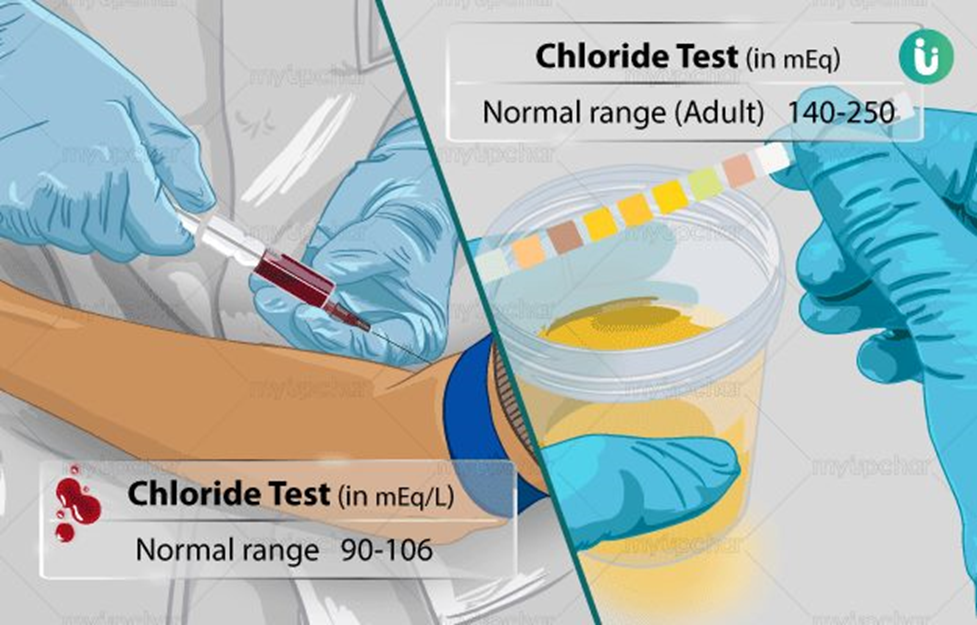
What is the normal range of serum chloride level in adults?
95-110 mg/dL
10-120 mEq/L
96-106 mEq/L
1.8-2.6 mEq/L
The Correct Answer is C
Choice A reason: This is incorrect because 95-110 mg/dL is the normal range of serum phosphorus level in adults, not chloride. Phosphorus is an electrolyte that is involved in energy metabolism, acid-base balance, and bone formation.
Choice B reason: This is incorrect because 10-120 mEq/L is not a realistic range for any electrolyte level in the blood. The units of mEq/L indicate the concentration of ions, not the mass of the substance. The normal range of serum chloride level in adults is expressed in mEq/L, not mg/dL.
Choice C reason: This is correct because 96-106 mEq/L is the normal range of serum chloride level in adults. Chloride is an electrolyte that is important for fluid balance, acid-base balance, and nerve transmission.
Choice D reason: This is incorrect because 1.8-2.6 mEq/L is the normal range of serum magnesium level in adults, not chloride. Magnesium is an electrolyte that is important for muscle and nerve function, as well as enzyme activity.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Choice A reason: Intracellular to the extracellular fluid shift is not the cause of low blood pressure in this case. This fluid shift occurs when the cells lose water due to osmosis, such as in dehydration or hypernatremia.
Choice B reason: Interstitial to intravascular fluid shift is not the cause of low blood pressure in this case. This fluid shift occurs when the fluid moves from the tissue spaces to the blood vessels, such as in hypovolemia or hypotonic fluid administration.
Choice C reason: Interstitial to the intracellular fluid shift is not the cause of low blood pressure in this case. This fluid shift occurs when the fluid moves from the tissue spaces to the cells, such as in overhydration or hyponatremia.
Choice D reason: Intravascular to the interstitial fluid shift is the cause of low blood pressure in this case. This fluid shift occurs when the fluid moves from the blood vessels to the tissue spaces, such as in edema, inflammation, or increased capillary permeability. This reduces the blood volume and lowers the blood pressure.
Correct Answer is A
Explanation
Choice A: Elderly patients are at a higher risk for dehydration due to physiological changes that come with aging, such as decreased kidney function and physical changes to the body's water balance systems. Additionally, fever increases metabolic rate and fluid loss, and nausea and vomiting prevent adequate fluid intake, further increasing the risk of dehydration.
Choice B: While intentionally limiting fluid intake can lead to dehydration, the body's thirst mechanism in a healthy teenager is typically strong enough to prevent severe dehydration.
Choice C: Diarrhea can certainly lead to dehydration, but a young, otherwise healthy patient typically has a stronger ability to recover from fluid loss than an elderly patient.
Choice D: Infants are at a higher risk for dehydration than older children and adults due to their smaller body weight and higher turnover of water and electrolytes, but in this case, the elderly patient's multiple risk factors put them at a higher risk overall.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
