Which of the following substances is responsible for donating H+ ions to act as a buffer when blood pH rises?
Carbon dioxide.
Carbon monoxide.
Carbonic acid.
Oxygen.
The Correct Answer is C
In the human body, maintaining the pH of the blood within a narrow range is critical for proper physiological functioning.
One of the buffering systems that helps to regulate blood pH involves the conversion of carbon dioxide (CO2) and water (H2O) into carbonic acid (H2CO3), which then dissociates into hydrogen ions (H+) and bicarbonate ions (HCO3-).
Carbonic acid (H2CO3) is responsible for donating H+ ions to act as a buffer when blood pH rises.
When blood pH rises (becomes more alkaline), carbonic acid dissociates, and the H+ ions combine with bicarbonate ions to form more carbonic acid.
This helps to remove excess H+ ions from the blood and prevent the pH from rising too much.
Option A, carbon dioxide, is involved in the buffering system through its conversion to carbonic acid.
However, it does not directly donate H+ ions to act as a buffer when blood pH rises.
Option B, carbon monoxide, is a toxic gas that binds to hemoglobin in red blood cells, preventing them from carrying oxygen.
It is not involved in the buffering system and does not donate H+ ions.
Option D, oxygen, is carried by hemoglobin in red blood cells and is essential for respiration.
It is not involved in the buffering system and does not donate H+ ions.
Nursing Test Bank
Naxlex Comprehensive Predictor Exams
Related Questions
Correct Answer is B
Explanation
In this cross, the plant heterozygous for red flowers has the genotype Rr (where R represents the dominant red allele and r represents the recessive white allele), while the plant with white flowers has the genotype rr.
The possible offspring from this cross would have the genotypes Rr (red), Rr (red), rr (white), and rr (white), resulting in a 1:1 ratio of red to white flowers.
Correct Answer is B
Explanation
Calcium ions play a crucial role in initiating muscle contraction.
When a muscle cell is stimulated to contract by an action potential, calcium channels open in the sarcoplasmic membrane and release calcium into the sarcoplasm.
Some of this calcium attaches to troponin, which causes it to change shape. This shape change exposes binding sites for myosin on the actin filaments.
Myosin’s binding to actin causes cross-bridge formation, and muscle contraction begins.
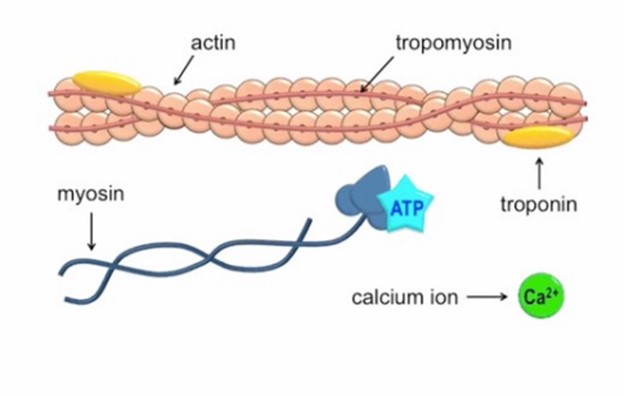
The other ions mentioned in the question do not have this specific role in muscle contraction.
Potassium ions are important for maintaining the resting membrane potential of cells, but they do not bind to the troponin complex.
Phosphorus ions are important for energy metabolism but do not bind to the troponin complex.
Sodium ions are important for generating action potentials but do not bind to the troponin complex.
Whether you are a student looking to ace your exams or a practicing nurse seeking to enhance your expertise , our nursing education contents will empower you with the confidence and competence to make a difference in the lives of patients and become a respected leader in the healthcare field.
Visit Naxlex, invest in your future and unlock endless possibilities with our unparalleled nursing education contents today
Report Wrong Answer on the Current Question
Do you disagree with the answer? If yes, what is your expected answer? Explain.
Kindly be descriptive with the issue you are facing.
