Reference Range: 1329545.
A client receiving mechanical ventilation has a pH of 7.26, PaCO2 of 68 mm Hg, and a PaO2 of 92 mm Hg. Which intervention should the nurse implement?
Decrease expiratory flow time.
Decrease expiratory pressure.
Increase rate of ventilation.
Increase ventilator tidal volume.
The Correct Answer is B
Choice A rationale:
Decreasing expiratory flow time is not the appropriate intervention in this case. The client's pH and PaCO2 levels suggest respiratory acidosis, which indicates inadequate ventilation. Increasing expiratory flow time might exacerbate the acidosis by reducing ventilation.
Choice C rationale:
Increasing the rate of ventilation (respiratory rate) is a potential intervention to improve the client's acid-base balance. However, it should be done cautiously and under medical supervision to avoid respiratory alkalosis. It is not the first-line intervention in this scenario.
Choice D rationale:
Increasing the ventilator tidal volume may help improve ventilation, but it should also be done under medical guidance to prevent barotrauma. It is not the initial intervention to address the client's respiratory acidosis.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Choice A rationale:
The client being the oldest of their siblings is not a contributing factor related to the development of conduct disorder. Family dynamics such as birth order may have some influence on personality traits, but they are not a primary factor in the development of conduct disorder.
Choice B rationale:
The fact that the client's father lives in the client's home is a family dynamic, but it does not necessarily contribute to the development of conduct disorder. Other factors related to parenting style, communication, and family interactions play a more significant role in the development of conduct disorder.
Choice C rationale:
The client's mother having asthma is a medical condition and not a family dynamic that directly contributes to the development of conduct disorder. Conduct disorder is more closely associated with social, environmental, and psychological factors.
Choice D rationale:
The presence of several siblings in the family dynamic can contribute to the development of conduct disorder. Increased family size can lead to competition for attention and resources, which may affect the child's behavior and interactions. Sibling relationships and family dynamics are crucial in shaping a child's behavior and psychological well-being.
Correct Answer is B
Explanation
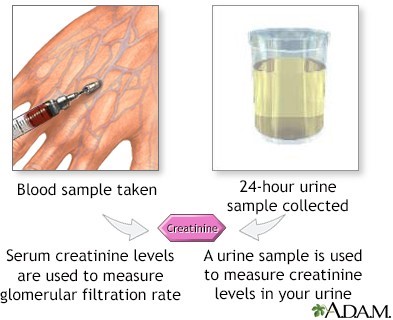
Serum creatinine is the most important laboratory value to review before administering an antibiotic that can cause nephrotoxicity. Nephrotoxicity is an alteration in the function of the kidney due to exposure to certain drugs or toxins.
It can be assessed by measuring the glomerular filtration rate (GFR), which is the rate of clearance of a substance from the blood by the kidneys. Serum creatinine is a waste product of muscle metabolism that is freely filtered by the glomeruli and not reabsorbed or secreted by the tubules.
Therefore, it is a reliable indicator of GFR and renal function. An increase in serum creatinine indicates a decrease in GFR and renal function, which may be caused by nephrotoxic drugs.
The other laboratory values are not directly related to nephrotoxicity or GFR:
- Serum calcium: This may be affected by renal function, but it is not a sensitive or specific marker of nephrotoxicity. It may be altered by other factors such as vitamin D, parathyroid hormone, and bone metabolism.
- Hemoglobin and hematocrit: These may be affected by renal function, but they are not sensitive or specific markers of nephrotoxicity. They may reflect the erythropoietin production by the kidneys, which stimulates red blood cell production in the bone marrow. However, they may also be influenced by other factors such as blood loss, hydration status, and iron deficiency.
- White blood cell count (WBC): This is not related to nephrotoxicity or GFR. It may reflect the presence of infection or inflammation, which may be a cause or a consequence of renal impairment, but it is not a direct measure of renal function.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
