For which of the following reasons does a chloride ion have a negative charge?
It gained an electron.
It lost an electron.
It lost a proton.
It gained a proton.
The Correct Answer is A
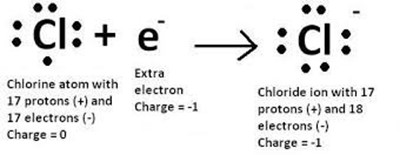
A chloride ion has a negative charge because it gained an electron. When an atom gains an electron, it becomes negatively charged because it now has more electrons than protons. In the case of a chloride ion, the neutral chlorine atom gains an electron to become a negatively charged chloride ion.
The other options are not correct because they do not result in a negative charge. Losing an electron would result in a positive charge. Losing or gaining a proton would change the identity of the atom and is not related to the formation of a chloride ion.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
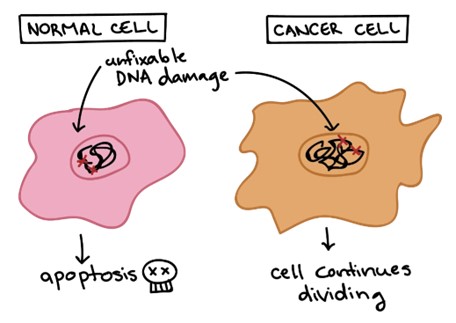
A genetic mutation that results in the uncontrolled division of a single cell within the body describes cancer. Cancer is a disease of uncontrolled cell division. Its development and progression are usually linked to a series of changes in the activity of cell cycle regulators. In most cases, these changes in activity are due to mutations in the genes that encode cell cycle regulator proteins.
The other options are not correct because they do not accurately describe a genetic mutation that results in the uncontrolled division of a single cell within the body. Gene therapy, stem cells, and translation are not processes that result in uncontrolled cell division.
Correct Answer is A
Explanation
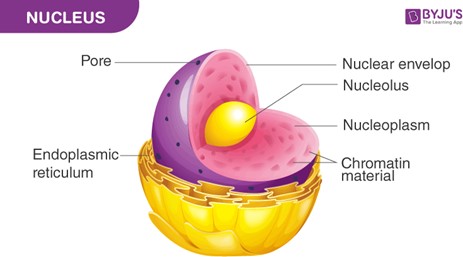
Nuclei. Genetic information describing the characteristics of an organism is found in the nuclei of its cells. The nucleus contains the organism's DNA, which carries the genetic information that determines its traits.
B. Membranes are structures that surround and enclose cells and organelles, but they do not contain genetic information.
C. Cilia are hair-like structures that protrude from the surface of some cells and are involved in movement, but they do not contain genetic information.
D. Ribosomes are organelles that are involved in protein synthesis, but they do not contain genetic information.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
