Which of the following properties of water explains its solvent abilities for certain substances?
Kinetic energy of liquid water molecules
High specific heat
High surface tension
Polarity of water molecules.
The Correct Answer is D
The polarity of water molecules explains its solvent abilities for certain substances.
Water is a polar molecule because it has a partial positive charge on one end and a partial negative charge on the other end due to the unequal sharing of electrons between the oxygen and hydrogen atoms.
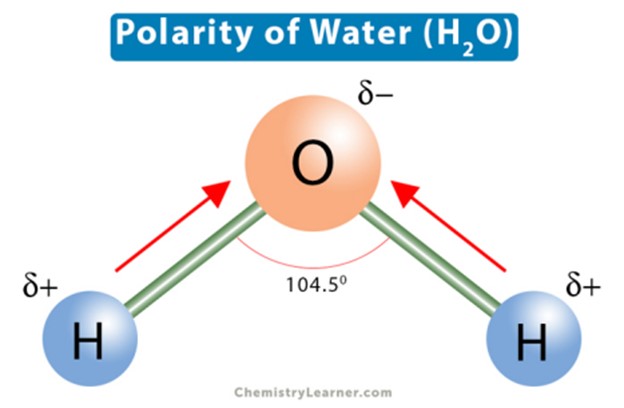
This polarity allows water to dissolve other polar substances and ionic compounds.
Choice A. Kinetic energy of liquid water molecules is not the correct answer because kinetic energy refers to the energy of motion and does not directly explain water’s solvent abilities.
Choice B. High specific heat is not the correct answer because specific heat refers to the amount of heat required to raise the temperature of a substance and does not directly explain water’s solvent abilities.
Choice C. High surface tension is not the correct answer because surface tension refers to the cohesive forces between liquid molecules and does not directly explain water’s solvent abilities.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is B
Explanation
The correct answer is choice B. 3’ TCGATCGCA 5’.
In DNA, the nitrogenous bases adenine (A) and thymine (T) pair together, while cytosine © and guanine (G) pair together.
Therefore, the complementary strand of the given DNA sequence “5’ AGCTAGCGT 3’” would have the nitrogenous bases T, C, G, A, T, C, G, C and A in that order.
Additionally, the 5’ to 3’ direction of the complementary strand is opposite to that of the given strand.
Choice A. 3’ AGCTAGCGT 5’ is not correct because it is not complementary to the given strand.
Choice C. 5’ UCGAUCGCA 3’ is not correct because it contains uracil (U), which is a nitrogenous base found in RNA, not DNA.
Choice D. 3’ TCGUTCGCU 3’ is not correct because it also contains uracil (U), which is a nitrogenous base found in RNA, not DNA.
Correct Answer is B
Explanation
Reverse transcriptase is a virus-specific enzyme that transcribes an RNA template to DNA1.
This allows the AIDS virus, which contains RNA, to insert viral DNA into the DNA of a host cell after the AIDS virus enters the cell.
Choice A, The phospholipids found on the envelope of the virus, is not the correct answer because phospholipids are a major component of cell membranes and do not play a direct role in inserting viral DNA into the DNA of a host cell.
Choice C, Receptor proteins located on the surface of the virus, is not the correct answer because receptor proteins located on the surface of the virus play a role in attachment and fusion of HIV virons to host cells2, but do not play a direct role in inserting viral DNA into the DNA of a host cell.
Choice D, The protein that makes up the capsid of the virus, is not the correct answer because capsid is the outer protein shell of a virus and does not play a direct role in inserting viral DNA into the DNA of a host cell.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
