After soaking for one hour in a solution of unknown concentration, a slice of potato appears to be very soft and limp. You can determine that the solution is
Hypertonic
Hypotonic
Isotonic
Tonic
The Correct Answer is A
Choice A rationale: A hypertonic solution has a higher solute concentration than the potato cell, which means it has a lower water potential. Water will move out of the potato cell by osmosis, causing it to shrink and become soft and limp.
Choice B rationale: A hypotonic solution has a lower solute concentration than the potato cell, which means it has a higher water potential. Water will move into the potato cell by osmosis, causing it to swell and become turgid and firm.
Choice C rationale: An isotonic solution has the same solute concentration as the potato cell, which means it has the same water potential. Water will move in and out of the potato cell at the same rate, causing it to remain unchanged in size and shape.
Choice D rationale: Tonic is not a valid term to describe the solute concentration of a solution. The correct terms are hypertonic, hypotonic, or isotonic.
Choice E rationale: I cannot determine anything without comparing multiple solutions is incorrect because the appearance of the potato slice after soaking in the solution provides enough information to determine the relative solute concentration of the solution.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is D
Explanation
Choice A rationale: 4x is incorrect because 4x is the lowest magnification objective lens that does not require oil to be applied. 4x is also called the scanning objective lens because it is used to scan the whole slide and find the specimen. 4x has a large field of view and a low resolution.
Choice B rationale: 10x is incorrect because 10x is the medium magnification objective lens that does not require oil to be applied. 10x is also called the low power objective lens because it is used to observe the specimen in more detail. 10x has a smaller field of view and a higher resolution than 4x.
Choice C rationale: 40x is incorrect because 40x is the high magnification objective lens that does not require oil to be applied. 40x is also called the high power objective lens because it is used to observe the specimen in greater detail. 40x has a smaller field of view and a higher resolution than 10x.
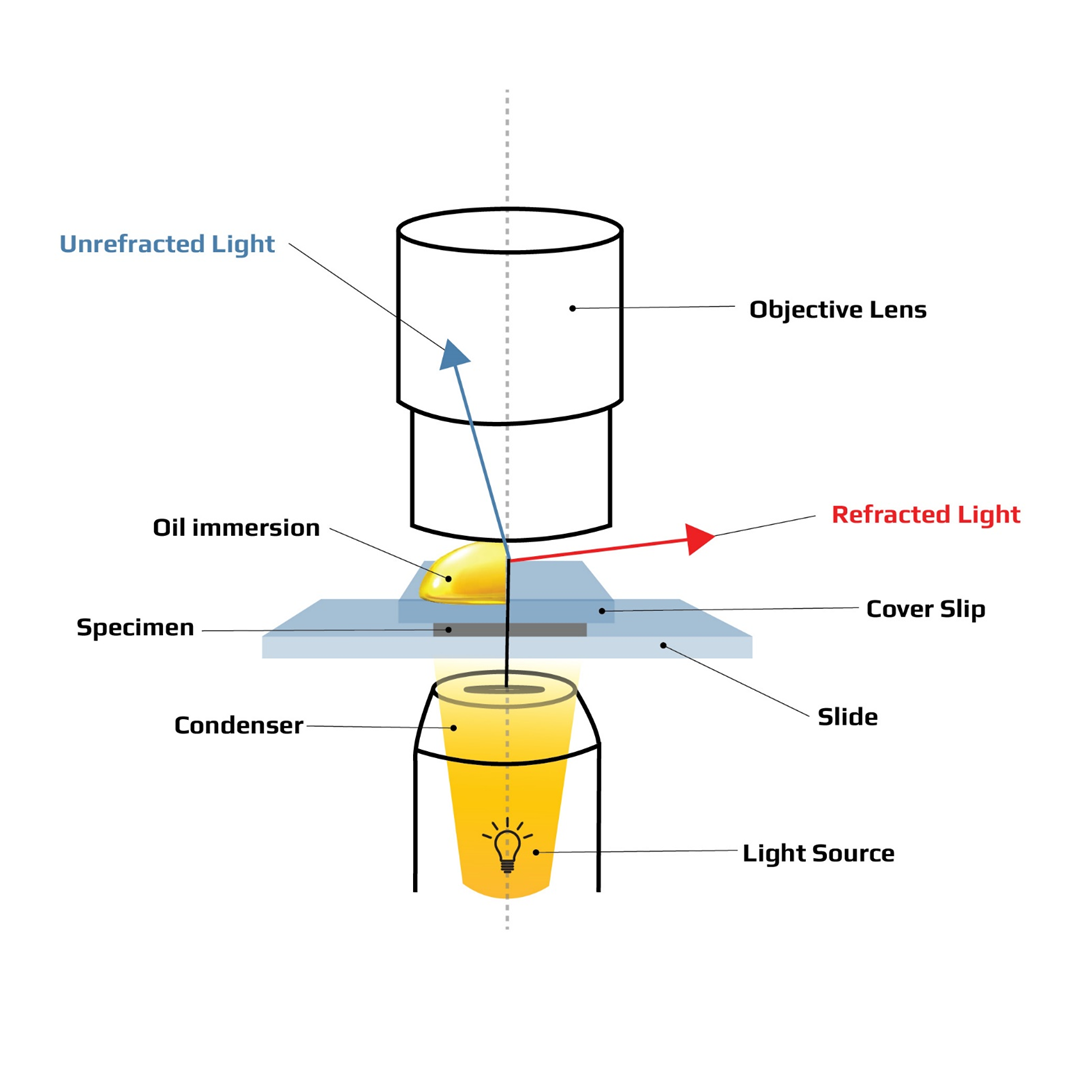
Choice D rationale: 100x is correct because 100x is the highest magnification objective lens that requires oil to be applied. 100x is also called the oil immersion objective lens because it is used to observe the specimen in the highest detail. 100x has a very small field of view and a very high resolution. Oil is applied between the slide and the lens to reduce the refraction of light and increase the clarity of the image.
Correct Answer is B
Explanation
Choice A rationale: High power is incorrect because high power is the second highest magnification objective lens, not the highest. High power is also called the 40x objective lens because it magnifies the specimen by 40 times. When combined with the 10x eyepiece lens, the total magnification is 400x.
Choice B rationale: Oil immersion is correct because oil immersion is the highest magnification objective lens. Oil immersion is also called the 100x objective lens because it magnifies the specimen by 100 times. When combined with the 10x eyepiece lens, the total magnification is 1000x. Oil immersion requires oil to be applied between the slide and the lens to reduce the refraction of light and increase the clarity of the image.
Choice C rationale: Low power is incorrect because low power is the second lowest magnification objective lens, not the highest. Low power is also called the 10x objective lens because it magnifies the specimen by 10 times. When combined with the 10x eyepiece lens, the total magnification is 100x.
Choice D rationale: Scanning is incorrect because scanning is the lowest magnification objective lens, not the highest. Scanning is also called the 4x objective lens because it magnifies the specimen by 4 times. When combined with the 10x eyepiece lens, the total magnification is 40x. Scanning is used to scan the whole slide and find the specimen.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
