORCH complex, it has a distinct transmission cycle compared to the parasitic nature of toxoplasmosis. Mixing food and saliva does not directly contribute to Toxoplasma transmission unless the food itself is already contaminated with raw cysts.
Test-taking strategy
- Identify the Pathogen: Recognize that toxoplasmosis is a parasitic infection (Toxoplasma gondii) primarily transmitted through contaminated meat, soil, and feline feces.
- Assess the Transmission Routes: Evaluate each option based on whether it addresses the fecal-oral or ingestion pathways associated with the parasite's life cycle.
- Apply Food Safety Principles: Focus on specific temperature requirements (≥71 degrees C) and hygiene practices that neutralize biological threats in the kitchen.
- Differentiate Between TORCH Pathogens: Distinguish between interventions for toxoplasmosis (avoiding raw meat/soil) and those for cytomegalovirus (avoiding toddler saliva) or listeriosis.
- Select Protective Barriers: Prioritize the use of personal protective equipment, such as gloves, for environmental exposures like gardening where feline fecal contamination is likely.
- Eliminate High-Risk Behaviors: Reject any option that encourages the consumption of raw or unpasteurized products, as these are universally contraindicated in pregnancy.
Take home points
- Pregnant clients must ensure all meats are cooked to a safe internal temperature to kill any encysted bradyzoites within the muscle tissue.
- Strict hand hygiene and the use of gloves during gardening are necessary to prevent the ingestion of oocysts found in contaminated soil.
- Avoiding unpasteurized dairy and raw milk is a critical safety measure to prevent both toxoplasmosis and other bacterial infections like listeriosis.
- Toxoplasmosis prevention focuses on food safety and soil contact, whereas cytomegalovirus prevention focuses on avoiding contact with the bodily fluids of young children.
A nurse is planning prenatal teaching for a client at risk for toxoplasmosis.
Which statement indicates effective understanding?
Explanation
Toxoplasmosis is a zoonotic infection caused by the obligate intracellular protozoan Toxoplasma gondii, which utilizes members of the Felidae family as definitive hosts. Transmission occurs via ingestion of oocysts shed in feline feces or bradyzoites found in the muscle tissue of intermediate hosts such as lamb, pork, or beef. Acute infection in pregnant individuals is often asymptomatic but can lead to severe fetal complications including chorioretinitis, intracranial calcifications, and obstructive hydrocephalus.
Rationale for correct answer
1. The definitive host for the parasite is the cat, which sheds infectious oocysts in its feces for a limited duration. Having a partner assume responsibility for cleaning the litter box removes the pregnant client from the direct environment where fecal-oral transmission occurs. This is a highly effective primary prevention strategy that minimizes the risk of maternal seroconversion and subsequent vertical transmission to the developing fetus.
Rationale for incorrect answers
2. Lamb, pork, and venison are high-risk sources for the transmission of bradyzoites contained within tissue cysts. Eating undercooked meat of any kind is strictly contraindicated during pregnancy because the parasite is only inactivated by reaching internal temperatures of ≥71 degrees C (160 degrees F). Consuming raw or rare lamb significantly increases the probability of acquiring an acute infection that can cross the placenta.
3. While wearing gloves provides a physical barrier against oocysts in the soil, it does not eliminate the need for handwashing. Microscopic tears in gloves or accidental contamination of the skin during removal can leave infectious agents on the hands. Diligent hand hygiene following gardening remains a mandatory precaution to ensure that any pathogens present are mechanically removed before they can be ingested.
4. Sharing food utensils with a toddler is a significant risk factor for the transmission of cytomegalovirus (CMV), not toxoplasmosis. CMV is frequently shed in the saliva and urine of young children and can cause severe congenital hearing loss and developmental delays. Although toxoplasmosis is not spread this way, the practice is unsafe in pregnancy due to the risk of other TORCH infections that utilize salivary transmission.
Test-taking strategy
- Identify the Core Pathogen: Recognize that toxoplasmosis involves the Toxoplasma gondii parasite and its specific life cycle involving cats and raw meat.
- Evaluate Behavioral Interventions:
- Assess choice 1 as a delegation strategy that completely avoids the source of infection (fecal-oral via cat feces).
- Assess choice 2 as a high-risk behavior; "occasionally" or "low risk" labels are distractors that do not change the pathophysiological danger of raw meat.
- Apply Hygiene Principles: Rule out choice 3 because the use of personal protective equipment (gloves) never replaces the requirement for sanitation (handwashing) in a clinical or safety context.
- Differentiate Pathogens: Use the "toddler/saliva" clue in choice 4 to identify it as a risk for CMV, thereby eliminating it as a correct answer for toxoplasmosis.
- Select the Safest Statement: Choose the option that demonstrates a proactive and complete avoidance of a known environmental reservoir.
Take home points
- Prevention of toxoplasmosis requires the avoidance of direct contact with cat feces, ideally by having someone else clean the litter box daily.
- Internal cooking temperatures for meat must reach at least 71 degrees C (160 degrees F) to ensure the total destruction of parasitic tissue cysts.
- Gloves used during gardening must be supplemented by immediate handwashing to prevent the accidental ingestion of oocysts found in soil.
- Clinical differentiation is required to distinguish toxoplasmosis risks (meat/soil/cats) from cytomegalovirus risks (toddler bodily fluids).
Practice Exercise 2
A nurse is assessing a pregnant client exposed to rubella during her first trimester. Which of the following is the priority concern for the nurse?
Explanation
Congenital rubella syndrome arises from the transplacental transmission of the rubella virus, a member of the Togaviridae family, during the period of embryonic organogenesis. Maternal infection during the first trimester results in a transmission rate of approximately 80% to 90%, leading to widespread teratogenic effects on the developing fetus. The virus disrupts normal cellular mitosis and induces vascular damage, resulting in permanent structural malformations known as the Gregg triad, which necessitates urgent clinical prioritization over maternal symptomatology.
Rationale for correct answer
2. The primary concern during a first-trimester exposure is the risk of profound fetal structural damage. Maternal viremia during the initial 12 weeks of gestation often leads to the Gregg triad, consisting of sensorineural deafness, cataracts, and congenital heart disease, specifically patent ductus arteriosus. Because these anomalies are irreversible and life-altering, assessing for fetal viability and developmental integrity is the highest clinical priority.
Rationale for incorrect answers
1. Maternal fever and a maculopapular rash are classic clinical manifestations of acute rubella infection in the adult host. While these findings confirm maternal illness, they are typically self-limiting and do not pose a direct threat to the mother's life. In the context of pregnancy, the physiological impact on the mother is secondary to the catastrophic developmental risks faced by the embryo.
3. Arthralgia and posterior cervical lymphadenopathy are common systemic responses to the rubella virus, particularly in female clients. These symptoms indicate the immune system's inflammatory response but do not require emergency intervention or stabilization. Prioritizing these symptoms would neglect the critical assessment of potential vertical transmission and subsequent congenital syndromes.
4. Reviewing the client's immunization history is an essential component of a thorough obstetrical assessment to determine susceptibility. However, once an exposure has already occurred in the first trimester, the history is retrospective data rather than an immediate clinical concern. The priority shifts from prevention to the management of the current gestational risk and potential fetal outcomes.
5. Mild conjunctivitis and headache are prodromal symptoms of rubella that often precede the characteristic exanthem. These findings are considered minor discomforts and do not carry the same clinical weight as the risk of permanent fetal disability. Focus remains on the pathogenic potential of the virus to cross the placenta and cause multi-organ damage.
Test-taking strategy
- Identify the Gestational Timing: Recognize that the "first trimester" is the most critical period for teratogenesis due to active organogenesis.
- Determine Priority: Use the principle of risk reduction to identify which outcome has the greatest long-term impact; fetal anomalies are more severe than transient maternal illness.
- Apply the TORCH Framework: Recall that rubella is a major component of the TORCH complex, where the primary concern is always congenital malformations rather than maternal symptoms.
- Differentiate Symptoms: Distinguish between "expected" maternal symptoms (rash, fever) and "critical" complications (fetal defects).
- Select the Fetal Focus: In maternal-newborn questions where the mother's life is not in immediate danger, the viability and health of the fetus often become the priority concern.
Take home points
- Rubella infection during the first trimester carries the highest risk for severe congenital anomalies, collectively known as congenital rubella syndrome.
- The classic triad of congenital rubella syndrome includes sensorineural hearing loss, ocular defects like cataracts, and cardiac malformations.
- Live-attenuated vaccines such as the MMR are contraindicated during pregnancy due to the theoretical risk of transmission to the fetus.
- Maternal symptoms like arthralgia and rash are generally mild and self-limiting, whereas fetal effects are permanent and often devastating.
A nurse is reviewing the prenatal laboratory results of a client. The results indicate rubella IgG negative and IgM positive. Which of the following actions should the nurse anticipate?
Explanation
The presence of immunoglobulin M (IgM) antibodies in a pregnant client is indicative of an acute or very recent primary infection, as these large pentameric molecules are the first to appear in the humoral immune response. Conversely, immunoglobulin G (IgG) negative status confirms that the client lacked pre-existing immunity through either prior infection or vaccination. This serological profile during pregnancy signifies a high risk for viremia and subsequent transplacental passage of the virus, which can cause severe teratogenic effects, especially if the infection occurs during the first or second trimester.
Rationale for correct answer
2. A positive IgM result combined with a negative IgG result confirms an acute infection, necessitating immediate notification of the primary healthcare provider. The provider must initiate specialized fetal monitoring, which may include high-resolution ultrasound to detect structural anomalies or amniocentesis for viral DNA detection. Early identification of maternal seroconversion is critical for discussing prognosis and coordinating multidisciplinary care for potential congenital rubella syndrome.
Rationale for incorrect answers
1. The client is not immune because her IgG titer is negative; immunity is defined by the presence of long-term memory cells and circulating IgG antibodies. A positive IgM without IgG indicates a current, vulnerable state rather than protection. Providing reassurance of immunity would be factually incorrect and dangerously delay the medical evaluation of a primary infection.
3. Administering the MMR vaccine during pregnancy is strictly contraindicated because it contains live-attenuated viruses. There is a theoretical risk that the vaccine strain could cross the placenta and cause congenital infection in the fetus. Immunization must be deferred until the postpartum period, and the client should be advised to avoid pregnancy for at least 28 days following the injection.
4. Scheduling a follow-up in 3 months is inappropriate because the window for detecting fetal damage and making time-sensitive obstetrical decisions would be lost. Acute rubella infection requires immediate diagnostic workup to determine the timing of the infection and the severity of fetal risk. Delaying care for several months constitutes a failure to manage a high-risk serological finding.
5. There is currently no effective antiviral therapy approved to treat acute rubella infection or to prevent its transmission to the fetus. Management is primarily supportive for the mother and focused on surveillance for the fetus. Unlike other viral infections like herpes simplex, rubella does not respond to drugs like acyclovir, making the initiation of such therapy non-evidence-based.
Test-taking strategy
- Analyze Serology Results: Recognize that IgM signifies "Miserable" (current infection) and IgG signifies "Gone" (past infection or immunity).
- Identify the Priority: In the context of an acute infection with a known teratogen (rubella), the priority is always notification of the provider and assessment of the fetus.
- Rule out Contraindications: Immediately eliminate any option suggesting the MMR vaccine during pregnancy, as live vaccines are a major safety "never event" in gestation.
- Apply the Stability Principle: An acute infection in a pregnant client is an unstable and urgent condition that precludes routine or delayed follow-up (ruling out choice 4).
- Scientific Rationale: Confirm that choice 2 is the only action that addresses the immediate pathophysiological threat to the fetus through appropriate medical escalation.
Take home points
- IgM positive and IgG negative status indicates a primary, acute rubella infection and high risk for vertical transmission.
- Immediate notification of the healthcare provider is required to begin diagnostic testing for fetal congenital rubella syndrome.
- The MMR vaccine must never be given during pregnancy; it is administered postpartum to non-immune mothers before discharge.
- Congenital rubella syndrome involves a constellation of defects including cardiac, ocular, and auditory impairments that require early detection.
A nurse is teaching a group of non-immune women of reproductive age about rubella prevention. Which of the following strategies should be included? Select all that apply
Explanation
Rubella is a viral exanthem characterized by a maculopapular rash and retroauricular lymphadenopathy caused by a Togaviridae agent. Prevention relies on achieving herd immunity through the administration of a live-attenuated vaccine to non-immune individuals. Primary infection during gestation results in severe embryopathy, including cardiac malformations and sensorineural deafness, making preconception screening for IgG titers essential.
Rationale for correct answers
2. The vaccine contains a live-attenuated virus that theoretically poses a risk of vertical transmission to the fetus. Clinical guidelines mandate that women avoid pregnancy for at least 28 days following the administration of the MMR injection. This safety window allows the viral load from the vaccine to dissipate, ensuring a safe environment for subsequent embryonic development.
4. Maternal exposure to individuals presenting with a febrile illness or an undiagnosed rash must be strictly avoided by non-immune women. Rubella is highly contagious via respiratory droplets and can be shed before the exanthem appears in the infected host. Reducing direct environmental exposure serves as a secondary protective measure when active immunization has not yet been achieved.
5. The rubella virus is transmitted through nasal secretions and droplets, making respiratory hygiene a fundamental preventative measure. Frequent hand hygiene disrupts the chain of infection by preventing accidental inoculation of the mucous membranes. These standard infection control practices are vital for non-immune individuals during seasonal outbreaks or in community settings where the virus is prevalent.
Rationale for incorrect answers
1. The administration of the MMR vaccine must occur prior to or after pregnancy, never after conception while the client is currently pregnant. Vaccination during pregnancy is avoided due to the theoretical risk of the attenuated virus crossing the placenta. Non-immune women should be identified during the preconception phase or prenatal screening to receive the dose in the immediate postpartum period.
3. The administration of live-attenuated vaccines is strictly contraindicated during any stage of pregnancy, including the first trimester. The live vaccine virus could potentially cross the placenta and infect the developing embryo, causing the very damage it is intended to prevent. Women who are found to be non-immune during prenatal screening must wait until the postpartum period to receive the immunization.
Test-taking strategy
- Analyze the Question Type: This is a "Select all that apply" (SATA) question focusing on prevention and safety for a non-immune reproductive-age population.
- Evaluate the Vaccine Type: Recognize that the MMR is a live-attenuated vaccine. This fundamental fact dictates its contraindications and the required waiting period for conception.
- Identify Timing:
- Preconception is the optimal time for live vaccines.
- Post-conception or during gestation (choices 1 and 3) is a high-risk period where live vaccines are banned.
- Apply Patient Safety Rules: The 28-day rule (choice 2) is a standard pharmacological precaution to protect a potential fetus from the attenuated virus.
- Consider General Prophylaxis: Evaluate choices 4 and 5 based on transmission routes (droplets and direct contact). Standard precautions are always appropriate for respiratory viruses.
- Final Review: Ensure all selected choices align with the goal of protecting the client and a potential future fetus without violating contraindications in pregnancy.
Take home points
- Preconception vaccination with the MMR vaccine is the gold standard for preventing congenital rubella syndrome in non-immune women.
- A minimum interval of 28 days must be maintained between MMR vaccination and conception to prevent theoretical live-virus exposure to the embryo.
- Live vaccines are never administered during pregnancy; non-immune clients receive the vaccine in the postpartum period before hospital discharge.
- Effective prevention also includes avoiding contact with potentially infected individuals and maintaining meticulous hand and respiratory hygiene.
A nurse is planning discharge teaching for a mother whose newborn has congenital rubella syndrome. Which of the following should the nurse include?
Explanation
Congenital rubella syndrome arises from transplacental transmission of the rubella virus, leading to persistent cellular infection. Affected neonates manifest high viral loads in nasopharyngeal secretions and urine, maintaining infectivity for extended durations. This systemic infection causes diffuse vascular endothelial damage and significant structural malformations, particularly involving the cardiac and auditory systems. Chronic shedding necessitates prolonged isolation to prevent community transmission to susceptible populations.
Rationale for correct answer
1. Neonates with congenital rubella syndrome function as a significant viral reservoir, shedding the pathogen through respiratory secretions and urine for 12 months or longer. Caregivers must adhere to strict infection control measures, including meticulous hand hygiene and contact precautions, to prevent horizontal transmission to non-immune individuals. This prolonged infectivity requires social distancing from pregnant women to mitigate the risk of further congenital outbreaks within the community.
Rationale for incorrect answers
2. While the newborn possesses the virus, the presence of congenital defects necessitates lifelong multidisciplinary monitoring rather than a cessation of care. These infants are at increased risk for late-onset manifestations such as endocrinopathies, progressive hearing loss, and neurodevelopmental delays. Dismissing the need for further surveillance would result in failure to manage the chronic complications associated with the persistent viral infection.
3. Administering the MMR vaccine to an infant with congenital rubella is redundant and clinically inappropriate as a preventative measure for transmission. The vaccine is a live-attenuated preparation and is not indicated for neonates, who typically receive their first dose between 12 and 15 months of age. Furthermore, vaccination does not halt the active shedding of the wild-type virus already present in the infant's biological systems.
4. Routine newborn screenings, including metabolic and hearing assessments, must be performed immediately rather than delayed for 6 months. Early detection of sensorineural hearing loss and cardiac defects is critical for providing timely surgical or rehabilitative interventions. A delay in screening would postpone essential treatments, potentially exacerbating the long-term disabilities and developmental deficits inherent in the syndrome.
Test-taking strategy
- Identify the Pathophysiology: Recognize that congenital rubella is a persistent viral infection, not a transient one, distinguishing it from adult rubella.
- Prioritize Public Health: Focus on infection control and the risk of transmission to others, which is a hallmark of managing neonates with this condition.
- Analyze Viral Shedding: Recall that the incubation and shedding period for congenital cases is uniquely long, lasting up to 1 year or until consecutive negative cultures are obtained.
- Eliminate Delay Tactics: Rule out choice 4, as medical care for a high-risk neonate should always be proactive and early rather than delayed.
- Evaluate Clinical Logic: Rule out choice 2 by recognizing that a syndrome with permanent structural defects requires continuous follow-up, and choice 3 by understanding vaccine protocols.
- Select the Safety Option: Choose choice 1 because it addresses the epidemiological risk posed by the patient to the general population.
Take home points
- Infants with congenital rubella syndrome are considered contagious and can shed the virus in body fluids for up to 1 year.
- Infection control measures must be maintained around pregnant women or non-immune individuals to prevent further congenital cases.
- Long-term monitoring is essential to address potential complications like diabetes mellitus, thyroid dysfunction, and progressive sensorineural deafness.
- Routine screenings and early interventions should never be delayed, as these infants require immediate assessment of cardiac and ocular health.
A nurse is providing education to a client about the fetal risks of rubella infection during pregnancy. Which of the following should the nurse include? Select all that apply
Explanation
Congenital rubella syndrome occurs when the rubella virus traverses the placenta, infecting fetal tissues during critical periods of organogenesis. The pathogen induces mitotic arrest and chromosomal breakage, resulting in structural defects across multiple organ systems, most notably the ocular, auditory, and cardiovascular systems. This constellation of anomalies is termed the Gregg triad, and the risk of severe teratogenic outcomes is highest (up to 90%) when maternal infection occurs before 12 weeks of gestation.
Rationale for correct answers
1. Sensorineural hearing loss is the most frequent clinical manifestation of congenital rubella syndrome, occurring in approximately 60 to 80 percent of infected neonates. The virus causes direct damage to the cochlea and the organ of Corti during embryonic development. This deficit may be bilateral or unilateral and often requires early intervention with hearing aids or cochlear implants to support linguistic development.
2. The virus interferes with the normal development of the lens fibers, leading to the formation of congenital cataracts. These ocular lesions are often described as "pearlescent" and can result in significant visual impairment or blindness if not surgically addressed. Ocular involvement is a hallmark of the classic rubella triad and serves as a primary diagnostic indicator in the neonatal period.
3. Cardiac malformations result from viral-induced necrotizing arteritis and impaired development of the great vessels during the first trimester. Patent ductus arteriosus and peripheral pulmonary artery stenosis are the pathognomonic cardiovascular defects associated with this infection. These hemodynamic abnormalities often require surgical ligation or balloon angioplasty to prevent progressive heart failure and pulmonary hypertension.
5. The rubella virus is highly neurotropic, leading to inhibited neuronal proliferation and resulting in microcephaly. This reduction in brain volume is frequently associated with long-term developmental delays, intellectual disabilities, and motor deficits. Neurological assessment is a critical component of the longitudinal care plan for children surviving the initial neonatal phase of the infection.
Rationale for incorrect answers
4. While the rubella virus can cause systemic fetal involvement, hydrops fetalis is not the most common or characteristic complication of this specific infection. Hydrops fetalis, involving severe fetal edema and heart failure, is more classically associated with Parvovirus B19 (Fifth disease) or Rh isoimmunization. In rubella, the primary focus is on permanent structural malformations rather than the acute hydropic changes seen in other viral pathologies.
Test-taking strategy
- Identify the Core Condition: Recognize that the question focuses on the specific teratogenic effects of the rubella virus as part of the TORCH complex.
- Recall the Gregg Triad: Use the acronym "Ear, Eye, Heart" to quickly identify choices 1, 2, and 3 as classic, high-probability findings in congenital rubella.
- Assess Gestational Timing: Note that rubella is primarily a disease of organogenesis (first trimester), leading to the structural defects listed in choices 1, 2, 3, and 5.
- Differentiate Viral Complications: Distinguish rubella from Parvovirus B19; remember that Parvovirus targets erythroid progenitor cells leading to anemia and hydrops, while rubella causes structural anomalies.
- Select the Multi-system Options: Choose all findings that reflect the systemic and permanent nature of the embryopathy, excluding choice 4 which describes a different clinical phenomenon.
Take home points
- Sensorineural deafness is the most common single finding in children with congenital rubella syndrome.
- The classic triad includes cataracts, sensorineural deafness, and cardiac defects (most commonly patent ductus arteriosus).
- Microcephaly and intellectual disabilities are common neurological sequelae resulting from viral interference with brain growth.
- Prevention through preconception vaccination remains the primary strategy to eliminate the risk of these irreversible fetal anomalies.
Practice Exercise 3
A nurse is educating a pregnant client about CMV infection. Which of the following statements by the client indicates correct understanding?
Explanation
Cytomegalovirus is a ubiquitous ubiquitous DNA virus belonging to the Herpesviridae family that establishes lifelong latency after primary infection. Transmission occurs via direct contact with infectious bodily fluids, most commonly saliva or urine from young children who shed the virus asymptomatically. Maternal primary infection or reactivation during gestation poses a significant risk for vertical transmission, leading to permanent sensorineural hearing loss or neurodevelopmental deficits. High-risk behaviors include sharing utensils or improper sanitation after handling pediatric excretions.
Rationale for correct answer
2. Since the virus is shed in high titers in pediatric urine and saliva, strict hand hygiene after handling diapers or oral secretions is the most effective preventative measure. This behavioral intervention physically removes viral particles from the skin, preventing accidental autoinoculation of maternal mucous membranes. Mitigating fecal-oral or fluid-to-mucosa contact significantly reduces the probability of primary maternal seroconversion. Consistent adherence to these sanitary practices is critical for pregnant women who provide childcare.
Rationale for incorrect answers
1. Complete avoidance of all contact with young children is a disproportionate and impractical recommendation that is not supported by current clinical guidelines. Prevention focuses on modifying high-risk hygiene behaviors rather than total social isolation from the pediatric population. Pregnant clients can safely interact with children if they utilize proper precautions, such as avoiding the sharing of food or kissing the child on the mouth.
3. The MMR vaccine provides immunity against measles, mumps, and rubella, but it confers no cross-protection against cytomegalovirus. Furthermore, the MMR is a live-attenuated vaccine and is strictly contraindicated during pregnancy due to the risk of fetal infection. There is currently no commercially available vaccine approved for the prevention of cytomegalovirus, making behavioral modification the primary defensive strategy.
4. Maternal cytomegalovirus infection is frequently asymptomatic or presents with non-specific, mild flu-like symptoms, yet it still poses a grave risk to the fetus. The absence of maternal clinical illness does not preclude the possibility of transplacental transmission or subsequent neonatal sequelae. Relying solely on the presence of symptoms would lead to a failure in identifying and managing the majority of at-risk pregnancies.
Test-taking strategy
- Identify the Pathogen: Recognize that cytomegalovirus (CMV) is a herpesvirus spread through bodily fluids, particularly those of young children.
- Assess Transmission Dynamics: Focus on the most common route, which is the saliva and urine of toddlers, often found in daycare or home settings.
- Evaluate Clinical Feasibility: Rule out choice 1 because "avoiding all contact" is an extreme, non-therapeutic intervention that is rarely the correct answer in nursing care.
- Differentiate Immunizations: Recall that CMV has no vaccine; therefore, any choice involving the MMR or other vaccines for CMV prevention is scientifically inaccurate.
- Apply Pathophysiological Knowledge: Understand that TORCH infections, especially CMV, are often subclinical in the mother, making choice 4 a dangerous misconception.
- Select the Standard Precaution: Choose the hygiene-based option (choice 2) as it directly addresses the mechanism of fluid-borne transmission.
Take home points
- Cytomegalovirus is the most common congenital viral infection and a leading cause of non-genetic sensorineural hearing loss in children.
- Primary prevention during pregnancy focuses on meticulous handwashing after contact with a child's urine, saliva, or contaminated toys and surfaces.
- Pregnant women should be advised not to share food, drinks, or utensils with young children to minimize exposure to infectious saliva.
- The risk of vertical transmission exists regardless of whether the maternal infection is primary, a reactivation, or a reinfection with a different strain.
A nurse is caring for a newborn diagnosed with congenital CMV. Which clinical manifestation should the nurse prioritize monitoring?
Explanation
Congenital cytomegalovirus results from maternal viraemia and subsequent transplacental infection, leading to systemic viral replication within fetal tissues. The pathogen exhibits high neurotropism, frequently causing inflammatory damage to the eighth cranial nerve and cerebral parenchyma. Clinical manifestations include periventricular calcifications, microcephaly, and hepatosplenomegaly, though 90% of affected neonates appear asymptomatic at birth. Permanent auditory deficits and neurodevelopmental delays represent the most significant long-term sequelae of this intrauterine infection.
Rationale for correct answer
3. Cytomegalovirus is the leading non-genetic cause of pediatric sensorineural hearing loss in developed nations. The virus causes direct damage to the structures of the inner ear, specifically the cochlea, often leading to progressive auditory impairment. Because this deficit can be late-onset or fluctuate, continuous monitoring is the priority to facilitate early intervention. Timely auditory rehabilitation is essential to optimize the child's linguistic and cognitive development.
Rationale for incorrect answers
1. Petechiae, often described as a blueberry muffin rash, and jaundice are common acute findings in symptomatic congenital cytomegalovirus. While these signs indicate systemic involvement and hepatic dysfunction, they are typically self-limiting or manageable in the neonatal period. Auditory deficits carry a higher risk of permanent, life-altering disability compared to these transient cutaneous and metabolic manifestations. Monitoring for hearing loss remains the priority for long-term functional outcomes.
2. Limb hypoplasia is not a characteristic clinical finding associated with congenital cytomegalovirus infection. This specific musculoskeletal deformity is a hallmark of congenital varicella syndrome, which occurs following maternal infection with the Varicella-Zoster virus. CMV pathology focuses on neurological, ocular, and visceral organs rather than disrupting the development of the limb buds. Therefore, monitoring for limb defects would be inappropriate for a neonate diagnosed with a CMV infection.
4. Structural congenital heart defects, such as patent ductus arteriosus or septal defects, are pathognomonic for congenital rubella syndrome. Cytomegalovirus does not typically cause primary cardiovascular malformations during embryonic organogenesis. While a neonate with CMV may have various comorbidities, heart defects are not a standard feature of the CMV clinical profile. Priority monitoring focuses on the neurological and sensory systems most frequently targeted by the cytomegalovirus.
Test-taking strategy
- Analyze the Pathogen: Identify that the question specifies cytomegalovirus (CMV), a member of the TORCH complex with specific organ affinities.
- Recall Pathognomonic Findings: Distinguish CMV from other TORCH infections by associating it with sensorineural hearing loss and periventricular calcifications.
- Differentiate by Exclusion:
- Rule out choice 2 (limb hypoplasia) as it belongs to Varicella.
- Rule out choice 4 (heart defects) as it belongs to Rubella.
- Prioritize Long-term Impact: Between choice 1 (transient) and choice 3 (permanent), prioritize the condition that significantly impacts developmental milestones.
- Focus on Prevalence: Recognize that hearing loss is the most common permanent sequela of congenital CMV, making it the most critical monitoring priority for the nurse.
Take home points
- Sensorineural hearing loss is the most frequent and significant long-term complication of congenital cytomegalovirus infection.
- Neonates with CMV require serial auditory evaluations because hearing loss can be progressive or delayed in onset beyond the newborn period.
- CMV is distinguished from other TORCH infections by the presence of periventricular calcifications rather than the diffuse calcifications seen in toxoplasmosis.
- While blueberry muffin spots and jaundice are visible signs, they do not carry the same long-term neurosensory priority as auditory monitoring.
A nurse is teaching a group of pregnant clients about preventing congenital CMV. Which of the following measures should the nurse include? Select all that apply
Explanation
Cytomegalovirus is a common herpesvirus that establishes lifelong latency and is characterized by periodic subclinical viral shedding. Maternal transmission typically occurs through direct mucosal contact with infectious bodily fluids, particularly the saliva and urine of asymptomatic toddlers. The virus is highly neurotropic, making it the leading non-genetic cause of congenital sensorineural hearing loss and a significant contributor to neurodevelopmental disabilities. Primary prevention relies entirely on behavioral modifications, as there are no pharmacological or immunogenic interventions currently approved for general prophylaxis.
Rationale for correct answers
1. Young children are the primary reservoir for the virus, often shedding it in high concentrations in their saliva for months. Avoiding the sharing of food, drinks, or utensils with toddlers prevents the direct transfer of viral particles to the maternal oral mucosa. This simple behavioral change is a cornerstone of preventing primary maternal infection and subsequent vertical transmission to the fetus during any stage of gestation.
2. Since the virus is excreted in high titers in pediatric urine, diaper changes represent a significant exposure risk for pregnant caregivers. Meticulous hand hygiene following any contact with urine, saliva, or nasal secretions physically removes the virus before autoinoculation can occur. Consistent use of soap and water after handling toys or cleaning up after toddlers significantly lowers the risk of acquiring the infection in household or daycare settings.
Rationale for incorrect answers
3. There is currently no commercially available or FDA-approved CMV vaccine for the prevention of congenital infection. While research into recombinant and mRNA vaccines is ongoing, clinical practice still relies solely on hygiene education to protect pregnant clients. Suggesting that a vaccine is an available strategy is scientifically inaccurate and provides a false sense of security to the client regarding their immune status.
4. Avoiding contact with cats and their litter boxes is the primary prevention strategy for toxoplasmosis, not cytomegalovirus. Toxoplasmosis is caused by a parasite (Toxoplasma gondii) found in feline feces, whereas CMV is a human herpesvirus spread through human bodily fluids. While both are members of the TORCH complex, the interventions for preventing them are distinct and should not be confused in patient education.
5. Limiting contact with toddlers in daycare is generally impractical and unnecessary if proper sanitary precautions are strictly maintained. Nursing education should focus on safe interaction techniques, such as avoiding kissing children on the mouth and practicing frequent handwashing, rather than social isolation. Recommending the avoidance of a specific demographic is not a standard or effective public health strategy for CMV prevention in pregnant women.
Test-taking strategy
- Identify the Pathogen: Recognize that cytomegalovirus (CMV) is transmitted through human bodily fluids (saliva, urine), not animals or food.
- Target the High-Risk Population: Focus on the relationship between pregnant women and young children, who are the most common source of infection.
- Evaluate Vaccine Availability: Recall that among the TORCH infections, rubella and varicella have vaccines, but CMV does not.
- Differentiate Precautions:
- Rule out choice 4 by associating cats with Toxoplasmosis.
- Rule out choice 5 by distinguishing between "avoiding contact" (incorrect) and "modifying behavior" (correct).
- Select Hygiene Measures: Choose the options that describe specific, actionable hygiene steps (choices 1 and 2) to interrupt the fluid-to-mucosa transmission route.
Take home points
- The most effective way to prevent congenital CMV is to avoid contact with the saliva and urine of young children.
- Pregnant women should be specifically instructed not to share food, cups, or utensils with toddlers and to avoid kissing them on the mouth.
- Frequent handwashing is mandatory after changing diapers, wiping noses, or handling children's toys that may be contaminated with saliva.
- There is no vaccine for CMV; therefore, prenatal education must emphasize behavioral changes as the primary defense against maternal seroconversion.
A nurse is reviewing prenatal laboratory results and notes that a client has positive CMV IgM and low IgG avidity. Which of the following is the most appropriate interpretation?
Explanation
Cytomegalovirus serology interpretation relies on the detection of specific immunoglobulins and the functional strength of their binding. The presence of IgM antibodies indicates a recent immune response, but it can occasionally persist or reappear during reactivation. To differentiate a first-time exposure, an avidity index is calculated; low results suggest that the IgG antibodies are recently produced and have not yet undergone the maturation process required for high-affinity binding to the viral antigens. High avidity (typically >60%) generally excludes a primary infection occurring within the preceding 3 to 4 months.
Rationale for correct answer
2. The combination of a positive IgM and low IgG avidity is the definitive serological marker for a primary infection occurring in the very recent past. Low avidity indicates that the immune system has not had sufficient time (usually less than 16 to 20 weeks) to produce mature, high-affinity antibodies. In a pregnant client, this profile signifies the highest risk period for vertical transmission and severe fetal sequelae, as the maternal immune system is encountering the virus for the first time.
Rationale for incorrect answers
1. Past infection with established immunity would be characterized by a negative IgM and a positive IgG with high avidity. High avidity demonstrates that the antibodies have matured over several months, indicating a latent infection state. While a woman with past infection can still transmit the virus to a fetus via reactivation, the statistical risk and typical severity are significantly lower than during a primary infection.
3. A client with no evidence of exposure would be seronegative for both IgM and IgG antibodies. This status indicates the client is non-immune and highly susceptible to a primary infection if exposed to infectious bodily fluids. If the lab results show any positive titers or avidity values, the "no exposure" interpretation is scientifically invalidated as the immune system has clearly interacted with the viral antigens.
4. Reactivation of a latent virus usually presents with a positive IgG and high avidity, although IgM may occasionally become positive again. However, the concept of "no fetal risk" is medically inaccurate, as even non-primary infections (reactivations or reinfections with new strains) can lead to congenital CMV. Furthermore, low avidity is specifically not associated with reactivation; it is a hallmark of the initial, primary phase of the disease process.
Test-taking strategy
- Analyze the Biomarkers: Focus on the specific meaning of IgM (acute) and the avidity index (duration of infection).
- Define Avidity: Recall that "low avidity" means "weak binding," which only occurs in the early stages of antibody production following a primary exposure.
- Determine Fetal Risk: Recognize that the primary infection (low avidity) carries a transmission risk of 30% to 40%, whereas secondary infection risk is approximately 1% to 2%.
- Process of Elimination:
- Rule out choice 3 because the results are positive, not negative.
- Rule out choice 1 because low avidity contradicts long-term immunity.
- Rule out choice 4 because low avidity is not a feature of reactivation.
- Select the High-Urgency Option: Choose choice 2 as it represents the most clinically significant and accurate interpretation of the data.
Take home points
- Low IgG avidity is the most reliable indicator that a primary CMV infection occurred within the last 3 to 4 months.
- Maternal primary CMV infection during pregnancy carries a significantly higher risk of symptomatic congenital disease compared to reactivation.
- IgM can remain positive for months after an initial infection, making the avidity test essential for accurate gestational dating of the exposure.
Confirmed primary infection in pregnancy warrants immediate referral for high-level fetal surveillance, including amniocentesis for viral PCR.
A nurse is caring for a neonate with symptomatic congenital CMV. Which intervention should the nurse anticipate?
Explanation
Congenital cytomegalovirus is an obligate intracellular viral infection that requires potent antiviral agents to inhibit DNA polymerase and reduce viral replication. Treatment is indicated for neonates with central nervous system involvement or multi-organ disease to mitigate the severity of sensorineural hearing loss and improve neurodevelopmental outcomes. Therapy must be initiated within the first 30 days of life to be effective, typically requiring prolonged administration and close monitoring for neutropenia and other hematologic toxicities.
Rationale for correct answer
2. The antiviral agent ganciclovir, or its prodrug valganciclovir, is the standard of care for symptomatic congenital CMV. It works by competitively inhibiting viral DNA synthesis, which limits the progressive damage to the auditory and central nervous systems. Clinical studies demonstrate that a 6 month course of treatment can significantly prevent the worsening of hearing deficits and improve long-term cognitive scores in affected infants.
Rationale for incorrect answers
1. Amoxicillin is a beta-lactam antibiotic that targets bacterial cell wall synthesis and is entirely ineffective against viral pathogens. Using antibiotics to treat a viral infection like CMV represents a misapplication of antimicrobial therapy and does not address the underlying pathophysiology. While secondary bacterial infections can occur, amoxicillin has no role in the primary management of congenital viral syndromes.
3. High-dose vitamin C supplementation lacks clinical evidence as an effective treatment for congenital viral infections. While vitamin C is an antioxidant, it cannot inhibit the replication of the cytomegalovirus or prevent the structural and neurological damage caused by the virus. Relying on vitamins for a severe neonatal infection would delay the administration of life-altering antiviral medications.
4. Providing phototherapy only addresses neonatal jaundice by converting bilirubin into water-soluble isomers for excretion. While jaundice is a common finding in CMV due to hepatitis, treating it in isolation ignores the critical neurological and auditory risks of the disease. A narrow focus on hyperbilirubinemia constitutes a failure to provide comprehensive care for the multi-system sequelae of the virus.
Test-taking strategy
- Analyze the Pathogen: Identify CMV as a DNA virus, which necessitates the use of a specific antiviral class rather than antibiotics or supplements.
- Assess Clinical Severity: Recognize that the question specifies "symptomatic" CMV, which is the specific trigger for intensive pharmacological intervention.
- Differentiate Drug Classes:
- Rule out choice 1; antibiotics are for bacteria, not viruses.
- Rule out choice 3; "supplements" are rarely the priority intervention for acute congenital diseases.
- Evaluate the Scope of Care: Rule out choice 4 because it is an "only" statement that addresses a minor symptom while neglecting the organ-threatening viral load.
- Identify the Gold Standard: Select ganciclovir as the specific, evidence-based antiviral used in neonatal intensive care for this condition.
Take home points
- Intravenous ganciclovir or oral valganciclovir is the primary treatment for neonates with symptomatic congenital CMV.
- Antiviral therapy is specifically aimed at preserving hearing and improving neurological development rather than just treating skin or liver symptoms.
- Treatment must be closely monitored for bone marrow suppression, specifically absolute neutrophil counts falling below 500 cells/mm3.
- Early initiation of therapy, ideally within the first month of life, is essential to achieve maximum therapeutic benefit for the infant.
Practice Exercise 4
A nurse is assessing a client who is at 39 weeks gestation and in active labor. The client has a documented history of genital Herpes Simplex Virus (HSV). Which of the following findings would require the nurse to prepare the client for a cesarean delivery?
Explanation
Genital herpes simplex virus infection during pregnancy necessitates rigorous clinical surveillance to prevent neonatal transmission, which occurs primarily during descent through an infected birth canal. The virus establishes latency within the sacral ganglia and can undergo reactivation, presenting as either visible vesicular lesions or subjective prodromal symptoms. If active replication is suspected at the onset of labor, the risk of vertical transmission increases significantly, potentially leading to disseminated encephalitis or multi-organ failure in the newborn.
Rationale for correct answer
1. The report of a tingling sensation or pruritus in the perineal area constitutes prodromal symptoms, which indicate that viral shedding is imminent or already occurring. Even in the absence of visible vesicles, these sensations suggest active reactivation of the virus along the neural pathways. Because the risk of neonatal transmission is high during active shedding, the presence of prodromal symptoms is a mandatory indication for cesarean delivery to bypass the infected birth canal.
Rationale for incorrect answers
2. While failure to adhere to the prophylactic acyclovir regimen increases the statistical probability of an outbreak, the missed doses alone do not mandate a surgical delivery. A cesarean is only indicated if the client presents with active lesions or prodromal symptoms at the time of labor. The nurse should perform a thorough physical assessment, but in the absence of clinical signs, a vaginal delivery remains the preferred and safe route.
3. The rupture of membranes with clear fluid is a normal physiological occurrence in active labor and does not indicate an HSV-related complication. If the fluid were meconium-stained or if the client had active lesions, the clinical priority would shift; however, 2 hours of rupture does not necessitate a cesarean in a stable client. The duration of membrane rupture is only a concern in HSV if active lesions are present, as the risk of ascending infection increases over time.
4. A history of a primary outbreak 5 years ago indicates that the client has recurrent HSV rather than a primary infection, but it is not a reason for surgical intervention today. The presence of maternal antibodies from a long-standing infection actually provides some passive protection to the fetus, lowering the transmission risk compared to a primary maternal infection. Only current clinical evidence of viral activity at 39 weeks would alter the planned route of delivery.
Test-taking strategy
- Identify the Decision Point: In HSV-positive pregnant clients, the decision for a cesarean is based entirely on the current clinical presentation at the time of labor.
- Recognize Subjective Data: Understand that prodromal symptoms (tingling, burning, itching) are treated with the same clinical gravity as visible, fluid-filled vesicles.
- Evaluate Risk of Transmission: Prioritize the intervention that prevents the neonate from coming into contact with active viral shedding in the vaginal vault.
- Eliminate Historical Data: Rule out choice 4, as a past history does not dictate current delivery mode; only the "here and now" assessment matters.
- Assess Medication Adherence: Recognize that while acyclovir prophylaxis (usually started at 36 weeks) is standard, a missed dose (choice 2) is not a surgical indication unless a lesion has actually formed.
- Select the Safety First Option: Choose choice 1 because it represents the earliest stage of an outbreak, making a vaginal delivery unsafe for the newborn.
Take home points
- Cesarean delivery is mandatory for pregnant clients in labor who have active genital herpes lesions or prodromal symptoms like tingling.
- Prophylactic antiviral therapy with acyclovir or valacyclovir is typically initiated at 36 weeks gestation to reduce the likelihood of an outbreak at term.
- If a client with HSV has no active lesions or prodromes during labor, a vaginal delivery is considered safe and is the recommended approach.
- Neonatal herpes is a life-threatening condition; therefore, any suspicion of viral shedding at the time of delivery must be managed surgically to protect the infant.
A nurse is caring for a client at 36 weeks gestation with a history of recurrent genital herpes. Which of the following instructions should the nurse include in the teaching plan?
Explanation
Genital herpes simplex virus infection during pregnancy requires a strategic pharmacological approach to minimize the risk of vertical transmission. Management focuses on preventing the reactivation of the virus during the third trimester, as the presence of pathogenic shedding at the time of labor necessitates surgical intervention. The virus maintains a state of latency within the dorsal root ganglia, but hormonal shifts and physical stress can trigger an outbreak, which is why prophylactic suppressive therapy is the standard of care to ensure the possibility of a safe vaginal delivery.
Rationale for correct answer
2. Clinical guidelines from major obstetric organizations recommend initiating suppressive antiviral therapy at 36 weeks gestation for all women with a history of recurrent genital herpes. Drugs such as acyclovir or valacyclovir effectively inhibit viral DNA polymerase, thereby reducing the frequency of symptomatic outbreaks and asymptomatic shedding at term. Maintaining a daily medication regimen until delivery significantly increases the likelihood that the client will be asymptomatic during labor, allowing for a vaginal birth and reducing the need for a cesarean section.
Rationale for incorrect answers
1. A scheduled cesarean section is not routinely required for clients with a history of HSV if they are asymptomatic at the onset of labor. Surgical delivery is reserved specifically for those presenting with active vesicular lesions or prodromal symptoms, such as burning or tingling, in the genital tract. Automatically scheduling a cesarean at 38 weeks without clinical evidence of an outbreak exposes the client to unnecessary surgical risks and contradicts evidence-based practice for managing recurrent HSV.
3. Frequent vaginal exams every 2 hours during labor are contraindicated because they increase the risk of introducing pathogens and may cause unnecessary trauma to the maternal tissues. In the context of HSV, exams should be limited to the minimum number necessary to assess cervical effacement and dilation. Excessive manipulation of the birth canal could potentially facilitate the transfer of the virus to the fetus if subclinical shedding is occurring, though the primary assessment for lesions is performed only once upon admission.
4. The use of internal fetal scalp electrodes is strictly avoided in clients with a history of genital herpes unless absolutely necessary and in the absence of lesions. Any procedure that breaks the fetal integumentary barrier provides a direct portal of entry for the virus, significantly increasing the risk of neonatal herpes simplex infection. External monitoring is the preferred method for assessing fetal heart rate patterns to maintain the integrity of the infant's skin and prevent iatrogenic viral inoculation.
Test-taking strategy
- Identify the Goal of Care: The primary objective for a pregnant client with recurrent HSV is to prevent an active outbreak at the time of delivery.
- Determine the Standard Protocol: Recall that 36 weeks is the established gestational milestone for starting suppressive therapy (acyclovir/valacyclovir).
- Evaluate Delivery Mode: Understand that a history of HSV does not equal an automatic cesarean; the route of delivery is determined by the physical assessment during labor.
- Apply Infection Control Principles:
- Rule out choice 4; internal monitors are invasive and increase infection risk.
- Rule out choice 3; frequent exams are generally discouraged in labor to prevent ascending infection.
- Select the Proactive Measure: Choose choice 2 as it is the only option that actively works to prevent the complication (an outbreak) rather than just reacting to it or using high-risk procedures.
Take home points
- Suppressive antiviral therapy is initiated at 36 weeks gestation to reduce the risk of active HSV lesions at the time of labor.
- Cesarean delivery is only indicated if active genital lesions or prodromal symptoms are present when labor begins or membranes rupture.
- Invasive fetal monitoring, such as scalp electrodes, should be avoided in HSV-positive clients to prevent direct viral inoculation into the fetus.
- Acyclovir and valacyclovir are considered safe for use in the third trimester and are the primary agents used for viral suppression.
A nurse is assessing a 4-day-old newborn client for suspected neonatal Herpes Simplex Virus (HSV). Which of the following clinical manifestations should the nurse identify as indicative of this infection? Select all that apply
Explanation
Neonatal herpes simplex virus is a life-threatening infection typically acquired during the peripartum period via contact with infectious maternal secretions. The virus exhibits strong neurotropic and organotropic properties, leading to three distinct clinical classifications: skin, eye, and mucous membrane (SEM) disease; central nervous system (CNS) disease; and disseminated disease. Without prompt administration of parenteral acyclovir, the infection can rapidly progress to irreversible neurological destruction, hepatic failure, and coagulopathy, often manifesting within the first 4 to 21 days of life.
Rationale for correct answers
1. Clustered vesicular lesions are the hallmark of SEM disease and often appear at the site of initial inoculation, such as the scalp in cases involving fetal electrodes or vacuum extraction. These vesicles contain high viral titers and can quickly spread or progress to more severe systemic involvement. Identifying these cutaneous markers is critical for early diagnosis, as they are present in approximately 60 to 70 percent of affected neonates.
2. The sudden onset of seizure activity or extreme irritability indicates that the virus has bypassed the blood-brain barrier, causing acute encephalitis. CNS involvement often presents with lethargy, tremors, and a bulging fontanelle as intracranial pressure increases due to inflammatory cerebral edema. These neurological signs represent a medical emergency and require immediate cerebrospinal fluid analysis and high-dose antiviral therapy to minimize permanent brain damage.
3. A temperature of 38.2 degrees C (100.8 degrees F) combined with poor feeding are non-specific but critical signs of neonatal sepsis, which can be the primary presentation of HSV. Neonates are unable to localize infections effectively, so systemic symptoms often precede focal findings. Poor feeding and thermoregulation instability reflect the metabolic stress of the underlying viremia and possible early multiorgan dysfunction.
5. Generalized petechiae and purpura are indicative of the disseminated form of neonatal herpes, which carries the highest mortality rate. This presentation reflects a state of disseminated intravascular coagulation (DIC) and fulminant hepatitis caused by widespread viral replication in visceral organs. These hematologic abnormalities signal a critical failure of the hemostatic system and require aggressive intensive care and blood product support.
Rationale for incorrect answers
4. Pink-tinged crystals in the diaper, often referred to as uric acid crystals, are a common and benign finding in newborns during the first week of life. They indicate concentrated urine as the infant's fluid intake transitions and do not signify a pathological process or infection. This finding is unrelated to the pathophysiology of the herpes simplex virus and should not be confused with the hemorrhagic manifestations of disseminated disease.
Test-taking strategy
- Identify the Pathogen: Recognize that neonatal HSV is a systemic and potentially neurological disease requiring high-acuity assessment.
- Categorize the Symptoms: Use the SEM, CNS, and Disseminated framework to validate choices 1, 2, and 5.
- Prioritize Life-Threatening Signs:
- Associate vesicles (choice 1) with the virus's primary cutaneous presentation.
- Associate seizures (choice 2) with the hallmark CNS involvement.
- Associate fever (choice 3) with the global inflammatory response seen in neonatal infections.
- Differentiate Normal vs. Abnormal: Distinguish choice 4 (uric acid crystals) as a physiological newborn variant that occurs independently of infectious states.
- Apply Hematologic Logic: Recognize that petechiae (choice 5) represent the severe coagulopathy seen in the disseminated form of the infection.
- Final Selection: Ensure the chosen options reflect the systemic and multi-organ impact of a viral pathogen rather than normal transition findings.
Take home points
- Neonatal HSV is classified into SEM, CNS, and disseminated disease, each requiring urgent clinical recognition.
- Vesicular rashes and neurological changes like seizures are pathognomonic and mandate immediate parenteral acyclovir therapy.
- Disseminated HSV mimics bacterial sepsis but is often characterized by severe liver dysfunction and coagulopathy.
- Uric acid crystals in a diaper are a normal finding related to hydration and are not a sign of congenital or neonatal infection.
A nurse is teaching a postpartum client who has an active orolabial herpes lesion (cold sore) about caring for her newborn. Which of the following statements by the client indicates a correct understanding?
Explanation
Orolabial herpes, caused by Herpes Simplex Virus Type 1 (HSV-1), involves viral replication within the epithelial cells and subsequent shedding from the oral mucosa or vermilion border. For a neonate, exposure to this virus is a medical emergency due to their immature immune system, which allows for rapid viral dissemination to the central nervous system or visceral organs. Maternal shedding can occur even when a lesion is crusting, making rigorous barrier precautions and hand hygiene essential to prevent horizontal transmission during the vulnerable neonatal period.
Rationale for correct answer
2. The use of a surgical mask serves as a critical physical barrier that prevents accidental direct contact between the infant and the maternal oral lesion. It also mitigates the risk of droplet transmission or autoinoculation if the mother were to touch her lip and then the infant. This intervention allows the mother to maintain the essential bonding process and continue breastfeeding safely while the lesion is in an infectious state.
Rationale for incorrect answers
1. The client does not need to stop breastfeeding as long as there are no herpetic lesions present on the breasts. HSV-1 is not transmitted through breast milk itself; rather, transmission occurs through direct contact with active vesicles. The mother should be encouraged to continue lactation to provide the infant with passive immunity, provided she adheres to strict hand hygiene and covers the oral lesion.
3. Applying diaper cream (typically containing zinc oxide) to an oral herpes lesion is not an evidence-based treatment and may lead to secondary contamination of the product. Maternal lesions should be treated with appropriate antiviral creams or oral medications like acyclovir as prescribed by a healthcare provider. Furthermore, using a product intended for the infant on a maternal infection increases the risk of fomite transmission to the newborn.
4. It is absolutely unsafe to kiss a newborn anywhere when an active orolabial lesion is present, regardless of whether the lesion is touched. The virus can be present in saliva and on the surrounding skin through asymptomatic shedding, making any oral contact extremely high-risk. Neonatal herpes acquired through kissing can lead to devastating encephalitis or "kiss of death" syndromes, which carry a high mortality rate.
Test-taking strategy
- Identify the Pathogen and Host: Recognize the high-risk relationship between HSV-1 and a neonate, where even minor exposure can lead to systemic disease.
- Evaluate the Transmission Route: Focus on preventing direct contact between the maternal lesion/saliva and the newborn's skin or mucous membranes.
- Analyze Barrier Precautions: Look for the most effective way to prevent accidental contact; a mask (choice 2) provides a constant physical shield during close contact.
- Assess Breastfeeding Safety: Apply the rule that breastfeeding is only contraindicated in HSV if mammary lesions are present.
- Eliminate High-Risk Behaviors: Rule out any choice that permits kissing (choice 4) or improper medication use (choice 3) as these compromise neonatal safety.
- Select the Practical Intervention: Choose the option that balances maternal-infant bonding with maximum infection control.
Take home points
- Mothers with active cold sores must wear a surgical mask and practice strict handwashing when handling their newborns.
- Kissing the infant is strictly prohibited until the orolabial lesion is completely healed and the crust has fallen off.
- Breastfeeding is safe and encouraged unless active herpetic lesions are identified on the mother's nipples or areolae.
- Neonatal HSV-1 infection can be just as severe as HSV-2, potentially causing permanent neurological damage or death.
The nurse is reviewing the primary health care provider's orders for a newborn with suspected disseminated Herpes Simplex Virus (HSV). Which of the following medications should the nurse expect to administer?
Explanation
Disseminated neonatal herpes simplex virus represents the most severe clinical manifestation of the infection, involving multi-organ failure and a mortality rate exceeding 80% if left untreated. The pathophysiology involves widespread viral replication within the visceral organs, leading to fulminant hepatitis, pneumonitis, and intravascular coagulopathy. Treatment must focus on the immediate inhibition of viral DNA polymerase to halt cellular destruction. Because the neonatal immune system is immature, the pharmacological intervention must be aggressive and delivered via a route that ensures 100% bioavailability and therapeutic plasma concentrations.
Rationale for correct answer
3. Intravenous Acyclovir is the gold standard and mandatory treatment for all forms of neonatal herpes, particularly the disseminated type. Acyclovir is a purine nucleoside analogue that selectively inhibits the replication of HSV-1 and HSV-2 by interfering with the viral DNA chain elongation. High-dose parenteral administration (typically 60 mg/kg/day divided every 8 hours) is required for a minimum of 21 days to ensure the viral load is suppressed across all affected organ systems. Delaying this specific antiviral therapy by even a few hours significantly increases the risk of permanent neurological deficits or death.
Rationale for incorrect answers
1. Penicillin G is a bactericidal antibiotic that acts by inhibiting the synthesis of the bacterial cell wall. It is the treatment of choice for neonatal syphilis but has zero efficacy against viral pathogens like the herpes simplex virus. Administering an antibiotic for a suspected viral viraemia would be a diagnostic and therapeutic error that fails to address the underlying cause of the neonate's clinical deterioration.
2. Spiramycin is a macrolide antibiotic used primarily in the treatment of maternal toxoplasmosis to reduce the risk of vertical transmission to the fetus. It does not possess antiviral properties and is not indicated for the management of the herpes simplex virus. Furthermore, it is generally administered to the mother during pregnancy rather than to the newborn as a primary treatment for acute disseminated viral disease.
4. Varicella-zoster immune globulin (VZIG) provides passive immunity specifically against the varicella-zoster virus (chickenpox). While varicella is also a member of the Herpesviridae family, VZIG does not contain the specific antibodies required to neutralize the herpes simplex virus (HSV-1 or HSV-2). Using VZIG for an HSV infection is an inappropriate use of blood products and would provide no protection against the disseminated viral replication occurring in the infant.
Test-taking strategy
- Identify the Pathogen Class: Recognize that HSV is a virus, which automatically rules out any pharmacological agent ending in "-cillin" or "-mycin" (antibiotics).
- Target the Delivery Route: In cases of "disseminated" or systemic disease in a neonate, always prioritize the intravenous route over oral or intramuscular routes for maximum efficacy.
- Differentiate within the Family: Distinguish between the treatments for different herpes viruses; acyclovir is for HSV, whereas VZIG is specifically for VZV.
- Evaluate Clinical Urgency: Understand that disseminated HSV is a medical emergency; the correct answer must be the most potent, specific, and fast-acting medication available.
- Match Drug to Bug: Connect acyclovir to herpes simplex as the definitive, evidence-based pairing in neonatal intensive care.
Take home points
- Intravenous acyclovir is the only effective treatment for neonatal disseminated herpes and must be started immediately upon clinical suspicion.
- Treatment for disseminated or CNS-involved herpes requires a 21-day course of high-dose parenteral antiviral therapy.
- Antibiotics like Penicillin G and Spiramycin have no role in treating viral infections and are used for Syphilis and Toxoplasmosis, respectively.
- Disseminated HSV often presents similarly to bacterial sepsis, but it is uniquely characterized by severe liver enzyme elevations and thrombocytopenia.
Practice Exercise 5
A nurse is caring for a client at 37 weeks gestation who tested positive for GBS. Which of the following interventions should the nurse implement during labor to prevent early-onset neonatal infection?
Explanation
Group B Streptococcus (Streptococcus agalactiae) is a Gram-positive coccus that colonizes the maternal anogenital tract, posing a risk for vertical transmission during parturition. This pathogen causes early-onset neonatal sepsis, pneumonia, and meningitis through the aspiration of contaminated vaginal fluids. Effective prevention requires therapeutic maternal serum levels of bactericidal agents to eliminate fetal exposure during the intrapartum window.
Rationale for correct answer
1. The administration of intrapartum penicillin G is the gold standard for prophylaxis, requiring an initial 5 million unit dose followed by 2.5 to 3 million units every 4 hours. Optimal efficacy is achieved when the antibiotic is administered for at least 4 hours prior to the expulsion of the fetus. This timeframe ensures adequate umbilical cord blood concentrations to prevent neonatal colonization and subsequent systemic infection.
Rationale for incorrect answers
2. Oral antibiotics are clinically insufficient because they fail to reach the high, rapid intravascular concentrations necessary to neutralize GBS during active labor. The pharmacokinetics of oral administration are too slow and unpredictable to provide the requisite protection against vertical transmission during the second stage of labor. Only parenteral routes are indicated for intrapartum antibiotic prophylaxis to ensure immediate bioavailability.
3. A cesarean section is not routinely indicated for GBS-positive status, as the primary prevention strategy remains pharmacological rather than surgical. While a cesarean performed before the onset of labor and rupture of membranes minimizes transmission risk, it introduces unnecessary maternal surgical morbidity. For clients at 37 weeks or beyond, a vaginal delivery with appropriate antibiotic coverage is the evidence-based standard of care.
4. The application of an antiseptic perineal scrub or vaginal douching has been proven ineffective in reducing the incidence of early-onset GBS disease. These topical methods do not eliminate the bacterial load within the cervical canal or address the risk of ascending infection once membranes have ruptured. Clinical guidelines prioritize systemic antimicrobials over localized cleansing to provide a comprehensive barrier against neonatal infection.
Test-taking strategy
- Identify the Pathogen Class: Recognize GBS as a bacterial colonization that requires specific antibiotic timing to prevent neonatal sepsis.
- Recall the Time-to-Efficacy Rule: Prioritize the 4 hour window as the critical pharmacological benchmark for intrapartum prophylaxis.
- Determine the Route of Choice: Eliminate any option suggesting oral medications (choice 2), as intravenous administration is mandatory for rapid maternal-fetal transfer.
- Assess Delivery Mode: Understand that GBS is not a surgical indication; vaginal birth remains the standard as long as antibiotics are used.
- Evaluate Non-Pharmacological Interventions: Rule out topical solutions (choice 4) as they lack the systemic depth to protect the fetus during the descent through the birth canal.
- Apply Patient Stability Principles: Focus on the intervention that provides the most direct and evidence-based protection for the neonate.
Take home points
- Penicillin G is the first-line agent for GBS prophylaxis, with ampicillin serving as a valid alternative for intrapartum care.
- Adequate prophylaxis is defined as receiving the initial antibiotic dose at least 4 hours before the delivery of the infant.
- Intrapartum antibiotic prophylaxis is only indicated once labor has started or membranes have ruptured in GBS-positive individuals.
- Clients with GBS who undergo a planned cesarean section before labor starts and with intact membranes do not require antibiotic prophylaxis.
A nurse is educating a client about GBS colonization. Which statement by the client indicates correct understanding?
Explanation
Group B Streptococcus (GBS) is a transient commensal bacterium found in the gastrointestinal and genitourinary tracts of 10 to 30 percent of pregnant individuals. While typically asymptomatic in the adult carrier, colonization at the time of delivery facilitates the vertical transmission of the pathogen to the neonate. This can result in early-onset disease, manifesting as respiratory distress, tachycardia, and temperature instability within the first 72 hours of life. Universal screening protocols utilize culture-based methods to identify carriers who require intrapartum antibiotic prophylaxis to protect the fetus.
Rationale for correct answer
2. Current clinical guidelines from major obstetric organizations mandate universal rectovaginal screening for all pregnant clients between 36 0/7 and 37 6/7 weeks of gestation. This specific timing is utilized because the culture results are highly predictive of colonization status for the subsequent 5 weeks, covering the period when most clients go into labor. A swab is used to sample the distal vagina and the anorectum to increase the sensitivity of the test, as the gastrointestinal tract serves as the primary reservoir for GBS.
Rationale for incorrect answers
1. Colonization with GBS is overwhelmingly asymptomatic in the mother and does not typically result in vaginitis or urinary tract symptoms unless the bacterial load is exceptionally high. Most women are entirely unaware of their carrier status until a screening culture is performed during the third trimester. Relying on the presence of symptoms would lead to the failure of identifying the vast majority of colonized individuals, leaving their newborns at significant risk for infection.
3. Starting antibiotics before the onset of labor is ineffective because GBS is a transient colonizer that can quickly return after a course of treatment is completed. Prophylaxis is only indicated during the intrapartum period—when labor begins or membranes rupture—to provide protection during the actual window of fetal exposure. Administering antibiotics during the antepartum phase does not prevent neonatal early-onset disease and contributes to the development of antimicrobial resistance.
4. While colonization is a prerequisite for vertical transmission, the majority of newborns born to GBS-positive mothers will not develop sepsis. Without treatment, approximately 1 to 2 percent of infants born to colonized women develop early-onset disease; with proper intrapartum antibiotic prophylaxis, this risk is reduced by over 80 percent. The development of sepsis is not automatic but rather depends on the bacterial load, the duration of membrane rupture, and the effectiveness of maternal prophylaxis.
Test-taking strategy
- Identify the Standard of Care: Recall that the universal screening window for GBS is 36 to 37 weeks (moving toward 37 weeks in recent updates).
- Define Colonization: Distinguish between "infection" (which has symptoms) and "colonization" (which is asymptomatic), effectively ruling out choice 1.
- Determine Timing of Intervention: Apply the principle that prophylaxis for GBS is intrapartum, not antepartum, which rules out choice 3.
- Assess Risk Probability: Avoid extreme words like "automatically" (choice 4) in nursing questions, as medical outcomes are rarely guaranteed for every patient.
- Confirm the Screening Method: Match the "rectovaginal" technique with the known anatomical reservoirs for GBS to validate choice 2.
- Apply Public Health Principles: Focus on the screening intervention that represents the current evidence-based protocol for preventing neonatal morbidity.
Take home points
- Universal rectovaginal GBS screening is performed between 36 and 37 weeks of gestation to determine the need for intrapartum antibiotics.
- GBS colonization is generally asymptomatic in the mother, making universal screening essential regardless of clinical presentation.
- Antibiotic prophylaxis is only effective when administered during labor or after the rupture of membranes, rather than during the prenatal period.
- While GBS is a major cause of neonatal sepsis, the absolute risk of disease in an infant is low when the mother receives timely intrapartum antibiotics.
A nurse is reviewing risk factors for neonatal GBS infection. Which of the following should the nurse identify? Select all that apply
Explanation
Group B Streptococcus (GBS) is a gram-positive coccus that colonizes the maternal gastrointestinal and genitourinary tracts. Vertical transmission occurs during labor or following membrane rupture, potentially leading to neonatal sepsis, pneumonia, or meningitis. Early-onset disease typically manifests within 72 hours of birth.
Rationale for correct answers
1. Preterm delivery at <37 weeks increases neonatal vulnerability due to an immature immune system and lower transplacental maternal IgG. This lack of protective antibodies facilitates rapid bacterial proliferation in the neonate. Early identification and intrapartum antibiotic prophylaxis are essential to mitigate this risk.
2. Rupture of membranes lasting >18 hours allows vaginal flora to ascend into the amniotic cavity. This prolonged exposure significantly increases the risk of vertical colonization to the fetus. Intrapartum antibiotics are indicated when this time threshold is exceeded to prevent infection.
3. A maternal intrapartum temperature of >38°C often indicates chorioamnionitis, an infection of the placental tissues. This inflammatory environment promotes fetal exposure to high bacterial loads during the birthing process. Fever serves as a primary clinical marker for initiating empirical intrapartum antibiotic treatment.
Rationale for incorrect answers
4. Nulliparity, or never having given birth, is not an independent risk factor for neonatal GBS infection. Risk is determined by colonization status and clinical labor factors rather than previous obstetric history. Maternal screening at 36 to 37 weeks gestation is the standard for determining risk.
5. Maternal asthma is a chronic respiratory condition that does not influence the presence or transmission of GBS. It involves airway inflammation but does not correlate with bacterial colonization of the birth canal. Clinical management focuses on respiratory stability rather than infectious prophylaxis for the neonate.
Test-taking strategy
- Analyze the Scenario and Question: The question asks for specific risk factors that increase the likelihood of neonatal GBS infection. The nurse must identify which maternal or labor circumstances facilitate bacterial transmission or neonatal susceptibility.
- Apply the Principle of Pathophysiology: GBS is an opportunistic infection transmitted from mother to infant.
- Evaluate labor duration and membrane status. Prolonged rupture (>18 hours) increases the window of exposure.
- Evaluate maternal physiological changes. Fever (>38°C) suggests an active infectious process like chorioamnionitis.
- Evaluate fetal maturity. Preterm infants (<37 weeks) have insufficient antibody protection and are more likely to succumb to early-onset sepsis.
- Differentiate Non-Contributory Factors:
- Rule out chronic conditions (Asthma) that do not affect bacterial flora.
- Rule out obstetric history (Nulliparity) that does not inherently increase colonization risk.
- Select the findings that meet the CDC criteria for intrapartum antibiotic prophylaxis.
Take home points
- Intrapartum antibiotic prophylaxis is indicated for women with GBS-positive cultures or those with unknown status and risk factors like prematurity or prolonged rupture.
- Neonatal GBS infection is categorized as early-onset (0-6 days) or late-onset (7-89 days), with early-onset being primarily prevented by intrapartum interventions.
- Maternal screening for GBS colonization typically occurs via vaginal and rectal swabs between 36 0/7 and 37 6/7 weeks of gestation.
- The primary antibiotic of choice for intrapartum prophylaxis is Penicillin G, which must be administered at least 4 hours before delivery for maximum efficacy.
A nurse is caring for a newborn whose mother received intrapartum GBS prophylaxis. Which finding would require immediate intervention?
Explanation
Neonatal early-onset sepsis frequently results from vertical transmission of gram-positive cocci during parturition. This bacterial invasion triggers a systemic inflammatory response, often manifesting as fulminant pneumonia or meningitis. Clinical presentation includes temperature instability, lethargy, and severe respiratory compromise.
Rationale for correct answer
1. Expiratory grunting and nasal flaring are hallmarks of neonatal respiratory distress and potential septicemia. These compensatory mechanisms indicate decreased pulmonary compliance and alveolar collapse. Immediate stabilization is mandatory to prevent respiratory failure and ensure adequate oxygenation.
Rationale for incorrect answers
2. The presence of bilateral Moro reflexes indicates a normal, symmetric neurological response in a neonate. Absence or asymmetry would suggest brachial plexus injury or central nervous system depression. Because this is an expected developmental finding, no clinical intervention is required.
3. Maintaining normal urine output, typically defined as 1 to 3 mL/kg/hr, reflects adequate renal perfusion and fluid balance. Oliguria would otherwise signal systemic hypoperfusion or septic shock in the newborn. This finding confirms hemodynamic stability and does not necessitate acute medical action.
4. Pink skin coloration indicates sufficient arterial oxygen saturation and peripheral perfusion without evidence of shunting. The absence of central cyanosis rules out immediate hypoxemia or significant cardiovascular distress. This is a reassuring physical assessment finding that requires only routine observation.
Test-taking strategy
- Analyze the Scenario: The infant is at risk for early-onset sepsis despite maternal prophylaxis, as no treatment is 100% effective. The question asks for a finding requiring immediate intervention, which points toward a life-threatening complication.
- Prioritize using ABCs: Apply the Airway, Breathing, and Circulation framework to the choices provided.
- Choice 1 addresses Breathing. Grunting and flaring are abnormal signs of respiratory effort and potential failure.
- Choices 2, 3, and 4 represent normal physiological findings for a newborn.
- Distinguish Normal vs. Abnormal:
- Identify that neurological symmetry (Moro), metabolic excretion (Urine), and integumentary color (Pink) are indicators of a stable transition to extrauterine life.
- Recognize that respiratory distress is the primary symptom of GBS-induced pneumonia, the most common manifestation of early-onset disease.
- Conclusion: Select the option that deviates from homeostasis and indicates acute physiological distress.
Take home points
- Respiratory distress in a neonate at risk for GBS infection is a clinical emergency requiring immediate evaluation for sepsis.
- Intrapartum antibiotic prophylaxis significantly reduces but does not entirely eliminate the risk of vertical transmission.
- Early-onset GBS disease typically presents within the first 24 to 48 hours of life with respiratory or hemodynamic instability.
- Normal neonatal findings such as symmetric reflexes and adequate voiding indicate stability and do not require acute intervention.
A nurse is teaching a client with a history of GBS-positive pregnancy about prevention in future pregnancies. Which statements should the nurse include?
Explanation
The bacterial colonization of the maternal lower genital tract by Streptococcus agalactiae is typically transient and asymptomatic. Maternal pathogenesis involves ascending infection after the rupture of membranes or during descent through the birth canal. Prevention relies on universal screening and targeted intrapartum chemoprophylaxis to reduce neonatal morbidity and mortality.
Rationale for correct answers
1. Maternal GBS status often changes between pregnancies, necessitating universal screening during every subsequent gestation. The American College of Obstetricians and Gynecologists recommends a distal vaginal and anorectal swab at 36 0/7 to 37 6/7 weeks. This ensures that current colonization status is accurately identified to guide intrapartum management.
3. Intrapartum antibiotic prophylaxis significantly reduces the risk of early-onset disease but does not eliminate it. Continuous neonatal assessment for signs of sepsis, such as temperature instability or poor feeding, remains essential post-delivery. Healthcare providers must remain vigilant for breakthrough infections that can occur despite appropriate maternal treatment.
Rationale for incorrect answers
2. Fever is only one of several criteria for initiating antibiotics when the maternal GBS status is unknown. In a client with a positive screen, prophylaxis is indicated regardless of the presence of intrapartum fever. Relying solely on temperature would result in missed opportunities for infection prevention and increased neonatal risk.
4. GBS colonization in the adult female is characterized by an asymptomatic carrier state in the majority of cases. It rarely causes symptomatic maternal disease, such as urinary tract infections or endocarditis, in healthy individuals. Because the condition is clinically silent, routine laboratory screening is the only reliable method for detection.
5. GBS is considered a component of the normal human gastrointestinal and vaginal commensal flora. Its presence is not related to personal cleanliness or hygiene practices and cannot be prevented through topical cleaning. Colonization patterns are influenced by complex biological factors rather than behavioral hygiene interventions.
Test-taking strategy
- Analyze the Question: The nurse is teaching a client about preventative measures and expectations for future pregnancies regarding GBS. The goal is to identify accurate clinical guidelines and safety protocols.
- Evaluate Clinical Guidelines:
- Recall the standard for maternal screening. Because colonization is transient, a previous positive result does not guarantee a positive status in the future; therefore, testing must be repeated.
- Identify the efficacy of chemoprophylaxis. Acknowledge that while antibiotics are highly effective, they are not a fail-safe, meaning infant monitoring is always a priority.
- Eliminate Misconceptions:
- Rule out the idea that GBS is symptomatic. It is a silent colonizer, which is why universal screening (Choice 1) is the gold standard.
- Discard the notion that hygiene prevents colonization. GBS is part of the microbiome, not a result of poor sanitation.
- Synthesize the Plan: Select the choices that reflect evidence-based practice for screening and post-birth vigilance.
Take home points
- Universal screening for Group B Streptococcus must be performed in every pregnancy between 36 and 37 weeks gestation.
- Intrapartum antibiotic prophylaxis is recommended for all women with a positive GBS culture in the current pregnancy to prevent vertical transmission.
- Newborns born to GBS-positive mothers require clinical observation for at least 12 to 24 hours to detect early signs of neonatal sepsis.
- GBS is a transient bacterium that colonizes the gastrointestinal and genitourinary tracts without causing symptoms in the mother.
Comprehensive Questions
A nurse is educating a pregnant client about preventing toxoplasmosis. Which of the following instructions should the nurse include?
Explanation
Toxoplasmosis is an infection caused by the protozoan Toxoplasma gondii, often transmitted via oocytes in feline feces or bradyzoites in contaminated tissues. Maternal infection triggers parasitemia, leading to transplacental transmission that causes congenital chorioretinitis, hydrocephalus, and intracranial calcifications. Preventive measures focus on eliminating exposure to definitive hosts and environmental reservoirs.
Rationale for correct answer
1. Felines serve as the definitive host, shedding infective oocytes in their feces, which pose a significant risk to seronegative pregnant women. Gardening exposes the individual to soil contaminated by feline excrement, necessitating the use of protective gloves. Avoiding litter box maintenance prevents the direct inhalation or ingestion of parasites during the sporulation phase.
Rationale for incorrect answers
2. Ingesting undercooked or raw meat is a primary route for parasitic transmission via tissue cysts containing bradyzoites. Pregnant women must ensure meat reaches an internal temperature of ≥63°C to ≥74°C to achieve pathogen eradication. Suggesting undercooked meat for iron intake is clinically dangerous and contradicts standard prenatal nutritional safety guidelines.
3. Handling raw meat without subsequent hand hygiene facilitates accidental ingestion of the parasite through cross-contamination of mucosal surfaces. Handwashing with soap and water is a fundamental aseptic technique required to disrupt the transmission cycle of Toxoplasma gondii. Failure to sanitize hands significantly increases the risk of maternal infection and subsequent fetal harm.
4. Sharing utensils with children is a risk factor for Cytomegalovirus (CMV) rather than toxoplasmosis infection. While reducing saliva sharing is important for general prenatal infection control, it does not target the specific transmission vector of Toxoplasma gondii. Toxoplasmosis is primarily foodborne or environmental, whereas CMV is typically spread through direct contact with infected bodily fluids.
Test-taking strategy
- Identify the Core Pathogen: The question focuses on toxoplasmosis, caused by Toxoplasma gondii. This requires an understanding of its specific transmission vectors, which include feline feces, contaminated soil, and undercooked meat.
- Apply Risk Reduction Principles:
- Analyze Choice 1: This directly addresses the feline-environmental link. Gloves and avoidance of litter are standard, evidence-based recommendations for pregnancy.
- Analyze Choices 2 and 3: These encourage high-risk behaviors (consuming raw meat and poor hygiene) that facilitate parasitic entry.
- Analyze Choice 4: This is a distractor related to a different pathogen (CMV).
- Use Rule of Exclusion: Eliminate options that promote infection transmission or address the wrong infectious agent.
- Conclusion: Select the intervention that prioritizes primary prevention by creating a physical barrier between the host and the known parasite reservoir.
Take home points
- Pregnant women should delegate the cleaning of cat litter boxes to others to avoid exposure to sporulated oocytes.
- All meat products must be cooked to appropriate internal temperatures to destroy tissue cysts and prevent foodborne transmission.
- Gardening activities require the use of gloves and thorough handwashing due to the potential presence of parasites in the soil.
- Toxoplasmosis screening is typically reserved for symptomatic women or those with high-risk exposures due to the risk of congenital anomalies.
A nurse is caring for a pregnant client who is GBS positive. Which intervention is most critical for preventing early-onset GBS disease in the newborn?
Explanation
Intrapartum antibiotic prophylaxis (IAP) involves the intravenous administration of bactericidal agents to achieve therapeutic concentrations in the fetal circulation and amniotic fluid. This pharmacological intervention interrupts vertical colonization during the descent of the fetus through the birth canal. Successful prevention requires maintaining an inhibitory concentration of antibiotics to neutralize the gram-positive cocci before neonatal pathogenesis begins.
Rationale for correct answer
2. Intravenous penicillin G or ampicillin must be initiated at least 4 hours prior to parturition to ensure adequate fetal tissue levels. This duration allows for sufficient transplacental passage and high concentrations within the amniotic compartment, effectively reducing the bacterial load. Clinical evidence confirms that achieving this time threshold is the most effective method for preventing early-onset sepsis in the neonate.
Rationale for incorrect answers
1. Oral antibiotics do not reach the necessary pharmacokinetic peaks in the maternal serum or amniotic fluid to prevent vertical transmission. The gastrointestinal absorption rate is too slow and inconsistent to provide a rapid bactericidal effect during active labor. Standard of care strictly mandates the parenteral route to ensure immediate and predictable bioavailability for the prevention of neonatal GBS disease.
3. A cesarean delivery is only indicated for GBS prevention if performed prior to the rupture of membranes and before the onset of labor. Once labor begins or membranes rupture, the fetus is potentially exposed to ascending bacteria, making surgical intervention alone insufficient for prophylaxis. Routine cesarean delivery for all GBS-positive clients is not recommended due to associated surgical risks and lack of additional benefit over IAP.
4. Topical application of antiseptic agents to the perineum or vagina does not eliminate the deep mucosal reservoirs of Group B Streptococcus. Bacterial counts quickly rebound after mechanical scrubbing, failing to protect the infant during the expulsive stage of labor. Intrapartum systemic antibiotics are the only proven method to reduce neonatal infection rates, as local antiseptics do not address the ascending nature of the pathogen.
Test-taking strategy
- Analyze the Scenario: The client is GBS positive, and the goal is to identify the most critical intervention for preventing newborn infection. This is a prioritization question focusing on evidence-based prophylaxis.
- Evaluate the Options:
- Route of Administration: Compare oral (Choice 1) versus intravenous (Choice 2). IV administration is the gold standard because it provides rapid, high-dose systemic coverage.
- Timing: Identify the critical time window. The 4-hour threshold is the specific clinical benchmark for "adequate" prophylaxis.
- Procedure Type: Contrast surgical (Choice 3) and mechanical (Choice 4) interventions against pharmacological ones.
- Rule Out Distractors:
- Eliminate Choice 4; local treatments are ineffective against systemic colonization.
- Eliminate Choice 1; oral routes are inappropriate for intrapartum emergencies.
- Eliminate Choice 3; cesarean birth is not a standard GBS prevention strategy unless it is pre-labor.
- Conclusion: Select the option that aligns with CDC and ACOG guidelines for intrapartum antibiotic timing and route.
Take home points
- Intravenous Penicillin G is the first-line antibiotic for intrapartum GBS prophylaxis due to its narrow spectrum and efficacy.
- Prophylaxis is considered adequate if administered at least 4 hours before the second stage of labor is completed.
- If a client has a penicillin allergy with a low risk for anaphylaxis, Cefazolin is the recommended alternative for prophylaxis.
- Clindamycin should only be utilized if the GBS isolate is known to be susceptible and the client is at high risk for penicillin anaphylaxis.
A nurse is educating a pregnant client about congenital rubella syndrome. Which of the following findings are associated with this infection? Select all that apply
Explanation
Congenital rubella syndrome (CRS) results from maternal viremia and subsequent transplacental infection, primarily during the first trimester. The virus disrupts fetal organogenesis, leading to a classic triad of clinical manifestations involving the auditory, ophthalmic, and cardiovascular systems. Prevention is achieved through pre-pregnancy vaccination, as the infection can cause permanent multisystemic damage to the developing embryo.
Rationale for correct answers
1. Sensorineural hearing loss is the most prevalent permanent sequela of congenital rubella infection. The virus causes direct damage to the organ of Corti or the auditory nerve during critical developmental windows. This deficit may be bilateral and is often not fully diagnosed until the neonatal period or early infancy during universal hearing screenings.
2. Ocular abnormalities, specifically congenital cataracts, occur due to viral interference with lens fiber development. These opacities are frequently accompanied by "salt-and-pepper" retinopathy and microphthalmia, leading to significant visual impairment. Surgical intervention is typically required shortly after birth to prevent irreversible amblyopia and restore visual input.
3. The most characteristic cardiovascular complication associated with CRS is a patent ductus arteriosus (PDA). The rubella virus inhibits the normal physiological closure of the ductus, leading to persistent left-to-right shunting of blood. Other common defects include pulmonary artery stenosis, which contributes to significant neonatal hemodynamic instability and respiratory strain.
Rationale for incorrect answers
4. Hydrops fetalis, characterized by generalized fetal edema and fluid accumulation in serous cavities, is more commonly associated with Parvovirus B19. While rubella causes systemic damage, it does not typically manifest as the profound erythroblastosis or congestive heart failure required to produce hydrops. This finding indicates a different etiology of fetal hydrops rather than the classic rubella phenotype.
5. Chorioretinitis is a classic finding in congenital toxoplasmosis and Cytomegalovirus (CMV) rather than rubella. Rubella typically presents with a non-inflammatory retinopathy described as a speckled or "salt-and-pepper" appearance. Distinguishing between these ophthalmic findings is essential for the differential diagnosis of various TORCH infections in the neonatal period.
Test-taking strategy
- Identify the Core Condition: The question focuses on Congenital Rubella Syndrome (CRS). Success depends on recalling the "Gregg Triad," which consists of specific defects in the ears, eyes, and heart.
- Categorize Findings:
- Apply knowledge of the triad: Deafness (Choice 1), Cataracts (Choice 2), and Heart Defects (Choice 3) fit the classic clinical picture.
- Evaluate the distractors: Hydrops fetalis (Choice 4) is a hallmark of Parvovirus B19 or Rh isoimmunization. Chorioretinitis (Choice 5) is the signature ocular lesion for Toxoplasmosis and CMV.
- Utilize Pathophysiology:
- Recognize that CRS involves teratogenic effects during the first trimester.
- Differentiate between the "salt-and-pepper" retinopathy of rubella and the inflammatory chorioretinitis of other TORCH pathogens.
- Conclusion: Select the three options that comprise the classic triad of rubella-induced embryopathy.
Take home points
- The risk of fetal infection and severe malformation is highest when maternal rubella occurs before 12 weeks gestation.
- Sensorineural deafness is the most common single clinical finding in infants born with congenital rubella syndrome.
- The live-attenuated MMR vaccine is contraindicated during pregnancy and should be administered at least 28 days before conception.
- Neonates with CRS are considered infectious and may shed the virus in nasopharyngeal secretions and urine for up to 1 year.
A nurse is caring for a newborn exposed to maternal HSV lesions at birth. Which is the most appropriate delivery method to prevent transmission?
Explanation
Neonatal herpes simplex virus (HSV) infection is a devastating condition often acquired through vertical transmission during transit through an infected birth canal. The virus undergoes retrograde transport along sensory neurons, potentially leading to disseminated disease, encephalitis, or localized skin, eye, and mouth (SEM) involvement. Prevention necessitates the avoidance of direct fetal contact with active herpetic vesicles or prodromal viral shedding.
Rationale for correct answer
2. Performing a cesarean delivery is the most effective intervention to bypass the colonized vaginal tract and prevent direct neonatal exposure to active lesions. Clinical guidelines mandate surgical intervention if prodromal symptoms or visible genital vesicles are present at the onset of labor. This prophylactic approach significantly reduces the risk of neonatal viral inoculation and subsequent central nervous system complications.
Rationale for incorrect answers
1. A vaginal delivery in the presence of active lesions poses a high risk for mucocutaneous transmission as the fetus descends through the birth canal. Contact with infectious secretions can lead to neonatal viremia, which carries a high mortality rate if untreated. While vaginal birth is acceptable if no lesions are present, it is contraindicated when active HSV manifestations are identified during labor.
3. Using forceps during delivery increases the risk of iatrogenic trauma and scalp abrasions, which serve as portals of entry for the virus. Any scalp instrumentation is strictly avoided in HSV-exposed pregnancies to minimize the risk of direct inoculation into the fetal bloodstream. Forceps do not provide a barrier to the infectious flora of the vagina and may exacerbate the risk of transmission.
4. An episiotomy is a surgical incision of the perineum that creates a large open wound surface during the second stage of labor. This procedure increases the surface area for potential viral contact and facilitates the entry of HSV pathogens into maternal and fetal tissues. Utilizing an episiotomy does not assist in infection control and is avoided to prevent the spread of the virus to the neonate.
Test-taking strategy
- Analyze the Scenario: The nurse is managing a delivery where the mother has active HSV lesions, which are highly infectious. The primary goal is to identify a delivery method that avoids fetal contact with these lesions.
- Apply Infection Control Principles:
- Evaluate the route: Vaginal delivery (Choice 1) and its variations (Choice 4) involve passage through the infected area.
- Evaluate the risk of trauma: Forceps (Choice 3) can cause skin breaks, making the neonate more susceptible to the virus.
- Identify the bypass method: Cesarean delivery (Choice 2) is the only option that avoids the vaginal canal entirely.
- Use Clinical Guidelines: Recall that ACOG recommendations prioritize surgical delivery when active genital herpes lesions or prodromal symptoms (pain, burning) are present at the time of labor to prevent neonatal herpes.
- Conclusion: Select the option that provides a barrier to transmission by altering the delivery route.
Take home points
- Cesarean delivery is indicated for pregnant women with active genital HSV lesions or prodromal symptoms at the time of labor.
- If no active lesions are present at the onset of labor, a vaginal delivery is typically safe and recommended.
- Neonatal HSV can manifest as disseminated disease involving the liver and lungs, which has a mortality rate exceeding 80% without treatment.
- Suppressive antiviral therapy, such as Acyclovir, is often initiated at 36 weeks gestation to reduce the likelihood of active lesions at term.
A nurse is providing education on maternal CMV infection prevention. Which of the following instructions should the nurse include? Select all that apply
Explanation
Cytomegalovirus (CMV) is a ubiquitous double-stranded DNA herpesvirus characterized by lifelong latency and periodic reactivation. Maternal infection is predominantly acquired through direct contact with infectious bodily fluids, particularly the saliva and urine of young children. Congenital transmission during primary viremia often results in permanent sensorineural hearing loss, microcephaly, and periventricular calcifications.
Rationale for correct answers
1. Young children are primary reservoirs for CMV and can shed the virus in their urine for months following an asymptomatic infection. Thorough handwashing after diaper changes is a critical mechanical barrier to prevent fecal-oral or mucosal inoculation of the mother. Consistent aseptic technique reduces the risk of primary maternal infection and subsequent transplacental transmission to the fetus.
2. Salivary transmission is a major vector for CMV, especially when pregnant women share food, cups, or utensils with toddlers. The virus remains stable on moist surfaces, facilitating horizontal transmission through oral contact with contaminated objects. Avoiding the sharing of saliva is the most effective behavioral intervention for reducing maternal exposure to the virus in household settings.
5. Daycare environments are high-risk zones for viral transmission due to the high density of children and shared fomites. Implementing strict hygiene protocols, such as sanitizing toys and frequent handwashing, disrupts the environmental spread of the virus. Because daycare workers are at increased risk, these precautionary measures are essential for protecting pregnant staff and parents from CMV.
Rationale for incorrect answers
3. The MMR vaccine is a live-attenuated preparation and is strictly contraindicated during pregnancy due to the theoretical risk of fetal infection. Furthermore, the MMR vaccine targets measles, mumps, and rubella, but provides zero immunological protection against CMV. There is currently no licensed vaccine available to prevent Cytomegalovirus infection in the general or pregnant population.
4. Avoiding undercooked meat is a preventive strategy specific to toxoplasmosis and Listeria, rather than Cytomegalovirus. CMV is not a foodborne pathogen associated with animal tissue; it is exclusively spread through human-to-human contact with bodily fluids. While dietary safety is important in pregnancy, it does not address the transmission dynamics of the CMV herpesvirus.
Test-taking strategy
- Analyze the Core Pathogen: The question focuses on Cytomegalovirus (CMV). The nurse must identify the specific transmission route to select the correct preventive measures. CMV is primarily spread through saliva and urine of small children.
- Evaluate the Options based on Transmission:
- Choices 1, 2, and 5 directly address the contact with pediatric fluids (diapers, utensils, daycare settings). These are the standard "catch-no-fever" hygiene guidelines for CMV.
- Choice 3 mentions a vaccine. Recall that MMR is for Rubella, not CMV, and is prohibited in pregnancy.
- Choice 4 addresses foodborne pathogens. Distinguish CMV from Toxoplasmosis, which is the pathogen linked to raw meat.
- Apply the Rule of Specificity: Select only the interventions that target the human fluid transmission route.
- Conclusion: Choose 1, 2, and 5 as they represent the primary prevention strategies recommended by major health organizations for CMV.
Take home points
- Cytomegalovirus is the most common congenital viral infection and a leading cause of non-genetic sensorineural hearing loss in children.
- Pregnant women should avoid kissing young children on the mouth or sharing food to minimize exposure to infectious saliva.
- CMV infection is often asymptomatic in the mother, making behavioral prevention strategies more critical than symptom monitoring.
- Hand hygiene after handling children's toys, tissues, or diapers is the cornerstone of reducing the risk of primary CMV acquisition.
A nurse is caring for a pregnant client who has a confirmed primary cytomegalovirus (CMV) infection at 12 weeks gestation. Which of the following is the most important fetal complication to monitor for?
Explanation
Cytomegalovirus (CMV) is a ubiquitous herpesvirus that remains the most frequent cause of congenital viral infection worldwide. Following primary maternal viremia, the virus crosses the placenta and infects the fetal germinal matrix, leading to irreversible neurological damage. Chronic viral replication within the fetal central nervous system and inner ear results in significant long-term developmental morbidities.
Rationale for correct answer
2. Sensorineural hearing loss and microcephaly are the hallmark complications of congenital CMV, particularly when infection occurs during the first trimester. The virus causes targeted destruction of the cochlear hair cells and interferes with neuronal migration, resulting in a reduced brain volume. These neurodevelopmental deficits represent the most common and severe sequelae identified in affected neonates, requiring long-term multidisciplinary management and monitoring.
Rationale for incorrect answers
1. Congenital heart defects are typically associated with Rubella or maternal pregestational diabetes rather than Cytomegalovirus. CMV does not primarily disrupt cardiac organogenesis during the embryonic period of development. While systemic infection can occur, the pathogen does not have a high affinity for myocardial or endocardial tissues during fetal maturation.
3. Limb hypoplasia is a characteristic finding of congenital Varicella syndrome, occurring after maternal chickenpox infection in early pregnancy. CMV infection does not interfere with the mesenchymal development required for limb bud formation or elongation. Monitoring for musculoskeletal deformities is not indicated for a diagnosis of primary CMV infection.
4. Neonatal jaundice is a common and often transient finding in many newborns, including those with sepsis or ABO incompatibility. While CMV can cause hepatosplenomegaly and conjugated hyperbilirubinemia, it is not the most significant or specific complication compared to permanent neurological damage. Jaundice is a manageable clinical sign, whereas neurosensory deficits are irreversible and represent a higher priority for long-term prognosis.
Test-taking strategy
- Analyze the Scenario: The client has a confirmed primary CMV infection in the first trimester (12 weeks), which is the period of highest risk for severe fetal damage.
- Categorize Clinical Findings:
- Apply knowledge of TORCH infections. Recall that CMV is the leading non-genetic cause of sensorineural hearing loss.
- Identify the target organ: CMV has a strong tropism for the central nervous system, leading to microcephaly and periventricular calcifications.
- Differentiate Pathogens:
- Eliminate Choice 1: Heart defects = Rubella.
- Eliminate Choice 3: Limb hypoplasia = Varicella.
- Evaluate Choice 4: Jaundice is a non-specific finding that does not carry the same prognostic weight as neurosensory loss.
- Conclusion: Select the option that identifies the permanent and specific neurological complications associated with the CMV herpesvirus.
Take home points
- Cytomegalovirus is the most common cause of non-hereditary sensorineural hearing loss in the pediatric population.
- First-trimester maternal infection carries the highest risk for severe fetal neurodevelopmental impairment.
- Ultrasound findings such as intracranial calcifications, ventriculomegaly, and microcephaly often suggest a congenital CMV infection.
- Long-term follow-up is essential for affected infants, as hearing loss may be late-onset or progressive even if the neonate is asymptomatic at birth.
A nurse is educating a client who is pregnant about preventing toxoplasmosis. Which statement by the client indicates understanding?
Explanation
Toxoplasmosis is a parasitic infection caused by the protozoan Toxoplasma gondii, characterized by an obligatory intracellular lifecycle. Transmission to the gravid host occurs via the ingestion of sporulated oocytes from feline feces or bradyzoites residing in skeletal muscle cysts of intermediate hosts. Following maternal parasitemia, the organism traverses the placenta, potentially inducing fetal hydrocephalus, intracranial calcifications, and retinochoroiditis.
Rationale for correct answer
1. Consuming meat heated to internal temperatures below 63°C to 74°C fails to denature the protective wall of parasitic tissue cysts. Additionally, fruits and vegetables can be contaminated by oocytes present in soil or irrigation water, necessitating mechanical removal through thorough washing. Adhering to these dietary safeguards is the primary method for preventing foodborne transmission of the parasite during pregnancy.
Rationale for incorrect answers
2. While wearing gloves provides a physical barrier, the safest clinical recommendation is for pregnant women to avoid litter box maintenance entirely. Feline feces contain oocytes that become infective after 1 to 5 days of sporulation in the environment. Delegating this task to a non-pregnant individual eliminates the risk of accidental inhalation or ingestion of the parasite during the cleaning process.
3. There is currently no commercially available or approved human vaccine for the prevention of toxoplasmosis in any population. Furthermore, the administration of live vaccines is generally contraindicated during pregnancy due to the risk of unintentional fetal infection. Preventive efforts must rely exclusively on behavioral modifications and environmental hygiene rather than active immunization.
4. Hand hygiene is a fundamental prophylactic measure that must be performed regardless of maternal symptomatic status. Many individuals remain asymptomatic during the initial phase of infection, though they can still transmit the parasite to the fetus. Rigorous handwashing is required specifically after handling raw meat, gardening, or contact with soil to prevent the mechanical transfer of pathogens to mucosal surfaces.
Test-taking strategy
- Identify the Pathogen and Host: The question focuses on toxoplasmosis (Toxoplasma gondii). Recall that the primary reservoirs are cats (definitive hosts) and contaminated food/soil (intermediate vehicles).
- Evaluate the Statements:
- Choice 1: Addresses two major transmission routes (undercooked meat and contaminated produce). This aligns with evidence-based prenatal education.
- Choice 2: Although gloves are mentioned, "avoidance" is the priority recommendation. Many boards consider any contact with the litter box a high-risk behavior to be discouraged.
- Choice 3: Recognize the scientific fact that a toxoplasmosis vaccine does not exist.
- Choice 4: Dismiss this as it ignores the asymptomatic nature of the infection and basic infection control principles.
- Distinguish the Best Answer: Select the statement that demonstrates a comprehensive understanding of food safety and environmental risks.
Take home points
- Pregnant women should ensure all meat is cooked to a safe internal temperature (71°C for ground meat, 63°C for whole cuts) to kill tissue cysts.
- Avoidance of cat litter box cleaning is the most effective way to prevent exposure to infective oocytes shed by felines.
- Fruits and vegetables must be peeled or washed thoroughly to remove parasites that may be present in contaminated soil.
- Hand hygiene after gardening or handling raw meat is essential, as the parasite can be accidentally ingested through hand-to-mouth contact.
A nurse is assessing a newborn whose mother had active genital herpes simplex virus (HSV) lesions at term. Which finding in the newborn would most likely indicate central nervous system involvement?
Explanation
Neonatal herpes simplex virus (HSV) infection is a high-mortality condition characterized by three distinct clinical patterns: disseminated disease, skin-eye-mouth (SEM) disease, and central nervous system (CNS) disease. The virus typically gains entry through the mucous membranes or breaks in the skin, followed by rapid retrograde axonal transport to the brain. CNS involvement occurs in approximately 33% of infected neonates and carries a poor neurological prognosis even with aggressive antiviral therapy.
Rationale for correct answer
2. Encephalitis manifesting as focal seizures, extreme lethargy, or bulging fontanelles is the hallmark of CNS-involved neonatal HSV. The virus induces hemorrhagic necrosis within the temporal lobes, leading to profound electrical instability in the brain. These symptoms typically appear between 2 to 3 weeks of life and require immediate cerebrospinal fluid analysis to confirm the presence of viral DNA.
Rationale for incorrect answers
1. Skin vesicles located on the torso, scalp, or periorbital areas are characteristic of SEM disease, the most limited form of neonatal HSV. While these vesicles provide a clinical clue to the diagnosis, they do not inherently confirm that the virus has breached the blood-brain barrier. Many infants with isolated skin involvement do not progress to encephalitis if treated promptly with systemic acyclovir.
3. Hepatosplenomegaly, or enlargement of the liver and spleen, is a primary indicator of disseminated disease rather than isolated CNS involvement. This form of the infection mimics bacterial sepsis and involves multiple organ systems, including the lungs and adrenal glands. While life-threatening, it represents visceral spread of the virus rather than localized central nervous system infection.
4. Jaundice is a frequent and non-specific finding in the neonatal period that may result from hepatic dysfunction or hemolysis. In the context of HSV, it is often a sign of viral hepatitis associated with the disseminated form of the disease. Because jaundice occurs in many neonatal conditions, it lacks the diagnostic specificity required to identify central nervous system pathology.
Test-taking strategy
- Analyze the Question: The nurse is looking for a finding that most likely indicates central nervous system (CNS) involvement in a newborn exposed to HSV.
- Categorize Clinical Manifestations:
- Recall the three stages of neonatal HSV: SEM (Skin, Eyes, Mouth), Disseminated (Multi-organ), and CNS (Brain).
- Match the symptoms to the stage:
- Skin vesicles (Choice 1) = SEM stage.
- Seizures/Encephalitis (Choice 2) = CNS stage.
- Hepatosplenomegaly (Choice 3) = Disseminated stage.
- Jaundice (Choice 4) = Disseminated/General sepsis.
- Prioritize Neurological Symptoms: Any mention of seizures, tremors, or altered consciousness in a neonate with a history of HSV exposure must be prioritized as a sign of brain infection.
- Conclusion: Select the option that specifically identifies brain inflammation and the resulting neurological dysfunction.
Take home points
- CNS involvement in neonatal HSV typically manifests between the second and third weeks of life, often presenting with focal seizures.
- Disseminated HSV infection carries the highest mortality rate and often presents earlier, within the first week of life, resembling bacterial sepsis.
- Skin vesicles are absent in up to 30 to 40 percent of neonates with CNS or disseminated HSV, making clinical suspicion vital.
- High-dose intravenous Acyclovir is the gold standard for treating all forms of neonatal HSV to improve survival and neurodevelopmental outcomes.
A nurse is providing education to a non-immune pregnant client exposed to rubella. Which intervention should the nurse prioritize?
Explanation
Maternal rubella infection is characterized by a maculopapular exanthem and retroauricular lymphadenopathy following a brief prodromal phase. Pathogenesis involves respiratory droplet transmission followed by systemic dissemination and transplacental migration during the viremic stage. Because the virus is highly teratogenic, especially during the first 12 weeks of gestation, management focuses on laboratory confirmation of seroconversion and longitudinal fetal surveillance.
Rationale for correct answer
2. Since no curative antiviral therapy exists for rubella, management necessitates monitoring for seroconversion through maternal IgM and IgG titers. IgM antibodies typically appear within 3 to 7 days after symptom onset and indicate an acute infection that may threaten fetal development. Supportive care is provided to manage maternal symptoms while utilizing serial ultrasounds to assess for signs of congenital rubella syndrome or fetal demise.
Rationale for incorrect answers
1. The MMR vaccine contains a live-attenuated virus which is strictly contraindicated during pregnancy due to the theoretical risk of placental infection. Administering this vaccine to a pregnant client could potentially cause the very embryopathy it is designed to prevent in the general population. Immunization must be deferred until the postpartum period, and the client should be advised to avoid conception for at least 28 days following the injection.
3. While rubella immune globulin (IG) may be considered for a non-immune pregnant woman who refuses pregnancy termination, it is not a prioritized or highly effective intervention. IG may lengthen the incubation period or modify maternal symptoms, but it does not reliably prevent fetal infection or viremia. It is not recommended for routine use because it often provides a false sense of security without mitigating the risk of congenital anomalies.
4. Strict bed rest has no pharmacological effect on the replication of the rubella virus or the prevention of vertical transmission to the fetus. While rest may alleviate maternal fatigue associated with the viral prodrome, it does not alter the clinical course of the disease or protect fetal organogenesis. Clinical prioritization must remain on diagnostic monitoring and counseling regarding fetal risks rather than activity restriction.
Test-taking strategy
- Analyze the Scenario: The client is non-immune, pregnant, and exposed. This is a high-risk situation for Congenital Rubella Syndrome (CRS).
- Apply Safety and Contraindications:
- Immediately rule out Choice 1. Live vaccines (MMR, Varicella) are never given during pregnancy.
- Evaluate Choice 3. Immune globulin is a weak intervention that does not effectively stop vertical transmission and is therefore not the priority.
- Prioritize Data Collection:
- In the absence of a cure, the most critical nursing action is to confirm if an infection has occurred.
- IgM testing (Choice 2) is the standard diagnostic step to determine acute maternal infection.
- Differentiate Treatments:
- Recognize that rubella management is supportive and diagnostic.
- Rule out "bed rest" (Choice 4) as it lacks scientific evidence for improving infectious outcomes.
- Conclusion: Select the option that focuses on diagnostic surveillance of maternal-fetal status.
Take home points
- Rubella is most dangerous during the first trimester, with a fetal infection risk of up to 90 percent.
- Maternal IgM antibodies are the primary marker used to diagnose a current or very recent rubella infection.
- The MMR vaccine is a live vaccine and must only be administered when the client is confirmed to be non-pregnant.
- Congenital rubella syndrome causes permanent defects, including cataracts, cardiac anomalies, and sensorineural deafness.
A nurse is teaching a group of pregnant clients about strategies to reduce the risk of TORCH infections. Which of the following should the nurse include? Select all that apply
Explanation
The TORCH complex constitutes a group of congenital infections that traverse the placenta to induce fetal teratogenesis. These pathogens cause multi-organ inflammatory damage resulting in microcephaly, chorioretinitis, and hepatosplenomegaly. Preventive strategies prioritize disrupting environmental and zoonotic transmission pathways to protect the developing embryo and fetus.
Rationale for correct answers
1. Toxoplasma gondii is an obligate intracellular protozoan parasite shed as oocytes in feline feces. Avoiding litter boxes and wearing gloves during soil contact prevents the accidental ingestion of these infectious stages. This intervention is critical for preventing congenital toxoplasmosis and its associated neurological and ocular sequelae.
2. Cytomegalovirus is frequently shed in the urine and saliva of asymptomatic young children. Rigorous hand hygiene after diaper changes disrupts the horizontal transmission of the virus to the pregnant host. This measure significantly reduces the risk of primary infection, the leading cause of non-genetic neonatal deafness.
4. Consuming meat with an internal temperature <63°C may harbor viable cysts, while unpasteurized dairy can contain Listeria monocytogenes. Ensuring thorough cooking and pasteurization eliminates these foodborne pathogens from the maternal diet. These dietary modifications protect the fetus from systemic infection, spontaneous abortion, and preterm labor.
5. Certain TORCH-related agents, such as the Zika virus, are transmitted via arthropod vectors like Aedes mosquitoes. Utilizing EPA-approved insect repellents serves as a barrier against vector-borne inoculation during pregnancy. This strategy is essential to prevent severe fetal developmental anomalies, including congenital Zika syndrome and microcephaly.
Rationale for incorrect answers
3. Live-attenuated vaccines, including MMR and varicella, utilize weakened pathogens that retain the potential for transplacental migration. Administering these during pregnancy is strictly contraindicated due to the theoretical risk of causing fetal infection. Immunization status should be assessed preconceptionally, and live vaccines must be deferred until the postpartum period.
Test-taking strategy
- Identify the Main Idea: The question seeks comprehensive preventive strategies for the TORCH acronym pathogens (Toxoplasmosis, Other/Listeria/Zika, Rubella, Cytomegalovirus, Herpes).
- Evaluate Each Pathogen Transmission Route:
- For Toxoplasmosis, evaluate environmental reservoirs. Choice 1 correctly addresses feline and soil exposure.
- For Cytomegalovirus, evaluate pediatric fluids. Choice 2 correctly addresses urine and saliva exposure via handwashing.
- For foodborne risks (Listeria/Toxoplasma), evaluate ingestion. Choice 4 correctly identifies undercooked meat and raw dairy.
- For "Other" pathogens like Zika, evaluate vector transmission. Choice 5 correctly identifies repellent use.
- Apply Safety Contraindications:
- Analyze Choice 3 regarding immunization. Recall the pharmacological principle that live vaccines are contraindicated in pregnancy to prevent iatrogenic fetal harm.
- Rule Out/Select: Exclude the unsafe vaccine option and select the remaining behavioral and environmental interventions.
Take home points
- Primary prevention of toxoplasmosis requires avoiding cat feces and ensuring all meat is cooked to a safe internal temperature.
- Cytomegalovirus risk is minimized by avoiding the sharing of food or utensils with children and practicing frequent hand hygiene.
- Live-attenuated vaccines are never administered during pregnancy but are recommended in the immediate postpartum period for non-immune clients.
- Vector-borne TORCH infections are prevented by using protective clothing and insect repellents in areas with known mosquito activity.
A nurse is caring for a newborn whose mother had untreated primary syphilis during pregnancy. Which long-term complication is most concerning for the newborn?
Explanation
Syphilis is caused by the spirochete Treponema pallidum, which possesses the unique ability to traverse the placental barrier at any stage of gestation. This hematogenous spread results in a multisystemic inflammatory response within the fetus, potentially causing osteochondritis, hepatosplenomegaly, and severe neurosyphilis. If maternal primary syphilis remains untreated, the risk of vertical transmission to the neonate is approximately 70% to 100%, often leading to permanent structural and neurological sequelae.
Rationale for correct answer
2. Untreated maternal syphilis leads to profound neurodevelopmental deficits and classic stigmata such as Hutchinson teeth, saddle nose, and interstitial keratitis. The pathogen invades the cerebrospinal fluid, causing chronic meningovascular inflammation that impairs cognitive and motor maturation. Long-term surveillance is mandatory to monitor for late-onset manifestations, including sensorineural deafness and skeletal deformities like saber shins.
Rationale for incorrect answers
1. Hyperbilirubinemia is a common neonatal finding that can occur due to physiologic transition or hematologic incompatibility. While syphilis can cause hepatic inflammation and jaundice, hyperbilirubinemia itself is usually an acute, manageable condition rather than a permanent long-term complication. It does not carry the same degree of chronic morbidity as the systemic damage caused by Treponema pallidum.
3. Transient tachypnea of the newborn (TTN) is a self-limiting respiratory condition caused by delayed fetal lung fluid clearance. It typically resolves within 24 to 72 hours and does not result in long-term pulmonary or neurological impairment. TTN is not a pathological outcome of syphilitic infection, which primarily targets the mesenchymal tissues and central nervous system.
4. Congenital cataracts are the hallmark of congenital Rubella syndrome rather than syphilis. While syphilis can cause ophthalmic issues like interstitial keratitis later in childhood, it does not typically disrupt the embryonic development of the lens. Distinguishing between these pathognomonic signs is essential for identifying the specific TORCH agent responsible for neonatal anomalies.
Test-taking strategy
- Analyze the Scenario: The mother had untreated primary syphilis, which carries the highest risk of fetal infection. The question asks for the most concerning long-term complication.
- Apply Pathophysiology:
- Recall that syphilis is a systemic infection with a high affinity for the bones and the central nervous system.
- Identify that "primary" and "untreated" status in the mother ensures a high bacterial load reached the fetus.
- Evaluate the Options:
- Eliminate acute/transient issues: Hyperbilirubinemia (Choice 1) and TTN (Choice 3) are short-term neonatal concerns.
- Differentiate among congenital anomalies: Cataracts (Choice 4) are specific to Rubella.
- Identify the chronic impact: Neurodevelopmental deficits and systemic stigmata (Choice 2) represent the permanent, life-altering effects of Treponema pallidum.
- Conclusion: Select the option that encompasses the multisystemic and irreversible nature of congenital syphilis.
Take home points
- Congenital syphilis is prevented through early prenatal screening and treatment with Penicillin G, the only effective agent for fetal therapy.
- Hutchinson triad, a classic sign of late congenital syphilis, includes notched incisors, interstitial keratitis, and eighth-nerve deafness.
- Early congenital syphilis manifestations (0-2 years) include snuffles (rhinitis), skin rashes, and pseudoparalysis of Parrot due to osteochondritis.
- All infants born to mothers with reactive syphilis serology must undergo a complete evaluation, including a physical exam and quantitative nontreponemal tests.
A client at 28 weeks gestation asks the nurse why she is being screened for Group B Streptococcus (GBS). Which response is most accurate?
Explanation
Group B Streptococcus (GBS) is a transient commensal bacterium that colonizes the maternal anorectal and vaginal mucosa. While typically non-pathogenic in the adult, vertical transmission during labor can result in rapid neonatal bacterial proliferation. Preventive medicine focuses on universal screening to identify candidates for intrapartum antibiotic prophylaxis, thereby reducing the incidence of early-onset neonatal pneumonia and meningitis.
Rationale for correct answer
2. Identifying maternal GBS colonization allows for the administration of intravenous antibiotics during the intrapartum period. This intervention maintains a bactericidal concentration in the fetal circulation and amniotic fluid, effectively neutralizing the bacteria before they can invade neonatal tissues. Prophylaxis is the primary defense against early-onset sepsis, which can otherwise lead to severe respiratory failure and systemic collapse in the newborn.
Rationale for incorrect answers
1. While GBS can indeed cause maternal urinary tract infections, the statement that it does not affect the infant is a significant medical error. GBS is the leading cause of neonatal sepsis and meningitis in the first week of life. Neglecting the fetal risk ignores the primary clinical purpose of prenatal screening and the potential for life-threatening neonatal complications.
3. GBS screening is performed via a vaginal-rectal swab, not through bloodwork, and it has no diagnostic value for evaluating renal function. Kidney function is typically monitored through serum creatinine and blood urea nitrogen levels or urinalysis for protein. Misidentifying the diagnostic modality and purpose of the test can lead to patient confusion regarding their prenatal care plan.
4. A positive GBS status is not an indication for an elective cesarean delivery; in fact, vaginal delivery is appropriate with proper antibiotic coverage. Cesarean birth is only indicated for GBS if the mother is already undergoing the procedure for other obstetric reasons before the rupture of membranes. Suggesting that GBS determines the mode of delivery is inaccurate, as antibiotics are highly effective at preventing transmission during a vaginal birth.
Test-taking strategy
- Analyze the Question: The client is asking for the scientific rationale for GBS screening at the end of the second trimester/start of the third trimester.
- Apply Clinical Knowledge:
- Pathogen Impact: Recall that GBS is primarily a threat to the neonate (sepsis, pneumonia), not just the mother.
- Timing and Method: Standard screening occurs between 36 0/7 and 37 6/7 weeks via swab.
- Goal of Care: The primary objective of identifying GBS is to trigger intrapartum antibiotic prophylaxis (IAP).
- Evaluate Options:
- Eliminate Choice 1: It incorrectly minimizes the fetal risk.
- Eliminate Choice 3: It incorrectly identifies the testing method and physiological target.
- Eliminate Choice 4: It incorrectly suggests a change in delivery route instead of a pharmacological intervention.
- Conclusion: Select the answer that connects the screening process to the prevention of neonatal infection via antibiotics.
Take home points
- The primary goal of GBS screening is to identify women who require intrapartum antibiotics to prevent early-onset neonatal sepsis.
- Screening is performed using a single swab of the distal vagina and the anorectum at 36 to 37 weeks gestation.
- Penicillin G is the preferred antibiotic for prophylaxis and must be administered intravenously during labor.
- If a woman has GBS bacteriuria during the current pregnancy or a previous infant with GBS disease, screening is bypassed and antibiotics are automatically indicated.
A nurse is reviewing preventive strategies for a client at risk of neonatal herpes simplex virus infection. Which measures are appropriate? Select all that apply
Explanation
Neonatal herpes simplex virus (HSV) infection is a high-mortality condition acquired through direct mucocutaneous contact with infectious secretions during parturition. Maternal pathogenesis involves viral replication in the squamous epithelium of the genital tract, followed by migration to the sacral ganglia where it establishes latency. Prevention strategies focus on suppressive pharmacotherapy and surgical avoidance of the infected birth canal to eliminate fetal viral inoculation.
Rationale for correct answers
1. Oral acyclovir or valacyclovir administration beginning at 36 weeks gestation effectively reduces the frequency of recurrent outbreaks at the time of delivery. This suppressive therapy lowers the rate of viral shedding, thereby decreasing the necessity for surgical delivery. Maintaining therapeutic serum levels during the final weeks of pregnancy is a standard clinical intervention to optimize the chances for a safe vaginal birth.
2. A cesarean delivery is the mandatory surgical intervention for clients presenting with visible genital vesicles or prodromal symptoms like burning or tingling at the onset of labor. Bypassing the birth canal prevents the fetus from contacting infectious fluids, which is the primary route for vertical transmission. This approach significantly lowers the risk of disseminated neonatal disease and permanent neurological impairment.
4. In clients with active genital lesions, frequent digital vaginal examinations are minimized to prevent the mechanical transfer of the virus from the external genitalia to the cervix and fetus. Reducing manual contact with vesicular fluid serves as an essential infection control measure during the first stage of labor. These restrictive protocols help maintain the integrity of the fetal environment until the delivery method is finalized.
Rationale for incorrect answers
3. While breastfeeding is generally encouraged, it is contraindicated if active herpetic lesions are present on the maternal breasts or nipples. Direct contact with a mammary vesicle can lead to neonatal inoculation and subsequent systemic infection. Clients must be educated to pump and discard milk from the affected breast until the lesions have completely crusted and healed.
5. There is currently no commercially available or approved HSV immune globulin for providing passive immunization to the newborn. Neonatal protection relies entirely on maternal suppressive therapy and appropriate obstetric management of the delivery route. Unlike other infections such as Hepatitis B, the prevention of HSV is focused on viral suppression rather than post-exposure antibody administration.
Test-taking strategy
- Analyze the Pathogen: The question focuses on Herpes Simplex Virus (HSV) and its prevention. The goal is to identify evidence-based strategies to protect the neonate.
- Evaluate the Timing of Interventions:
- Choice 1 identifies the correct window for suppressive therapy (36 weeks), which is a standard of care to prevent late-term outbreaks.
- Choice 2 identifies the correct mode of delivery (Cesarean) when lesions are present.
- Assess Infection Control:
- Choice 4 correctly applies the principle of minimizing exposure during labor by limiting examinations.
- Choice 3 is incorrect because it fails to account for lesion location on the breasts, which poses a direct threat during feeding.
- Differentiate Treatments:
- Choice 5 describes a treatment (Immune Globulin) that does not exist for HSV, making it a scientific distractor.
- Conclusion: Select the pharmacological and surgical interventions that align with ACOG guidelines for maternal-fetal HSV management.
Take home points
- Suppressive antiviral therapy starting at 36 weeks gestation reduces the risk of active HSV lesions at the time of delivery.
- The presence of active genital lesions or prodromal symptoms at the onset of labor is a definitive indication for cesarean birth.
- Fetal scalp electrodes and other invasive monitoring should be avoided in clients with a history of HSV to prevent iatrogenic viral entry.
- Breastfeeding is safe as long as no herpetic lesions are present on the breasts and proper hand hygiene is maintained.
A nurse is caring for a client with a primary parvovirus B19 infection at 18 weeks gestation. Which fetal complication should the nurse monitor for using ultrasound?
Explanation
Parvovirus B19 is a small, non-enveloped DNA virus that exhibits a specific tropism for erythroid progenitor cells in the fetal bone marrow. By binding to the P antigen on these cells, the virus inhibits erythropoiesis, leading to profound medullary aplasia and a shortened red blood cell lifespan. This cessation of hematopoiesis results in high-output cardiac failure and the accumulation of fluid in fetal serous cavities, a condition known as hydrops fetalis.
Rationale for correct answer
1. The primary complication of maternal parvovirus B19 infection is the development of fetal anemia due to the destruction of erythroblasts. As hemoglobin levels drop, the fetus develops high-output heart failure, leading to ascites, pleural effusions, and generalized edema. Serial ultrasound monitoring, including Middle Cerebral Artery (MCA) peak systolic velocity, is essential to detect hemodynamic changes and guide potential intrauterine blood transfusions.
Rationale for incorrect answers
2. Neural tube defects, such as spina bifida or anencephaly, result from the failure of the neural folds to fuse during the first 4 weeks of gestation. This developmental error is typically associated with folic acid deficiency or genetic factors rather than viral infections. Parvovirus B19 does not disrupt the early embryological processes involved in the formation of the spinal cord or cranium.
3. A cleft palate is a structural malformation caused by the failure of the palatine shelves to fuse during the late embryonic period. While certain environmental teratogens or genetic syndromes can cause orofacial clefts, viral pathogens like parvovirus B19 are not linked to these specific anatomical defects. Monitoring for this condition would not be a priority for a client with a confirmed viral viremia.
4. Limb hypoplasia is a pathognomonic finding associated with the congenital Varicella syndrome, occurring following a primary maternal chickenpox infection. Parvovirus B19 primarily targets the hematopoietic system and does not interfere with the mesenchymal development of the extremities. Consequently, assessing for shortened or cicatricial limbs is not indicated for the management of parvovirus B19.
Test-taking strategy
- Analyze the Scenario: The client has a confirmed primary parvovirus B19 infection at 18 weeks gestation, which is a high-risk period for fetal complications.
- Apply Pathophysiology:
- Recall that parvovirus B19 is also known as Fifth Disease and is famous for causing "slapped-cheek" rash in children.
- Identify its specific target: Erythroid progenitor cells (red blood cell precursors).
- Connect the target to the outcome: No red blood cells = Severe Anemia = Heart failure = Hydrops.
- Differentiate Pathogens:
- Rule out Choice 4: Limb hypoplasia = Varicella.
- Rule out Choice 2: Neural tube defects = Folate deficiency.
- Rule out Choice 3: Cleft palate = Structural/Genetic etiology.
- Conclusion: Select the option that reflects the hematologic destruction and subsequent fluid overload characteristic of this specific virus.
Take home points
- Parvovirus B19 infection during pregnancy can lead to fetal hydrops, characterized by fluid accumulation in at least two fetal compartments.
- The virus causes fetal anemia by infecting and lysing erythroid precursor cells in the fetal liver and bone marrow.
- Middle cerebral artery (MCA) Doppler ultrasound is the gold standard for non-invasively screening for fetal anemia in at-risk pregnancies.
- While many fetuses recover spontaneously, severe cases may require an intrauterine fetal blood transfusion to prevent fetal demise.
Exams on Infections (TORCH)
Custom Exams
Login to Create a Quiz
Click here to loginLessons
 Naxlex
Just Now
Naxlex
Just Now
Notes Highlighting is available once you sign in. Login Here.
Objectives
- To provide a comprehensive understanding of maternal infections classified under TORCH (Toxoplasmosis, Other [Syphilis, Varicella-zoster, Parvovirus B19], Rubella, Cytomegalovirus, Herpes Simplex Virus) and Group B Streptococcus (GBS).
- To understand the pathophysiology, maternal and fetal manifestations, diagnosis, management, and prevention strategies of these infections.
- To develop nursing competencies in assessing, educating, and caring for pregnant clients with TORCH infections or GBS.
- To enhance the nurse’s ability to anticipate neonatal complications associated with vertical transmission.
- To provide NCLEX-style practice questions for application of theoretical knowledge in clinical scenarios.
Introduction
- Maternal Infections During Pregnancy: These infections can cross the placenta (transplacental) or be transmitted during labor and delivery, potentially leading to congenital anomalies, fetal death, preterm birth, or neonatal sepsis.
- Vertical Transmission: Infection passed from the mother to fetus via the placenta, birth canal, or breast milk. Vertical transmission risk is highest in primary maternal infections acquired during pregnancy.
- Fetal Risks: Include congenital anomalies (e.g., microcephaly, intracranial calcifications, cataracts), growth restriction, prematurity, neurologic impairment, and neonatal death.
- Epidemiology: TORCH infections are worldwide, with varying prevalence depending on region, sanitation, vaccination coverage, and food safety practices.
- Definition of TORCH:
- T – Toxoplasmosis
- O – Other infections (Syphilis, Varicella-Zoster Virus, Parvovirus B19)
- R – Rubella
- C – Cytomegalovirus (CMV)
- H – Herpes Simplex Virus (HSV)
Nursing Insights:
- Early recognition and education of at-risk clients are essential for prevention.
- Nurses must understand the specific transmission routes to provide targeted interventions.
- Maternal immune status (IgG/IgM antibodies) is critical for assessing risk.
Toxoplasmosis
3.1 Etiology and Pathophysiology
- Causative Agent: Toxoplasma gondii, an obligate intracellular protozoan parasite.
- Transmission:
- Ingestion of tissue cysts from undercooked or raw meat (pork, lamb, venison).
- Contact with oocysts from cat feces.
- Transplacental transmission during acute maternal infection.
- Pathophysiology:
- After ingestion, tachyzoites disseminate via the bloodstream.
- Placental invasion occurs, leading to fetal infection.
- Fetal impact depends on gestational age at infection:
- 1st trimester: High risk of severe congenital defects, low transmission.
- 2nd trimester: Moderate risk, congenital anomalies may develop.
- 3rd trimester: High transmission rate, usually milder or asymptomatic neonatal disease.
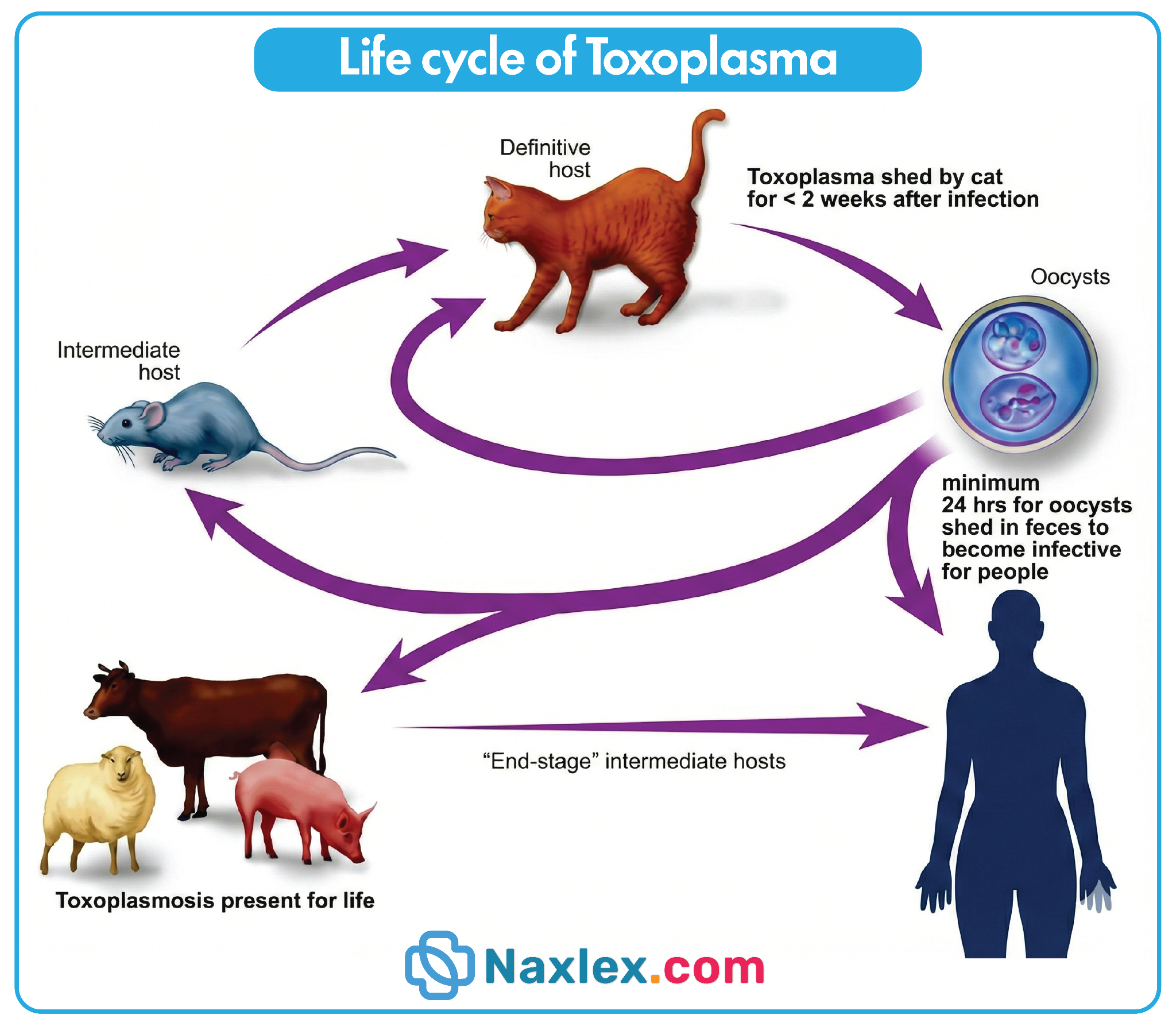
Nursing Insights:
- Educate clients to avoid handling cat litter or gardening without gloves.
- Emphasize proper cooking of meat and washing of fruits/vegetables.
- Be aware that asymptomatic maternal infection can still result in fetal infection.
3.2 Maternal Clinical Manifestations
- Often asymptomatic in immunocompetent women.
- Mild flu-like symptoms: fever, myalgia, fatigue, lymphadenopathy.
- Rarely, hepatosplenomegaly and rash may occur.
Nursing Insights:
- High index of suspicion is needed in pregnant clients with unexplained febrile illness.
- Counsel that mild maternal symptoms do not rule out significant fetal risk.
3.3 Fetal and Neonatal Manifestations
- Classic triad:
- Chorioretinitis – visual impairment, retinal lesions.
- Hydrocephalus – enlarged ventricles, macrocephaly.
- Intracranial calcifications – periventricular, diffuse.
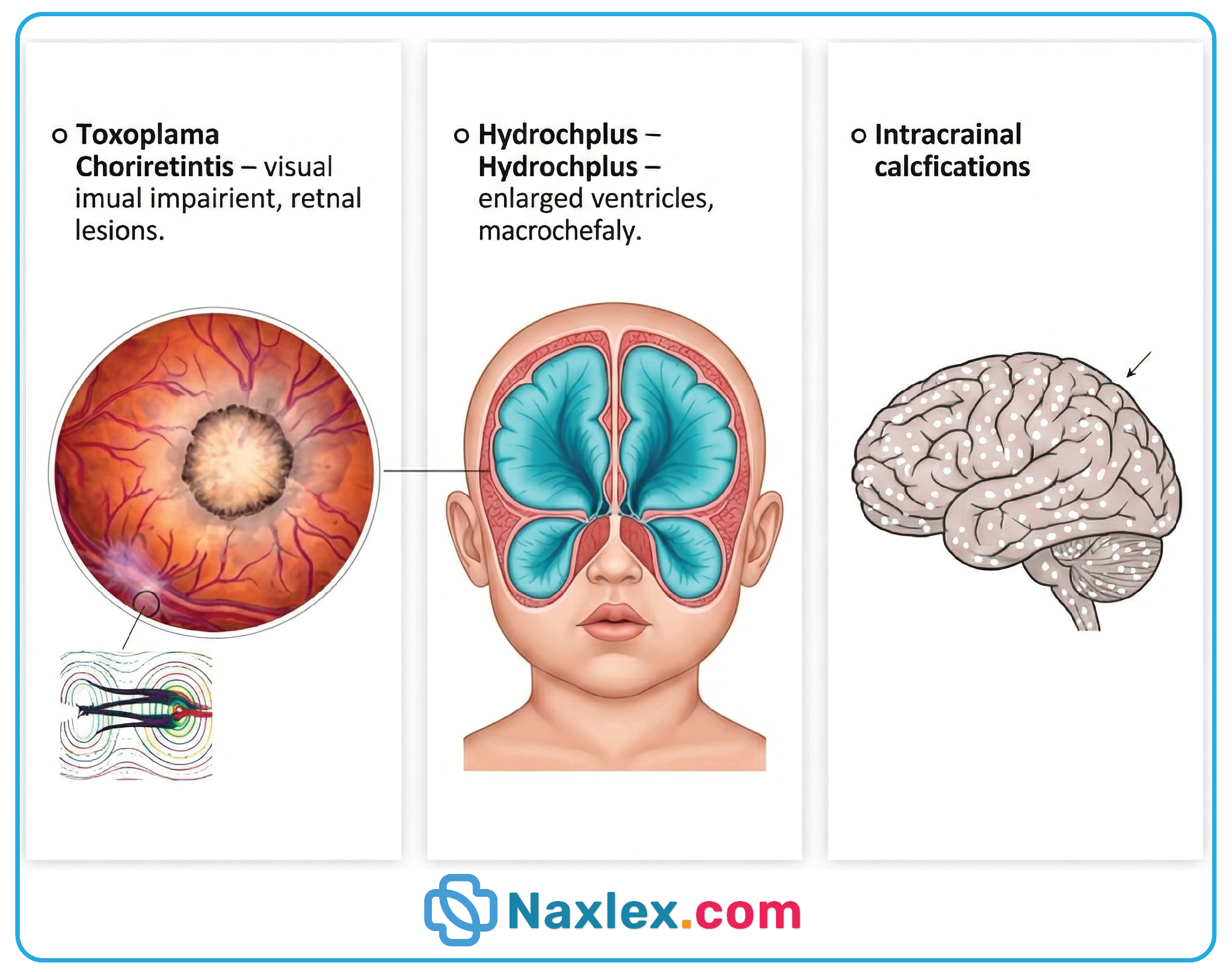
- Other manifestations:
- Jaundice, hepatosplenomegaly, thrombocytopenia.
- Neurologic deficits: seizures, developmental delay.
- Sensorineural hearing loss may appear later in childhood.
Nursing Insights:
- Monitor newborns for neurologic signs, vision/hearing deficits.
- Early identification allows prompt referral to pediatric specialists.
3.4 Diagnosis
- Maternal Diagnosis:
- Serologic testing for IgG and IgM antibodies to T. gondii.
- Avidity testing may determine timing of infection.
- Fetal Diagnosis:
- Amniocentesis for PCR testing of T. gondii DNA.
- Ultrasound for hydrocephalus, intracranial calcifications, hepatosplenomegaly.
- Neonatal Diagnosis:
- Serology (IgM positive indicates congenital infection).
- PCR testing of cord blood, urine, or cerebrospinal fluid.
Nursing Insights:
- Teach pregnant clients about the importance of serologic testing if exposed or symptomatic.
- Provide support during invasive procedures (amniocentesis).
- Collaborate with maternal-fetal medicine for high-risk pregnancies.
3.5 Treatment and Management
- Maternal Treatment:
- Spiramycin – reduces transplacental transmission (first-line for acute infection during pregnancy).
- Pyrimethamine + Sulfadiazine + Folinic acid – for confirmed fetal infection (after 1st trimester due to teratogenic risk).
- Neonatal Treatment:
- Pyrimethamine + Sulfadiazine + Folinic acid for 12 months.
- Symptom-based management: anticonvulsants for seizures, ophthalmologic therapy for chorioretinitis.
Nursing Insights:
- Monitor for drug toxicity (bone marrow suppression, hepatotoxicity).
- Educate parents about medication adherence and follow-up.
- Coordinate multidisciplinary care including neonatology, ophthalmology, and neurology.
3.6 Prevention Strategies
- Avoid handling cat litter or soil contaminated with cat feces.
- Cook meat to at least 71°C (160°F).
- Wash hands and kitchen utensils thoroughly after handling raw meat.
- Avoid unpasteurized milk or dairy products.
Nursing Insights:
- Reinforce hand hygiene and safe food practices at prenatal visits.
- Provide culturally sensitive guidance on dietary adjustments.
- Encourage family support to minimize exposure risk.
3.7 Nursing Insights
- Toxoplasmosis can be asymptomatic but cause severe fetal complications.
- Nurses must educate on primary prevention (food safety, hygiene, cat exposure).
- Early identification through serologic testing is essential.
- Nurses coordinate care between obstetric, pediatric, and infectious disease teams.
- Monitor for long-term neonatal complications: vision, hearing, neurodevelopment.
Rubella
5.1 Etiology and Pathophysiology
- Etiology
- Rubella is caused by the Rubella virus, a single-stranded RNA virus of the Togaviridae family.
- Transmission occurs via respiratory droplets from infected individuals, primarily through coughing or sneezing.
- Maternal infection during early pregnancy, especially the first trimester, poses the greatest risk for fetal teratogenesis.
- Pathophysiology
- Virus enters the maternal respiratory tract and replicates in the nasopharynx and regional lymph nodes.
- Maternal viremia develops within 5–7 days, allowing transplacental infection of the fetus.
- Fetal infection disrupts cellular mitosis, organogenesis, and vascular development, leading to congenital rubella syndrome (CRS).
- Severity of fetal outcomes is inversely proportional to gestational age at infection:
- First trimester: Highest risk of severe anomalies.
- Second trimester: Moderate risk, mainly hearing and ocular defects.
- Third trimester: Rarely causes major malformations but may result in postnatal infection.
Nursing Insights:
- Understanding gestational timing is essential; first trimester infection is catastrophic for the fetus.
- Nurses should recognize that maternal rubella infection may be asymptomatic or mild, but fetal effects are severe.
- Educate clients on respiratory precautions during rubella outbreaks to prevent maternal infection.
5.2 Maternal Clinical Manifestations
- Prodromal phase (1–5 days)
- Low-grade fever: 37.5–38.5°C (99.5–101.3°F)
- Malaise, headache, mild conjunctivitis
- Lymphadenopathy: postauricular, occipital, and cervical nodes
- Rash phase (3 days)
- Erythematous maculopapular rash begins on the face and spreads cephalocaudally to trunk and extremities.
- Pruritus may be mild.
- Rash resolves within 3 days.
- Other symptoms
- Arthralgia, especially in adult women
- Mild upper respiratory symptoms
Nursing Insights:
- Maternal symptoms may be subtle; high index of suspicion is required if the client is exposed during early pregnancy.
- Monitor for arthralgia and lymphadenopathy, which may cause maternal discomfort.
- Isolate infected clients to prevent nosocomial transmission, particularly in maternity units.
5.3 Fetal and Neonatal Manifestations (Congenital Rubella Syndrome, CRS)
- Classic Triad of CRS:
- Sensorineural deafness
- Ocular defects (cataracts, glaucoma, microphthalmia)
- Congenital heart defects (patent ductus arteriosus, pulmonary artery stenosis)
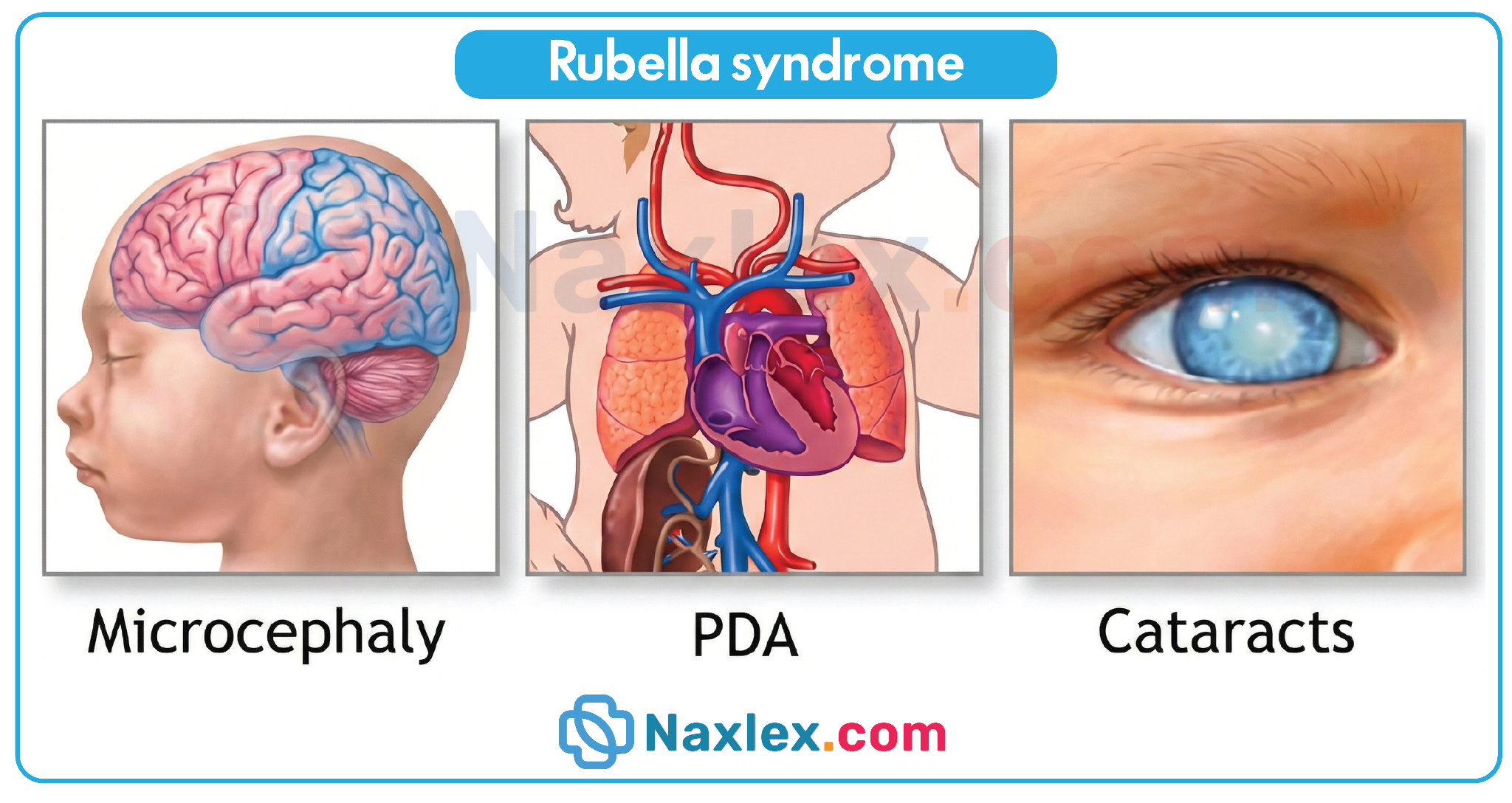
- Other manifestations:
- Microcephaly
- Hepatosplenomegaly
- Intrauterine growth restriction (IUGR)
- Thrombocytopenic purpura
- Radiolucent bone disease
- Developmental delay
Nursing Insights:
- Screen newborns of infected mothers for hearing loss, ocular defects, and cardiac anomalies immediately after birth.
- Educate parents about lifelong monitoring for neurodevelopmental delays.
- Early intervention programs may improve outcomes for affected infants.
5.4 Diagnosis
- Maternal Diagnosis:
- Serology: Detection of Rubella-specific IgM indicates recent infection; IgG indicates immunity or past infection.
- PCR testing of amniotic fluid may confirm fetal infection.
- Prenatal ultrasound: May detect IUGR, microcephaly, congenital heart defects, and cataracts.
- Neonatal Diagnosis:
- Positive Rubella IgM in newborn indicates intrauterine infection.
- Isolation of virus from nasopharyngeal swabs, urine, or blood confirms diagnosis.
- Serial audiologic and ophthalmologic testing for early detection of CRS complications.
Nursing Insights:
- Maternal serology should be routinely checked early in pregnancy to identify susceptible women.
- Prenatal ultrasound assists in planning delivery and neonatal interventions.
- Nurses should prepare parents for confirmatory neonatal testing and potential long-term follow-up.
5.5 Treatment and Management
- No specific antiviral therapy exists for rubella during pregnancy.
- Maternal care: Symptomatic management
- Antipyretics for fever
- Analgesics for arthralgia
- Hydration and rest
- Fetal/neonatal care:
- Multidisciplinary follow-up for cardiac, auditory, ocular, and developmental issues.
- Surgical interventions for congenital heart defects or cataracts if indicated.
- Early audiology referral for hearing impairment.
Nursing Insights:
- Emphasize supportive care for symptomatic mothers.
- Coordinate with pediatric cardiology, ophthalmology, and audiology for CRS management.
- Counsel parents on infection prevention for newborns with CRS who may shed the virus for up to 1 year.
5.6 Prevention Strategies
- Vaccination:
- MMR (Measles, Mumps, Rubella) vaccine prior to conception is the most effective preventive strategy.
- Live attenuated vaccine is contraindicated during pregnancy.
- Screening and counseling:
- Check Rubella IgG antibodies in early prenatal visits.
- Educate non-immune women to avoid pregnancy for at least 28 days post-vaccination.
- Infection control:
- Avoid contact with infected individuals during outbreaks.
- Implement standard respiratory precautions in healthcare settings.
Nursing Insights:
- Nurses must educate women of childbearing age about preconception vaccination.
- Monitor seronegative pregnant women for potential exposure.
- Promote community awareness to reduce rubella prevalence.
5.7 Nursing Insights Summary
- Early gestation rubella infection poses highest fetal risk.
- Maternal symptoms are often mild or subclinical, requiring vigilance.
- Newborns should be screened for CRS manifestations immediately after birth.
- Prevention through preconception vaccination is paramount.
- Coordinate care with a multidisciplinary team for affected neonates.
- Educate parents about virus shedding and infection control.
Cytomegalovirus
7.1 Etiology and Pathophysiology
- Cytomegalovirus (CMV) is a double-stranded DNA virus belonging to the Herpesviridae family.
- It is the most common congenital viral infection, affecting 0.5–2.5% of all live births worldwide.
- Transmission:
- Vertical (transplacental) transmission during primary maternal infection is most dangerous in the first trimester.
- Horizontal via body fluids: urine, saliva, breast milk, blood, sexual contact.
- Pathophysiology:
- Maternal infection triggers viral replication in the placenta, leading to placental inflammation and direct fetal tissue infection.
- CMV exhibits latency in mononuclear cells, allowing reactivation in immunocompromised or seropositive mothers.
- Fetal damage occurs via cytopathic effect, causing apoptosis of infected cells, particularly in the central nervous system (CNS), liver, and reticuloendothelial system.
Nursing Insights: Etiology and Pathophysiology
- CMV infection is often asymptomatic in healthy pregnant women, making early detection difficult.
- Awareness of high-risk exposures (childcare settings, toddlers with frequent saliva contact) is critical.
- Reactivation in seropositive women usually causes less severe fetal disease, but primary infections are associated with significant neurodevelopmental outcomes.
7.2 Maternal Clinical Manifestations
- Most maternal infections are asymptomatic.
- When symptoms occur, they are non-specific and flu-like, including:
- Fever (≥38°C / 100.4°F)
- Malaise and fatigue
- Myalgia and arthralgia
- Sore throat and lymphadenopathy
- Rarely: hepatosplenomegaly or mild hepatitis.
Nursing Insights: Maternal Manifestations
- Do not rely solely on symptoms to detect CMV; lab testing is essential.
- Educate clients that exposure does not always result in illness, but risk to fetus exists during primary infection.
- Emphasize hygiene measures as the primary prevention in asymptomatic cases.
7.3 Fetal and Neonatal Manifestations
- Congenital CMV manifestations:
- Sensorineural hearing loss (most common long-term complication)
- Microcephaly and intracranial calcifications
- Chorioretinitis and visual impairment
- Hepatosplenomegaly
- Thrombocytopenia and petechiae
- Jaundice and elevated liver enzymes
- Intrauterine growth restriction (IUGR)
- Timing of infection:
- First trimester: higher risk of severe CNS anomalies.
- Third trimester: often subclinical but can still cause hearing loss.
Nursing Insights: Fetal/Neonatal Manifestations
- Newborn screening should include urine PCR within 3 weeks of birth.
- Monitor hearing and vision longitudinally, as complications may develop after birth.
- Recognize that asymptomatic neonates may still develop delayed sensorineural hearing loss.
7.4 Diagnosis
- Maternal diagnosis:
- Serology: IgM (recent infection), IgG (past exposure), and IgG avidity testing to determine timing of infection.
- Fetal diagnosis:
- Amniocentesis for PCR detection of CMV DNA in amniotic fluid (optimal at ≥21 weeks gestation and ≥6 weeks post-maternal infection).
- Neonatal diagnosis:
- Urine or saliva PCR within first 3 weeks of life.
- CBC and liver function tests to assess systemic involvement.
- Neuroimaging (cranial ultrasound, MRI) for structural CNS anomalies.
Nursing Insights: Diagnosis
- Early detection is crucial to provide parental counseling and early antiviral therapy.
- Inform clients that prenatal CMV screening is not routinely recommended in all settings but is considered for high-risk exposures.
7.5 Treatment and Management
- Maternal treatment:
- Supportive care for mild symptoms (hydration, antipyretics).
- No FDA-approved antiviral treatment during pregnancy; experimental use of hyperimmune globulin in some settings.
- Neonatal treatment:
- Ganciclovir or valganciclovir for symptomatic neonates (CNS involvement, hepatitis, thrombocytopenia) to improve hearing and developmental outcomes.
- Monitor CBC and liver/kidney function during therapy.
Nursing Insights: Treatment
- Coordinate care with pediatric infectious disease and audiology teams.
- Educate parents on adherence to antiviral therapy and monitoring.
- Recognize that asymptomatic neonates typically do not receive antiviral treatment but require long-term follow-up.
7.6 Prevention Strategies
- Hygiene practices:
- Frequent handwashing after diaper changes or contact with saliva.
- Avoid sharing eating utensils with young children.
- Pregnancy counseling:
- Women who are seronegative should minimize exposure to high-risk environments (daycare, toddlers).
- No current vaccine available; research ongoing.
Nursing Insights: Prevention
- Prevention relies entirely on behavioral modification; early education is essential.
- Teach pregnant clients that even asymptomatic contact with CMV can lead to fetal infection.
- Reinforce that CMV cannot be completely eliminated from the environment, but risk reduction is feasible.
7.7 Nursing Insights
- Always consider CMV in newborns with unexplained hearing loss, IUGR, or neurological deficits.
- Prenatal counseling is crucial for seronegative women in high-exposure occupations (e.g., daycare, healthcare).
- Long-term follow-up includes hearing assessments, developmental screenings, and early intervention services.
- CMV infection has no routine universal prevention via vaccination, making client education and infection control paramount.
Herpes Simplex Virus
9.1 Etiology and Pathophysiology
- Causative Agent: Herpes Simplex Virus Type 1 (HSV-1) and Type 2 (HSV-2). While HSV-2 has traditionally been associated with genital infections, HSV-1 is increasingly identified as a cause of primary genital herpes.
- Mechanism of Infection: The virus is highly contagious and is transmitted through direct contact of mucous membranes or non-intact skin with viral lesions or infected secretions.
- Latency: After the initial infection, the virus migrates along sensory neurons to the dorsal root ganglia, where it remains latent. It can reactivate periodically, leading to viral shedding and recurrent lesions.
- Vertical Transmission: Transmission to the fetus or neonate occurs through three main routes:
- In Utero (Rare): Transplacental transmission (5% of cases).
- Intrapartum (Most Common): Direct contact with infected maternal secretions in the birth canal during delivery (85% of cases).
- Postnatal: Contact with infected caregivers or breast lesions (10% of cases).
- Risk Factors: The risk of neonatal infection is significantly higher (30% to 50%) during a primary maternal infection near the time of delivery compared to a recurrent episode (less than 1% to 3%).
9.2 Maternal Clinical Manifestations
- Primary Infection: * Often characterized by severe, painful, vesicular, or ulcerative lesions in the perineal area.
- Systemic symptoms include fever, malaise, myalgia, and inguinal lymphadenopathy.
- Dysuria and vaginal discharge may be present.
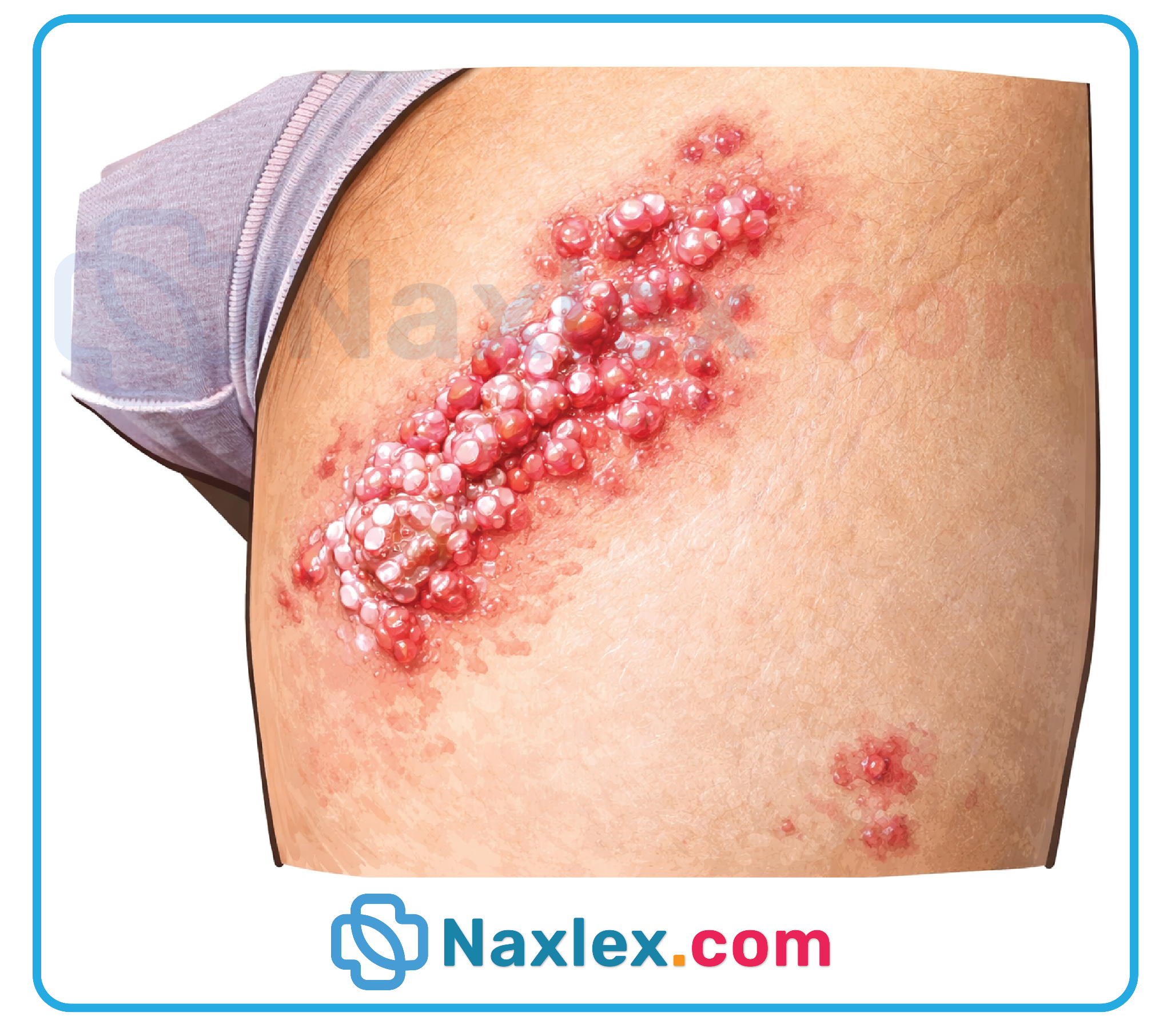
- Recurrent Infection: * Usually less severe than the primary episode.
- Prodromal symptoms such as tingling, itching, or burning at the site of future lesions often precede the outbreak.
- Asymptomatic Shedding: The client may shed the virus even in the absence of visible lesions, which still poses a risk for transmission.
9.3 Fetal and Neonatal Manifestations
- In Utero Infection: Can result in microcephaly, chorioretinitis, and cutaneous scarring.
- Neonatal Herpes Classification:
- Skin, Eye, and Mouth (SEM) Disease: Characterized by vesicular lesions on the skin or around the eyes and mouth. This has the best prognosis but can progress if untreated.
- Central Nervous System (CNS) Disease: Presents with seizures, lethargy, irritability, and poor feeding. It may occur with or without skin lesions.
- Disseminated Disease: The most severe form, involving multiple organs (liver, lungs, adrenal glands). It carries a high mortality rate and mimics neonatal sepsis.
9.4 Diagnosis
- Maternal Diagnosis:
- Viral Culture: The gold standard for symptomatic lesions; it is most sensitive during the vesicular stage.
- PCR (Polymerase Chain Reaction): Highly sensitive and often preferred for detecting viral DNA in lesions or cerebrospinal fluid (CSF).
- Type-Specific Serology: To differentiate between HSV-1 and HSV-2 and to determine if an infection is primary or recurrent.
- Neonatal Diagnosis: Swabs of the conjunctiva, oropharynx, and any skin lesions, as well as CSF analysis via PCR.
9.5 Treatment and Management
- Maternal Therapy: * Acyclovir, Valacyclovir, or Famciclovir: These antiviral agents are used to shorten the duration of symptoms and suppress viral shedding.
- Suppressive Therapy: Recommended starting at 36 weeks gestation for all pregnant clients with a history of genital herpes to reduce the likelihood of an active outbreak at the time of delivery.
- Neonatal Therapy: * Intravenous Acyclovir: Should be initiated immediately for any newborn suspected of having HSV infection. Treatment duration depends on the disease classification (14 days for SEM, 21 days for CNS or disseminated).
9.6 Delivery Considerations
- Vaginal Delivery: Indicated if there are no active lesions or prodromal symptoms (tingling/pain) at the onset of labor.
- Cesarean Delivery: Recommended for clients with active genital lesions or prodromal symptoms at the time of labor to minimize the risk of vertical transmission.
- Avoidance of Scalp Electrodes: Internal fetal monitoring and vacuum/forceps should be avoided in clients with a history of HSV if possible, as skin trauma increases the risk of viral entry.
9.7 Prevention Strategies
- Education on the use of condoms and the importance of avoiding sexual contact during active outbreaks.
- Routine screening of all pregnant clients for a history of genital herpes.
- Strict hand hygiene for caregivers with orolabial herpes ("cold sores") to prevent postnatal transmission to the newborn.
9.8 Nursing Insights
- The nurse must perform a thorough physical assessment of the perineum of any client in labor with a history of HSV to check for "hidden" lesions.
- During the third trimester, the nurse should verify that the client has received a prescription for suppressive antiviral therapy (usually Acyclovir) and emphasize the importance of compliance.
- In the delivery room, the nurse should explicitly document the absence of lesions if a vaginal birth is planned to ensure a clear legal and clinical record.
- For the newborn, the nurse must monitor for signs of sepsis-like illness or new-onset skin vesicles, reporting these immediately to the primary health care provider.
- If the mother has an active oral herpes lesion (cold sore), the nurse should instruct her to wear a mask when holding the infant and to avoid kissing the infant until the lesion is completely crusted over.
Group B Streptococcus
11.1 Etiology and Pathophysiology
- Etiology:
- Group B Streptococcus (GBS), also known as Streptococcus agalactiae, is a gram-positive, beta-hemolytic bacterium.
- It colonizes the lower gastrointestinal and genitourinary tracts in approximately 15–30% of pregnant women.
- Colonization can be asymptomatic in the mother but can cause serious neonatal infection.
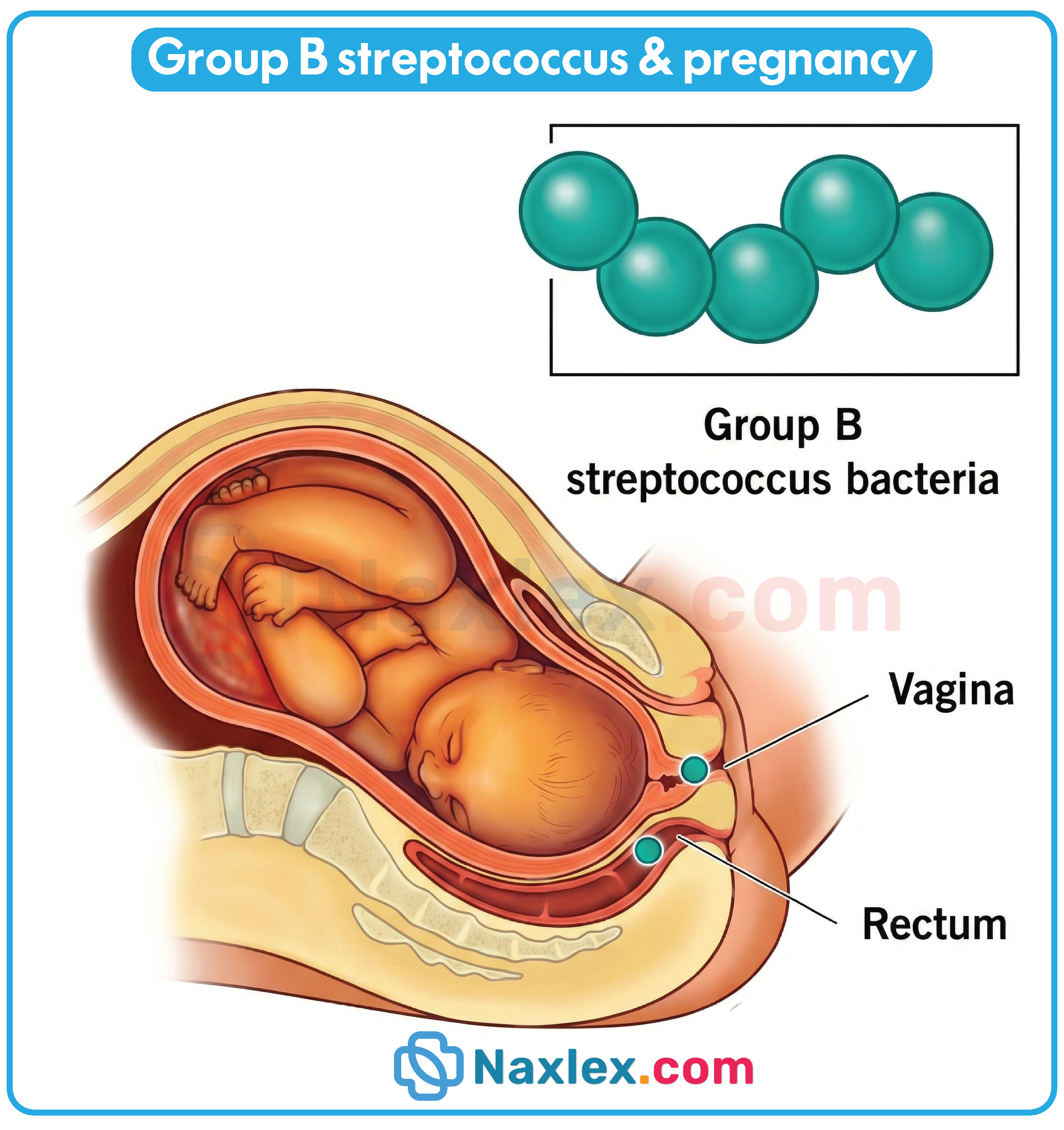
- Pathophysiology:
- Maternal colonization can lead to vertical transmission to the fetus during labor and delivery.
- GBS can ascend from the vagina or rectum into the amniotic sac, leading to:
- Preterm labor
- Chorioamnionitis
- Intrauterine fetal infection
- Early-onset neonatal GBS disease (occurring within the first 7 days of life) often manifests as:
- Sepsis
- Pneumonia
- Meningitis
- Late-onset disease (7–90 days postpartum) usually presents as meningitis or bacteremia without maternal colonization risk factors.
- Risk Factors for Neonatal GBS Infection:
- Preterm birth (<37 weeks)
- Prolonged rupture of membranes (>18 hours)
- Maternal intrapartum fever (>38°C / 100.4°F)
- Previous infant with GBS disease
- GBS bacteriuria during current pregnancy
- Multiple gestation
Nursing Insights:
- Nurses must recognize that GBS colonization is often asymptomatic; maternal symptoms may be absent.
- Understanding the timeline of early- vs. late-onset GBS disease informs surveillance and neonatal care.
- Nurses should educate mothers on risk factors for vertical transmission.
11.2 Maternal Clinical Manifestations
- Most colonized mothers are asymptomatic.
- Symptomatic infections may include:
- Urinary tract infection (dysuria, urgency, frequency)
- Chorioamnionitis (maternal fever, uterine tenderness, tachycardia)
- Bacteremia (rare, may present with fever, malaise)
- GBS bacteriuria indicates heavy colonization and increases risk of neonatal infection.
Nursing Insights:
- Nurses must screen for maternal risk factors at prenatal visits.
- Maternal fever, tachycardia, and uterine tenderness in labor should prompt evaluation for GBS and other infections.
- Documenting colonization and history of GBS is critical for intrapartum prophylaxis planning.
11.3 Fetal and Neonatal Manifestations
- Early-Onset GBS Disease (first 7 days):
- Sepsis (temperature instability, apnea, hypotension)
- Respiratory distress (tachypnea, grunting, nasal flaring)
- Pneumonia (hypoxia, cyanosis)
- Meningitis (less common in early-onset)
- Late-Onset GBS Disease (7–90 days):
- Bacteremia without focus
- Meningitis
- Occasional soft tissue infections
- Complications:
- Long-term neurodevelopmental delay
- Hearing loss (after meningitis)
- Death if untreated early
Nursing Insights:
- Early recognition of neonatal signs is essential for prompt antibiotic therapy.
- Nurses must monitor temperature, respiratory status, and neurologic signs in at-risk neonates.
- Communication with neonatal care team ensures timely intervention.
11.4 Screening and Diagnosis
- Screening Recommendations:
- Universal rectovaginal culture at 36–37 weeks gestation.
- Alternative: risk-based screening if preterm labor occurs before cultures are obtained.
- Diagnosis:
- Maternal colonization confirmed via culture.
- Neonatal infection confirmed with:
- Blood culture
- Cerebrospinal fluid (CSF) analysis
- Urine culture (occasionally)
- Additional Considerations:
- Positive GBS bacteriuria anytime in pregnancy indicates need for intrapartum prophylaxis.
- Rapid antigen testing may be used intrapartum if culture results unavailable.
Nursing Insights:
- Nurses must ensure proper collection technique: swab of vaginal introitus and rectum.
- Documentation of maternal GBS status is crucial for labor planning.
- Nurses should educate clients about the purpose of GBS screening.
11.5 Treatment and Intrapartum Prophylaxis
- Maternal Treatment:
- Penicillin G is the first-line intrapartum antibiotic prophylaxis (IAP).
- Dose: 5 million units IV initially, then 2.5 million units IV every 4 hours until delivery.
- Alternative for penicillin-allergic clients:
- Cefazolin if low risk for anaphylaxis
- Clindamycin or vancomycin if high risk and strain susceptible
- Penicillin G is the first-line intrapartum antibiotic prophylaxis (IAP).
- Timing:
- Administer ≥4 hours before delivery for optimal fetal protection.
- Neonatal Treatment:
- Empiric IV antibiotics (ampicillin + gentamicin) if early-onset sepsis suspected.
- Supportive care: oxygen, fluids, vasopressors if septic shock develops.
Nursing Insights:
- Nurses must monitor for maternal allergic reactions to antibiotics.
- Accurate timing of IAP is essential for neonatal prophylaxis.
- Documenting start and stop times, maternal response, and labor progression is crucial.
11.6 Prevention Strategies
- Universal prenatal screening at 36–37 weeks.
- Administer intrapartum prophylactic antibiotics for:
- Positive culture
- GBS bacteriuria during pregnancy
- Previous infant with GBS disease
- Educate clients on:
- Hygiene to reduce colonization risk
- Prompt reporting of fever or urinary symptoms in late pregnancy
- Avoid elective vaginal delivery in GBS-positive clients if membranes rupture >18 hours without prophylaxis.
Nursing Insights:
- Nurses play a key role in educating clients about GBS prevention.
- Ensuring compliance with screening and prophylaxis guidelines reduces neonatal morbidity and mortality.
- Coordination with obstetric and neonatal teams enhances early detection.
11.7 Nursing Insights
- Documentation: Accurate recording of maternal GBS status, allergies, and antibiotic administration.
- Monitoring: Vital signs, labor progression, neonatal status, early sepsis signs.
- Education: Clear communication with clients about GBS, risks, and preventive measures.
- Collaboration: Work with primary health care providers and neonatal teams for at-risk newborns.
- Early Detection: Recognize early signs of neonatal sepsis (lethargy, respiratory distress, temperature instability).
Summary
- TORCH Infections Overview: * TORCH is an acronym for a group of infections that can cross the placenta and cause significant fetal morbidity or mortality.
- Toxoplasmosis: Caused by the protozoan Toxoplasma gondii. Primary transmission is via raw/undercooked meat or contact with infected cat feces. Classic triad includes chorioretinitis, hydrocephalus, and intracranial calcifications.
- Other (Syphilis, Varicella, Parvovirus B19): Syphilis (Treponema pallidum) causes multisystem failure; Varicella causes limb hypoplasia; Parvovirus B19 causes fetal hydrops and severe anemia.
- Rubella: A viral infection that, if contracted in the first trimester, leads to Congenital Rubella Syndrome (CRS), characterized by deafness, cataracts, and cardiac defects.
- Cytomegalovirus (CMV): The most common congenital viral infection. Often asymptomatic in the mother but leads to sensorineural hearing loss and microcephaly in the neonate.
- Herpes Simplex Virus (HSV): Primarily transmitted during birth through an active lesion. Causes Skin, Eye, and Mouth (SEM) disease, CNS disease, or disseminated infection.
- Group B Streptococcus (GBS):
- Gram-positive bacteria found in the vaginal/rectal flora.
- Screening: Universal screening at 36 0/7 to 37 6/7 weeks gestation.
- Prophylaxis: Intrapartum Antibiotic Prophylaxis (IAP) with Penicillin G is mandatory for positive screens, GBS bacteriuria, or unknown status with risk factors (fever >38.0°C/100.4°F or ROM >18 hours).
- Nursing Insights:
- The nurse must prioritize screening history and maternal education regarding preventive hygiene (e.g., handwashing, cooking meat, avoiding cat litter).
- Timely administration of IAP (at least 4 hours prior to delivery) is the gold standard for preventing early-onset neonatal GBS sepsis.
COMPARISON TABLE: TORCH AND GBS
|
Infection |
Transmission |
Maternal Symptoms |
Fetal/Neonatal Manifestations |
Prevention/Management |
|
Toxoplasmosis |
Cat feces, undercooked meat |
Usually asymptomatic |
Chorioretinitis, hydrocephalus, intracranial calcifications |
Avoid cat litter, Spiramycin, hygiene |
|
Rubella |
Respiratory droplets |
Rash, low-grade fever |
Sensorineural deafness, cataracts, PDA |
Pre-pregnancy MMR vaccine |
|
CMV |
Body fluids |
Often asymptomatic |
Hearing loss, microcephaly |
Hand hygiene, supportive care |
|
HSV |
Genital lesions |
Painful vesicles, dysuria |
Skin vesicles, encephalitis |
Cesarean if lesions, Acyclovir |
|
GBS |
Vaginal/rectal colonization |
Often asymptomatic |
Early-onset sepsis, meningitis |
IAP with Penicillin G, culture at 36–37 wks |
Naxlex
Videos
Login to View Video
Click here to loginTake Notes on Infections (TORCH)
This filled cannot be empty
Join Naxlex Nursing for nursing questions & guides! Sign Up Now

