What is the relationship between viruses and their host cells during replication?
Viruses use the host’s replication processes to produce progeny virus particles.
Viruses divide inside the host cell to produce progeny virus particles.
Viruses have their own replication processes and do not need the host cell.
Viruses infect the host cell and destroy it completely.
The Correct Answer is A
Viruses use the host’s replication processes to produce progeny virus particles.
Viruses are obligate intracellular parasites, meaning that they require a host cell to replicate.
They hijack the host cell’s machinery to produce new virus particles.
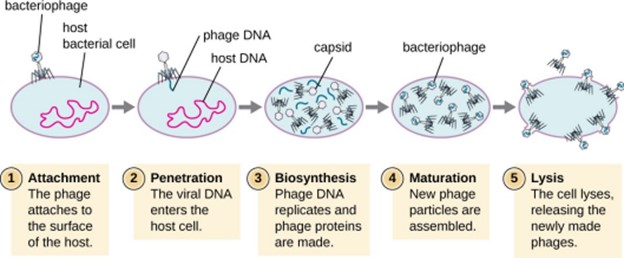
Choice B is incorrect because viruses do not divide inside the host cell.
Instead, they use the host cell’s machinery to produce new virus particles.
Choice C is incorrect because viruses do not have their own replication processes and rely on the host cell for replication.
Choice D is incorrect because while some viruses may destroy the host cell during replication, this is not always the case and is not the primary relationship between viruses and their host cells during replication.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
Hydrogen bonds are responsible for the unique properties of water and play a crucial role in the structure of DNA and proteins.
Hydrogen bonds are weak electrostatic attractions between a hydrogen atom covalently bonded to an electronegative atom and another electronegative atom.
Choice B.
Covalent bonds is incorrect because covalent bonds are strong chemical bonds formed by the sharing of electrons between two atoms.
Choice C.
Ionic bonds is incorrect because ionic bonds are chemical bonds formed by the transfer of electrons from one atom to another, resulting in the formation of ions.
Choice D.
Van der Waals forces is incorrect because Van der Waals forces are weak intermolecular forces that arise from temporary dipoles induced in atoms or molecules.
Correct Answer is A
Explanation
Hydrogen bonding is an interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons.
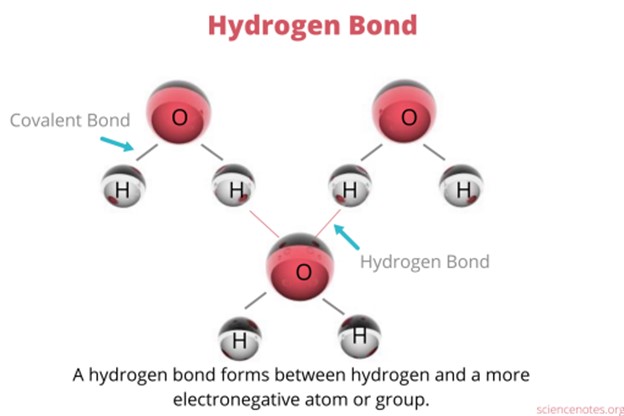
One atom of the pair (the donor), generally a fluorine, nitrogen, or oxygen atom, is covalently bonded to a hydrogen atom, whose electrons it shares unequally; its high electron affinity causes the hydrogen to take on a slight positive charge.
The other atom of the pair (the acceptor), also typically F, N, or O, has an unshared electron pair, which gives it a slight negative charge.
Mainly through electrostatic attraction, the donor atom effectively shares its hydrogen with the acceptor atom, forming a bond.
Choice B) The repulsion between the positive and negative charges of two molecules is incorrect because hydrogen bonding involves attraction, not repulsion.
Choice C) The attraction between two nonpolar molecules is incorrect because hydrogen bonding involves polar molecules.
Choice D) The attraction between two ionic molecules is incorrect because hydrogen bonding involves polar molecules and not ionic molecules.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
