A patient with a history of heart failure is prescribed a medication that increases urine output to reduce fluid buildup.
Which of the following statements best describes the mechanism of action of the prescribed medication?
Inhibits the renin-angiotensin-aldosterone system.
Blocks beta receptors.
Increases sodium and water reabsorption.
Enhances glomerular filtration rate.
The Correct Answer is D
The medication prescribed to the patient is a diuretic, which removes water and electrolytes from the body by increasing urination 1.
This helps reduce fluid buildup in the body.
Choice A, Inhibits the renin-angiotensin-aldosterone system, is not the correct answer because it describes a different mechanism of action.
Choice B, Blocks beta receptors, is not the correct answer because it describes a different mechanism of action.
Choice C, Increases sodium and water reabsorption, is not the correct answer because it would have the opposite effect of reducing fluid buildup.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is A
Explanation
Ionization is the process in which an atom loses or gains electrons to form an ion.
An ion is an atom or molecule that has a net electrical charge due to the loss or gain of one or more electrons.
Choice B is not the best answer because oxidation refers to the loss of electrons from an atom or molecule.
Choice C is not the best answer because reduction refers to the gain of electrons by an atom or molecule.
Choice D is not the best answer because isotopic decay refers to the process in which an unstable atomic nucleus loses energy by emitting radiation.
Correct Answer is A
Explanation
Hydrogen bonding is an interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons.
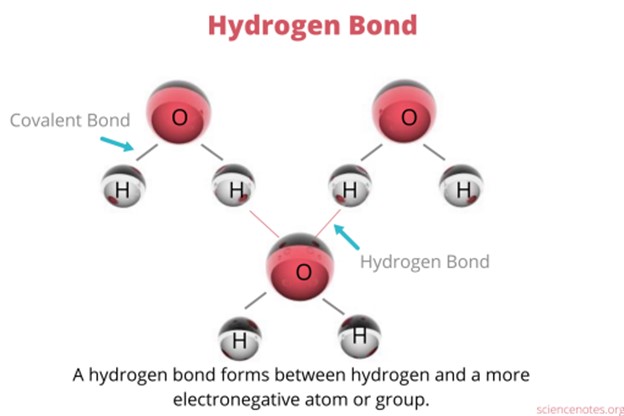
One atom of the pair (the donor), generally a fluorine, nitrogen, or oxygen atom, is covalently bonded to a hydrogen atom, whose electrons it shares unequally; its high electron affinity causes the hydrogen to take on a slight positive charge.
The other atom of the pair (the acceptor), also typically F, N, or O, has an unshared electron pair, which gives it a slight negative charge.
Mainly through electrostatic attraction, the donor atom effectively shares its hydrogen with the acceptor atom, forming a bond.
Choice B) The repulsion between the positive and negative charges of two molecules is incorrect because hydrogen bonding involves attraction, not repulsion.
Choice C) The attraction between two nonpolar molecules is incorrect because hydrogen bonding involves polar molecules.
Choice D) The attraction between two ionic molecules is incorrect because hydrogen bonding involves polar molecules and not ionic molecules.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
