A nurse is monitoring the fluid replacement of a client who has sustained burns. The nurse should administer which of the following fluids in the first 24 hr following a burn injury?
Dextrose 5% in 0.9% sodium chloride
Dextrose 5% in water
0.9% sodium chloride
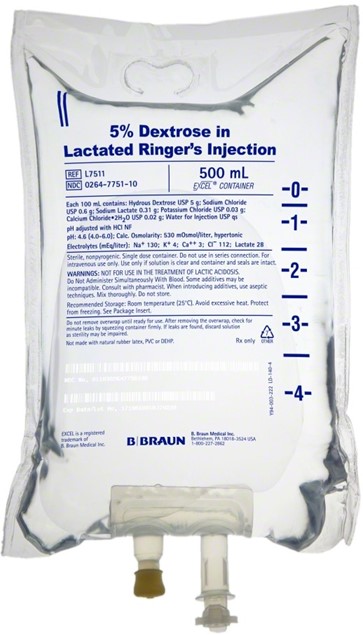
Lactated Ringer's
The Correct Answer is D
Choice A Reason: This is incorrect because dextrose 5% in 0.9% sodium chloride is a hypertonic solution that can cause fluid overload and pulmonary edema in a client who has burns.
Choice B Reason: This is incorrect because dextrose 5% in water is a hypotonic solution that can cause fluid shifts from the intravascular space to the interstitial space, resulting in hypovolemia and hypotension in a client who has burns.
Choice C Reason: This is incorrect because 0.9% sodium chloride is an isotonic solution that can cause hypernatremia and hyperchloremia in a client who has burns, as the fluid loss from burns is greater than the sodium loss.
Choice D Reason: This is correct because lactated Ringer's is an isotonic solution that contains electrolytes similar to plasma, such as sodium, potassium, calcium, and chloride. It also contains lactate, which is converted to bicarbonate in the liver and helps correct the metabolic acidosis that occurs in a client who has burns.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is C
Explanation
Choice A Reason: This is incorrect because a client who is post-CABG and has high cholesterol is not in immediate danger, as atorvastatin is a long-term medication that lowers cholesterol and prevents cardiovascular complications.
Choice B Reason: This is incorrect because a client who has pneumonia and a slightly elevated WBC count is not in immediate danger, as piperacillin is an antibiotic that treats bacterial infections.
Choice C Reason: This is correct because a client who has renal failure and a high serum potassium level is in immediate danger, as sodium polystyrene sulfonate is an emergency medication that lowers potassium and prevents cardiac arrhythmias.
Choice D Reason: This is incorrect because a client who has anemia and a mild hemoglobin deficiency is not in immediate danger, as epoetin alfa is a long-term medication that stimulates red blood cell production and improves oxygen delivery.
Correct Answer is D
Explanation
Choice A Reason: This is incorrect because magnesium 2.5 mEq/L is a normal value and does not indicate an increased risk of AKI. Magnesium is an electrolyte that plays a role in muscle and nerve function, blood pressure regulation, and energy production. The normal range for magnesium is 1.5 to 2.5 mEq/L.
Choice B Reason: This is incorrect because serum osmolality 290 mOsm/kg H2O is a normal value and does not indicate an increased risk of AKI. Serum osmolality is a measure of the concentration of solutes in the blood, such as sodium, glucose, and urea. The normal range for serum osmolality is 275 to 295 mOsm/kg H2O.
Choice C Reason: This is incorrect because blood urea nitrogen (BUN) 20 mg/dL is a normal value and does not indicate an increased risk of AKI. BUN is a measure of the amount of urea, a waste product of protein metabolism, in the blood. The normal range for BUN is 7 to 20 mg/dL.
Choice D Reason: This is correct because serum creatinine 1.8 mg/dL is an elevated value and indicates an increased risk of AKI. Creatinine is a waste product of muscle metabolism that is filtered by the kidneys. The normal range for serum creatinine is 0.6 to 1.2 mg/dL for women and 0.7 to 1.3 mg/dL for men. An increase in serum creatinine indicates a decrease in kidney function and glomerular filtration rate (GFR).
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
