In the following single-replacement reaction, ______ replaces ______.
Cl2+2NaI→2NaCl+I2
sodium, iodine
chlorine, iodine
chlorine, sodium
sodium, chlorine
Correct Answer : B
In this reaction, chlorine (Cl2) is an element in the reaction that replaces iodine in the compound sodium iodide (NaI). This allows chlorine to form a compound with sodium (NaCl) and leaves iodine (I2) as an element.
Synthesis reactions involve two or more reactants (A and B) combining to form one product (AB). In the example provided, hydrogen (H2) and oxygen (O2) begin as separate elements. At the end of the reaction, the hydrogen and oxygen atoms are bonded in a molecule of water (H2O).
Decomposition reactions have only one reactant (AB) that breaks apart into two or more products (A and B). In the example above, hydrogen peroxide (H2O2) breaks apart into two smaller molecules: water (H2O) and oxygen (O2).
Single-replacement reactions involve two reactants, one compound (AB) and one element (C). In this type of reaction, one element replaces another to form a new compound (AC), leaving one element by itself (B). In the example, zinc replaces hydrogen in hydrochloric acid (HCl). As a result, zinc forms a compound with chlorine, zinc chloride (ZnCl2), and hydrogen (H2) is left by itself.
Double-replacement reactions involve two reactants, both of which are compounds made of two components (AB and CD). In the example, silver nitrate, composed of silver (Ag1+) and nitrate (NO31-) ions, reacts with sodium chloride, composed of sodium (Na1+) and chloride (Cl1-) ions. The nitrate and chloride ions switch places to produce two compounds that are different from those in the reactants.
Combustion reactions occur when fuels burn, and they involve specific reactants and products, as seen in the examples below. Some form of fuel that contains carbon and hydrogen is required. Examples of such fuels are methane, propane in a gas grill, butane in a lighter, and octane in gasoline. Notice that these fuels all react with oxygen, which is necessary for anything to burn. In all combustion reactions, carbon dioxide, water, and energy are produced. When something burns, energy is released, which can be felt as heat and seen as light.
TEAS 7 Exam Quiz Bank
HESI A2 Exam Quiz Bank
Find More Questions 📚
Teas 7 Questions: We got the latest updated TEAS 7 questions
100% Money Refund: 100% money back guarantee if you take our full
assessment pass with 80% and fail the actual exam.
Live Tutoring: Fully customized live tutoring lessons.
Guaranteed A Grade: All students who use our services pass with 90%
guarantee.
Related Questions
Correct Answer is C
Explanation
Because more solute could be added and dissolve, the solution has not yet reached its limit and is considered unsaturated. Because all the solute dissolves, the particles in the mixture are evenly distributed as a homogenous mixture.
- Amixtureis when elements and compounds are physically, but not chemically, combined.
- Ahomogeneousmixture is when substances mix evenly and it is impossible to see individual components. Aheterogeneousmixture is when the substances mix unevenly and it is possible to see individual components.
- Asolutionis a type of homogeneous mixture that is formed when a solute dissolves in a solvent.
- The concentration of a solution is the amount of a substance in a given amount of solution. Anunsaturatedsolution has the ability to dissolve more solute and asaturatedsolution has already reached the limit of solute it can dissolve.
Correct Answer is B
Explanation
Nitrogen and oxygen are both nonmetals, which means they will share electrons in a covalent bond. For example, two oxygen atoms form a double bond, in which two pairs of electrons (four electrons total) are shared. Similarly, two nitrogen atoms form a molecule with a triple bond, in which three pairs of electrons (six electrons total) are shared.
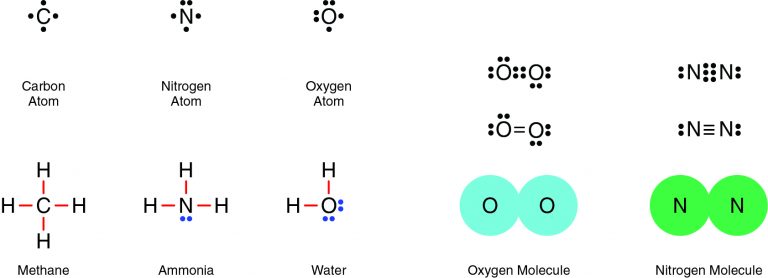
Correct Answer is B
Explanation
A cell copies its DNA during the S phase, and nucleotides are the building blocks of DNA. Thus, the step preceding the S phase, theG1phase, is the phase of the cell cycle when the cell would contain the most nucleotides.
For a cell to divide into more cells, it must grow, copy its DNA, and produce new daughter cells. Thecell cycleregulates cellular division. This process can either prevent a cell from dividing or trigger it to start dividing.
The cell cycle is an organized process divided into two phases:interphaseand theM (mitotic) phase. During interphase, the cell grows and copies its DNA. After the cell reaches the M phase, division of the two new cells can occur. The G1, S, and G2phases make up interphase.
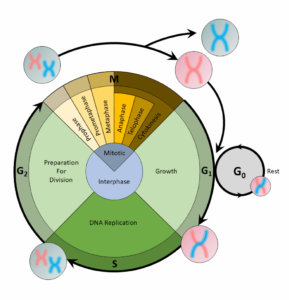
- G1:The first gap phase, during which the cell prepares to copy its DNA
- S:The synthesis phase, during which DNA is copied
- G2:The second gap phase, during which the cell prepares for cell division
It may appear that little is happening in the cell during the gap phases. Most of the activity occurs at the level of enzymes and macromolecules. The cell produces things like nucleotidesfor synthesizing new DNA strands, enzymes for copying the DNA, and tubulin proteins for building the mitotic spindle. During the S phase, the DNA in the cell doubles, but few other signs are obvious under the microscope. All the dramatic events that can be seen under a microscope occur during the M phase: the chromosomes move, and the cell splits into two new cells with identical nuclei.
Correct Answer is D
Explanation
In this reaction, two elements are trading places hence double-replacement. In the reactants, zinc and bromide ions are together, and potassium and hydroxide ions are together. In the products, zinc and hydroxide ions are together, and potassium and bromide ions are together.
Correct Answer is A
Explanation
Correct Answer is B
Explanation
A control group is a factor that does not change during an experiment. Due to this, it is used as a standard for comparison with variables that do change such as a dependent variable.
Recall that these make up thescientific method,described below:
- Problem:The question created because of an observation.Example: Does the size of a plastic object affect how fast it naturally degrades in a lake?
- Research:Reliable information available about what is observed.Example: Learn how plastics are made and understand the properties of a lake.
- Hypothesis:A predicted solution to the question or problem.Example: If the plastic material is small, then it will degrade faster than a large particle.
- Experiment:A series of tests used to evaluate the hypothesis. Experiments consist of anindependent variablethat the researcher modifies and adependent variablethat changes due to the independent variable. They also include acontrol groupused as a standard to make comparisons.
- Example: Collect plastic particles both onshore and offshore of the lake over time. Determine the size of the particles and describe the lake conditions during this time period.
- Observe:Analyze data collected during an experiment to observe patterns.
- Example: Analyze the differences between the numbers of particles collected in terms of size.
- Conclusion:State whether the hypothesis is rejected or accepted and summarize all results.
- Communicate:Report findings so others can replicate and verify the results.
Correct Answer is C
Explanation
Inductive reasoning involves making specific observations and using them to make broad statements. The student observes that all of the tigers have the same stripe pattern. He can use this observation to make the broad statement that all the tigers’ offspring will have the same stripe pattern.
Inductive reasoninginvolves drawing a general conclusion from specific observations. This form of reasoning is referred to as the “from the bottom up” approach. Information gathered from specific observations can be used to make a general conclusion about the topic under investigation. In other words, conclusions are based on observed patterns in data.
Correct Answer is B
Explanation
The number of protons, 28, gives the atomic number, which identifies this atom as nickel. The mass is the number after the dash in the isotope name, which is determined by adding the numbers of protons and neutrons (28 + 32 = 60).
Correct Answer is A
Explanation
Only plant cells have cell walls, which help protect the cell and provide structural support. The cell wall also enforces the overall structural integrity of the plant cell, and it is found outside the cell membrane. The next organelle is a chloroplast. It is found in the cytoplasm of only plant cells.Chloroplastsare photosynthetic compounds usedto make food for plant cells by harnessing energy from the sun. These organelles play a role in photosynthesis.
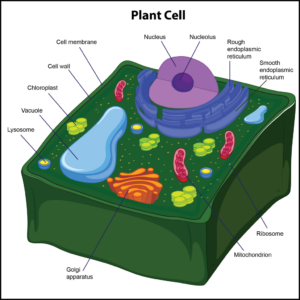
Correct Answer is D
Explanation
A neutralization reaction is a type of acid-base reaction where an acid and base react to form a salt and water.
In an aqueous solution, a base increases the hydroxide concentration (OH–), while an acid increases the hydrogen ion (H+) concentration. Sometimes,neutralization reactionsalso occur. This type of reaction happens when an acid and a base react with each other to form water and salt. Salt is typically defined as anionic compoundthat includes any cation except H+and any anion except OH–. Consider the following example of a neutralization reaction between hydrobromic acid (HBr) and potassium hydroxide (KOH).
HBr+KOH→KBr+H2O
Not all neutralization reactions proceed in the manner where all reactants are in the aqueous phase. In some chemical reactions, one reactant may be a solid. The neutralization reaction can still proceed to completion.
This question was extracted from the actual TEAS Exam. Ace your TEAS exam with the actual TEAS 7 questions, Start your journey with us today
Visit Naxlex, the Most Trusted TEAS TEST Platform With Guaranteed Pass of 90%.
Money back guarantee if you use our service and fail the actual exam. Option of personalised live tutor on your area of weakness.
